All products and services are For Research Use Only and CANNOT be used in the treatment or diagnosis of disease.
Are you currently grappling with challenges such as prolonged drug development timelines, high recurrence rates in solid tumors, and safety concerns related to allogeneic T-cell therapies? Creative Biolabs' CAR-NK92 Cell Products are designed to address these issues by leveraging advanced engineering of NK-92 cells, a modular universal CAR architecture, and tailored manufacturing workflows. We provide precisely controllable and scalable solutions to facilitate more efficient clinical translation and enhance therapeutic performance.
The NK92 cell line is a human NK cell line characterized by its sustained proliferative capacity, and it serves as a critical immune effector widely utilized in cancer immunotherapy research. Through genetic engineering with chimeric antigen receptors (CARs), CAR-NK92 cells not only exhibit potent and specific tumor-killing activity but also offer distinct advantages such as "off-the-shelf" availability, enhanced safety profiles, and controllable cytotoxicity. Furthermore, by incorporating modular and programmable universal CAR designs, this platform enables multi-antigen targeting and flexible immune regulation, presenting a promising strategy to overcome tumor heterogeneity and prevent relapse in solid tumors.
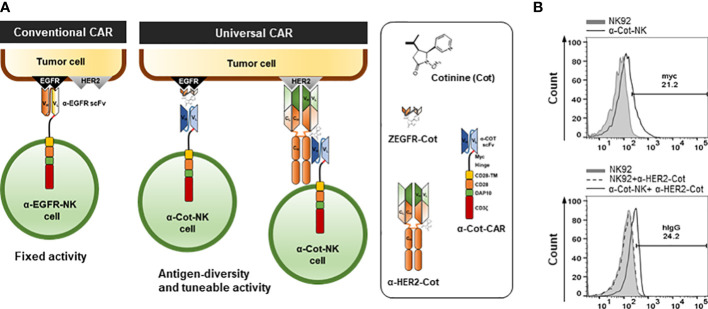 Fig.1 The α-Cot-NK92 cells: a universal chimeric antigen receptor system for adaptive immunotherapy.1
Fig.1 The α-Cot-NK92 cells: a universal chimeric antigen receptor system for adaptive immunotherapy.1
Creative Biolabs' CAR-NK92 Cell Products are designed to overcome key challenges in cell therapy, including antigen escape and manufacturing scalability. Utilizing the well-characterized NK-92 cell line as an off-the-shelf platform, we deliver targeted solutions against both solid and hematological malignancies. Our approach ensures enhanced safety, controllable activity, and accelerated clinical translation, positioning your pipeline for success with a versatile and cost-effective immunotherapy strategy.
Our integrated CAR-NK92 platform delivers a comprehensive solution, combining advanced cellular engineering, scalable industry-standard manufacturing, and rigorous quality systems to ensure reliable, off-the-shelf cell products with validated efficacy and safety.
Our CAR-NK92 manufacturing follows a systematic process from cell banking to cryopreserved final product, ensuring standardized, scalable, and quality-assured "off-the-shelf" therapeutics for reliable application.
Required Starting Materials: Our process initiates with a stable NK92 cell line and a validated CAR construct.
Key Steps:
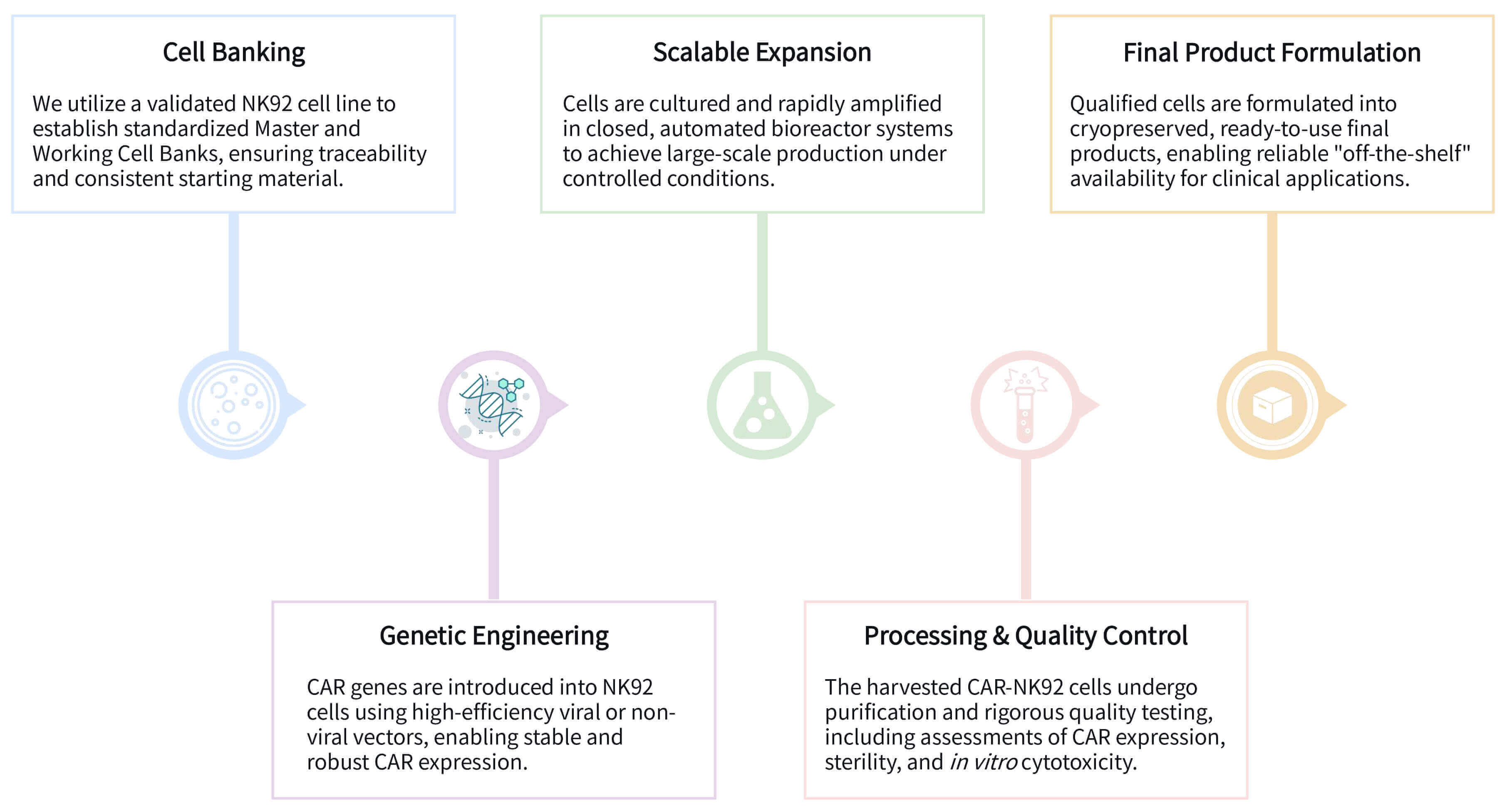
Final Deliverables: We provide cryopreserved, off-the-shelf CAR-NK92 cell products, fully characterized for CAR expression, sterility, and cytotoxic potency, ready for application.
What are the key distinctions between CAR-NK92 and CAR-T cell therapies?
CAR-NK92 fundamentally differs from autologous CAR-T through its allogeneic NK-92 cell source, enabling true "off-the-shelf" use. It presents a superior safety profile with minimal risk of severe CRS or GVHD, and offers scalable, cryopreserved manufacturing, eliminating patient-specific production delays.
