All products and services are For Research Use Only and CANNOT be used in the treatment or diagnosis of disease.
Developing effective non-viral cell therapies often confronts challenges such as transient transgene expression, complex viral manufacturing, and on-target/off-tumor toxicity in solid tumors. Creative Biolabs' CAR IVT circRNA platform is engineered to overcome these limitations by delivering durable, high-level CAR expression through a covalently closed RNA structure resistant to degradation. We offer an end-to-end service, from sequence design and proprietary scarless circularization to LNP formulation and functional validation, all supported by rigorous quality control. This integrated approach provides clients with a clinically translatable, target-agnostic solution that combines the safety of RNA with the persistence of viral systems, accelerating the path from discovery to development.
Conventional CAR-T therapies face manufacturing and safety hurdles. Circular RNA (circRNA) offers a transient, non-viral alternative with superior stability. This study demonstrates that electroporation of anti-DLL3 CAR circRNA into primary T cells generates highly potent effectors. These circRNA CAR-T cells exhibit enhanced cytotoxicity and achieve complete tumor eradication in SCLC models, outperforming mRNA-based counterparts and validating this platform for advanced cell therapies.
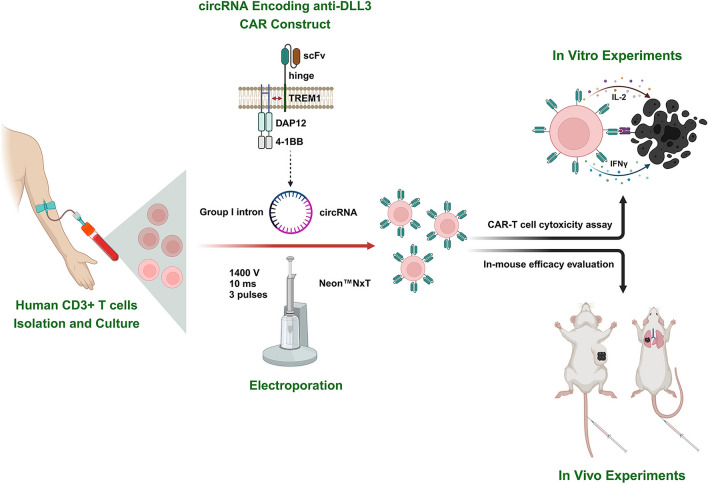 Fig.1 Circular RNA as a non-viral platform for DLL3-specific CAR-T cells in SCLC.1
Fig.1 Circular RNA as a non-viral platform for DLL3-specific CAR-T cells in SCLC.1
As a premier partner in advanced cell therapy development, Creative Biolabs offers an integrated, end-to-end platform for converting conventional mRNA or viral vector designs into highly stable circular RNA therapeutics. We deliver sequence-engineered, chromatography-purified circRNA constructs pre-formulated in cell-selective lipid nanoparticles (LNPs), optimized for direct functional evaluation in both cellular and animal models. Our proprietary system is engineered to address the intrinsic instability of linear RNA, enabling sustained transgene expression critical for overcoming the persistence barriers in solid tumor immunotherapy.
Our proprietary CAR IVT circRNA platform unites advanced sequence engineering, high-fidelity synthesis, and stringent functional assessment to generate high-potency, non-viral T cell therapies with durable activity.
Required Starting Materials:
Key Steps:
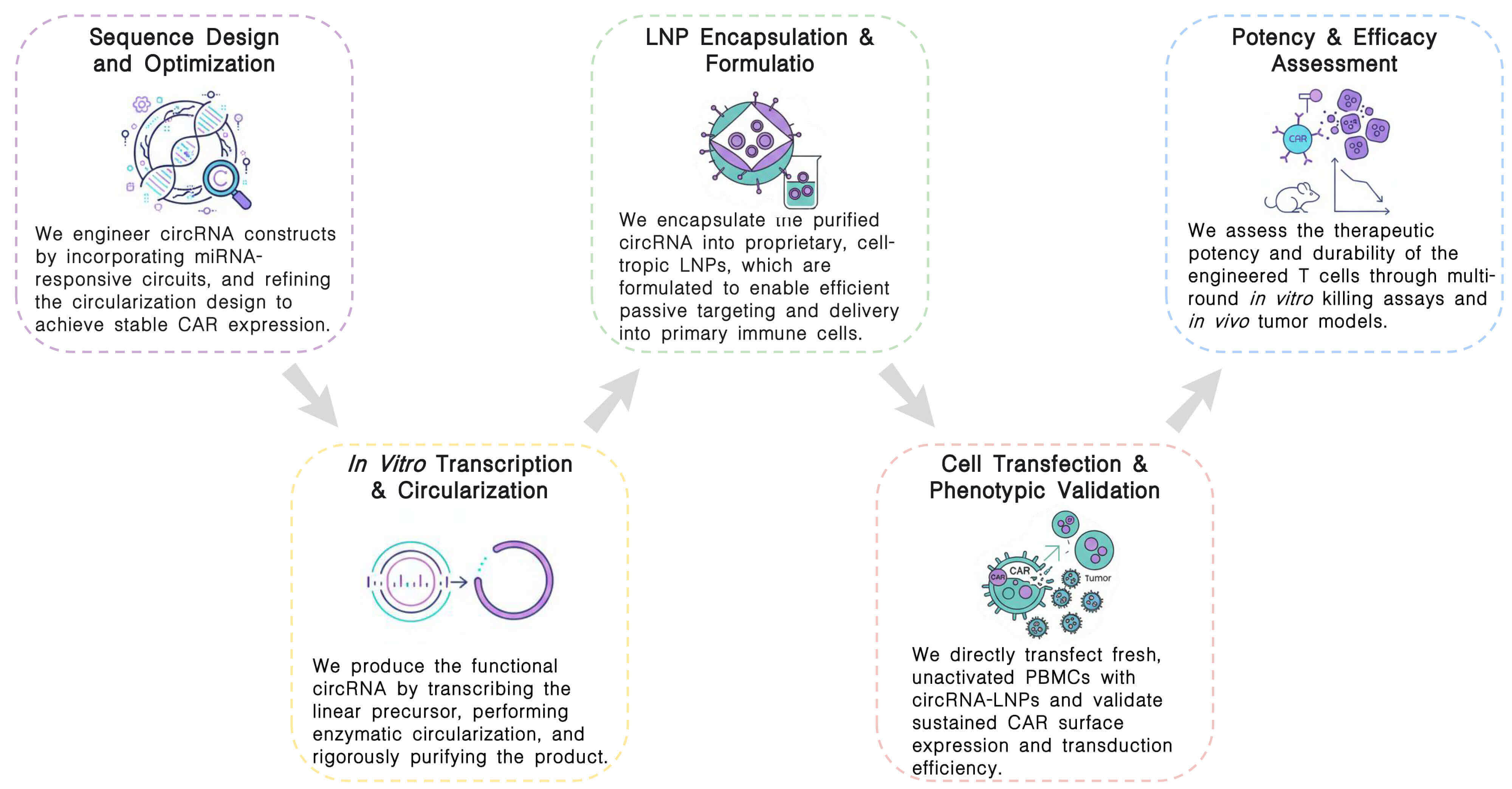
Final Deliverables:
How does circRNA compare to Lentivirus for CAR-T cell engineering?
Unlike lentiviral vectors, which integrate semi-randomly into the host genome and carry a theoretical risk of insertional mutagenesis, circRNA operates through a non-integrating, episomal mechanism. Its covalently closed, continuous structure confers exceptional resistance to exonuclease degradation, enabling viral vector-like durability of transgene expression without the associated genomic alteration risks. This makes it a favorable platform for applications requiring high safety profiles and sustained, but not permanent, expression.
Is the circularization process truly "scarless"?
Yes. Our proprietary circularization methodology utilizes a precise enzymatic ligation system. This process cleanly removes all exogenous scaffolding or splicing sequences, resulting in a seamless "scarless" junction within the final circRNA product. This junction is designed to be non-immunogenic and does not interfere with the open reading frame, ensuring the translation of an accurate, full-length functional protein, a critical factor for therapeutic efficacy and safety.
We offer a precision non-viral CAR-T platform that uniquely solves the core challenge of solid tumors: achieving durable and safe anti-tumor activity. Our patented IVT circRNA technology combines the high, sustained expression needed for efficacy with intelligent miRNA-sensing circuits for enhanced safety. Backed by peer-reviewed data, our platform is engineered to deliver the functional persistence required for clinical impact.
To accelerate the development and manufacturing of next-generation cell therapies, we offer a fully integrated suite of in vitro transcription materials and solutions. Our CAR IVT products series encompasses the entire workflow from gene cloning to final delivery, delivering high-quality, highly flexible, and extensively customizable core reagents. In addition to our CAR IVT circRNA products, we also offer a complementary suite including CAR IVT Plasmid Products, CAR IVT mRNA Products, and CAR IVT LNP Products, providing comprehensive support for diverse research, development, and manufacturing requirements across all stages.
To advance your CAR-T development program, our proprietary IVT circRNA platform offers a precise and potent solution. It is engineered to deliver durable, high-level transgene expression while maintaining an enhanced safety profile through its non-viral, non-integrating mechanism. Our dedicated team of PhD scientists collaborates closely with you to tailor constructs and optimize delivery strategies that directly align with your specific antigen target and accelerate your critical preclinical milestones. Connect us to discuss your project timeline and therapeutic objectives.
Reference
For any technical issues or product/service related questions, please leave your information below. Our team will contact you soon.
 NEWSLETTER
NEWSLETTER
The latest newsletter to introduce the latest breaking information, our site updates, field and other scientific news, important events, and insights from industry leaders
LEARN MORE NEWSLETTER NEW SOLUTION
NEW SOLUTION
CellRapeutics™ In Vivo Cell Engineering: One-stop in vivo T/B/NK cell and macrophage engineering services covering vectors construction to function verification.
LEARN MORE SOLUTION NOVEL TECHNOLOGY
NOVEL TECHNOLOGY
Silence™ CAR-T Cell: A novel platform to enhance CAR-T cell immunotherapy by combining RNAi technology to suppress genes that may impede CAR functionality.
LEARN MORE NOVEL TECHNOLOGY NEW SOLUTION
NEW SOLUTION
Canine CAR-T Therapy Development: From early target discovery, CAR design and construction, cell culture, and transfection, to in vitro and in vivo function validation.
LEARN MORE SOLUTION