What advantages does the NK92 cell line offer over primary NK cells?
The NK92 cell line exhibits unlimited in vitro proliferative capacity, providing a consistent and scalable cell source ideal for standardized industrial production. In contrast, primary NK cells face significant challenges in achieving large-scale expansion while maintaining functional uniformity, making NK92 a more reliable and practical platform for reproducible therapeutic development.
Choose our CAR-NK92 platform for a strategically superior cell therapy solution. We overcome critical barriers in oncology—antigen escape and safety concerns—through a universal CAR system enabling modular targeting and controllable activity. Combined with intrinsic NK cell safety and scalable off-the-shelf manufacturing, we deliver both therapeutic precision and commercial viability for your pipeline.
"The strategic decision to leverage Creative Biolabs' CD3-specific CAR-NK92 utility cells transformed our allogeneic CAR-T manufacturing. It provided the dual benefit of driving TCR+ impurities below the 0.01% threshold, which standard techniques cannot achieve, and amplifying final product yield by 300%, thereby drastically improving our cost structure." S**a M**n.
"Facing heterogeneous relapse, Creative Biolabs' universal CAR-NK92 products provided a decisive solution. The unique capability to dynamically switch targets mid-study outperformed conventional CARs and revealed a straightforward, adaptable path for developing effective combination therapies." Dr. J***s.
"We were concerned about off-tumor toxicity for our high-density target. Creative Biolabs' CAR-NK92 cells, leveraging the NK cell's intrinsic HLA-I modulation, provided a necessary safety buffer. Their fine-tuning of the CAR design ensured that only high-antigen-density malignant cells were targeted, sparing non-malignant tissue." Dr. R***r A**d.
Select Creative Biolabs to access a comprehensive CAR-NK92 platform designed to overcome the core challenges in cell therapy. We deliver validated, off-the-shelf cell products with modular targeting to overcome antigen escape, engineered safety controls for enhanced precision, and scalable manufacturing for reliable supply. Let us help you accelerate your oncology program's path. Contact our team to explore a tailored collaboration.
Reference
Associated Antigen Target
For any technical issues or product/service related questions, please leave your information below. Our team will contact you soon.
 NEWSLETTER
NEWSLETTER
The latest newsletter to introduce the latest breaking information, our site updates, field and other scientific news, important events, and insights from industry leaders
LEARN MORE NEWSLETTER NEW SOLUTION
NEW SOLUTION
CellRapeutics™ In Vivo Cell Engineering: One-stop in vivo T/B/NK cell and macrophage engineering services covering vectors construction to function verification.
LEARN MORE SOLUTION NOVEL TECHNOLOGY
NOVEL TECHNOLOGY
Silence™ CAR-T Cell: A novel platform to enhance CAR-T cell immunotherapy by combining RNAi technology to suppress genes that may impede CAR functionality.
LEARN MORE NOVEL TECHNOLOGY NEW SOLUTION
NEW SOLUTION
Canine CAR-T Therapy Development: From early target discovery, CAR design and construction, cell culture, and transfection, to in vitro and in vivo function validation.
LEARN MORE SOLUTION