Choosing the Right Method for Dermal Delivery:
Franz Diffusion Cells vs. Dialysis
Struggling to decide whether Franz diffusion cells or dialysis will generate more credible dermal delivery data? Learn which method answers which question, where each can mislead, and how to build a stronger, defensible evaluation strategy for topical liposomal formulations.
The Analytical Dilemma for Formulation Developers
For topical and transdermal formulation scientists, preclinical researchers, and biotech project managers, navigating the human skin barrier requires highly optimized lipid-based nanocarriers. However, generating reproducible, defensible data for these vehicles presents a persistent challenge.
Whether you are selecting a method for early-stage formulation screening, generating data for high-impact publication, comparing free drug versus liposomal formulations, or outsourcing topical delivery testing, you face a critical dilemma: Which methodology provides the most actionable and credible data?
The industry relies heavily on two techniques: the dialysis bag method and Franz diffusion cells. Frequently, researchers treat these as interchangeable tools or fall into the trap of confusing "release" with "permeation/retention." This misunderstanding often leads to data that is rejected by reviewers, questioned by regulatory bodies, or worse, results in flawed formulation decisions when engineering liposomes for dermal delivery.
What Makes a Dermal Delivery Release Profile Credible?
A credible release profile is not just a smooth curve on a graph. To produce data that withstands rigorous academic and regulatory scrutiny, your chosen methodology must clearly define its scope and limitations.
Key Evaluation Criteria
- Method-to-Question Match: Does the method directly answer whether the drug leaves the liposome, or whether the drug penetrates the stratum corneum?
- Distinguishing Drug States: Can the assay reliably differentiate between the unencapsulated (free) drug and the carrier-associated drug?
- Endpoint Separation: Are *release*, *permeation*, and *skin retention* treated as distinct, independent endpoints rather than synonymous terms?
Common Methodological Pitfalls
- Sink Conditions: Failure to maintain adequate sink conditions, especially for highly lipophilic APIs, leading to artificially truncated release curves.
- Membrane Resistance: Overlooking the artificial resistance generated by dialysis cellulose or the extreme variability of biological tissue in Franz cells.
- Stability Issues: Ignoring potential API degradation within the receptor medium over extended sampling periods.
Addressing these factors is at the core of robust formulation analysis and characterization.
Dialysis Method: Mapping Intrinsic Carrier Release
The dialysis method isolates the dissociation of the drug from its nanocarrier. Utilizing a specific Molecular Weight Cut-Off (MWCO), the dialysis sac retains the intact liposomes while allowing the free API to diffuse into a vast receptor medium driven purely by thermodynamic concentration gradients.
For an in vitro release kinetics analysis, dialysis is the gold standard to identify burst release phenomena, confirm structural integrity, and model sustained release mathematical profiles.
Crucial Limitations & Caveats
Despite its utility, dialysis is strictly an *in vitro* tool with specific boundaries:
- Not a Skin Proxy: A cellulose membrane lacks the complex lipid matrix, cellular architecture, and enzymes of the epidermis. It measures *diffusion*, not biological *permeation*.
- The Membrane Bottleneck: For highly lipophilic or large APIs, the dialysis membrane itself can become the rate-limiting step, causing researchers to mistake "slow membrane crossing" for "slow carrier release."
- Formulation Blind Spots: Dialysis cannot evaluate whether formulation excipients (e.g., penetration enhancers) will successfully fluidize human skin lipids.
The Rule of Dialysis
"Dialysis confirms that a liposome can release its payload; it does absolutely nothing to confirm if that payload will successfully navigate biological skin."
Franz Diffusion Cells: Establishing Dermal Relevance
When moving closer to clinical reality, Franz diffusion cells introduce the biological barrier. Equipped with ex vivo human/porcine skin or synthetic skin constructs (e.g., Strat-M®), this apparatus evaluates how lipid nanoparticles interact with the stratum corneum, capturing vesicle fusion, appendageal transport, and epidermal partitioning.
Data from Franz cells—quantified as cumulative transdermal permeation or localized skin retention—provides unparalleled insight into the bio-fate of the topical application, making it indispensable for formulation efficacy prediction.
Crucial Limitations & Caveats
Franz cell data is powerful but highly sensitive to experimental design:
- Membrane Dependency: Results vary wildly depending on whether you use human epidermis, full-thickness porcine skin, or synthetic mimics.
- Cumulative Amount ≠ Release: The concentration measured in the receptor chamber represents the drug that successfully permeated the *entire* skin thickness. It is an amalgamation of carrier release, skin partitioning, and diffusion—not a pure carrier release curve.
- Operational Sensitivity: Skin integrity, thickness, temperature control, and receptor fluid composition (especially the addition of BSA or surfactants for lipophilic drugs) drastically alter outcomes.
The Rule of Franz Cells
"Franz cells evaluate whether the formulation interacts effectively with a biological barrier to facilitate permeation or retention—but they obscure the intrinsic release mechanism of the nanocarrier itself."
The Synergistic Strategy for Defensible Data
The conclusion isn't a simple binary choice. High-impact research and IND-enabling studies require a strategic integration:
- Use Dialysis when the key question is release from the carrier.
- Use Franz cells when the key question is skin permeation or retention.
- Use Both when you need a credible, publishable evaluation package.
To illustrate this distinction, examine a comprehensive study investigating the delivery of Licochalcone A (LA) utilizing skin keratin liposomes.
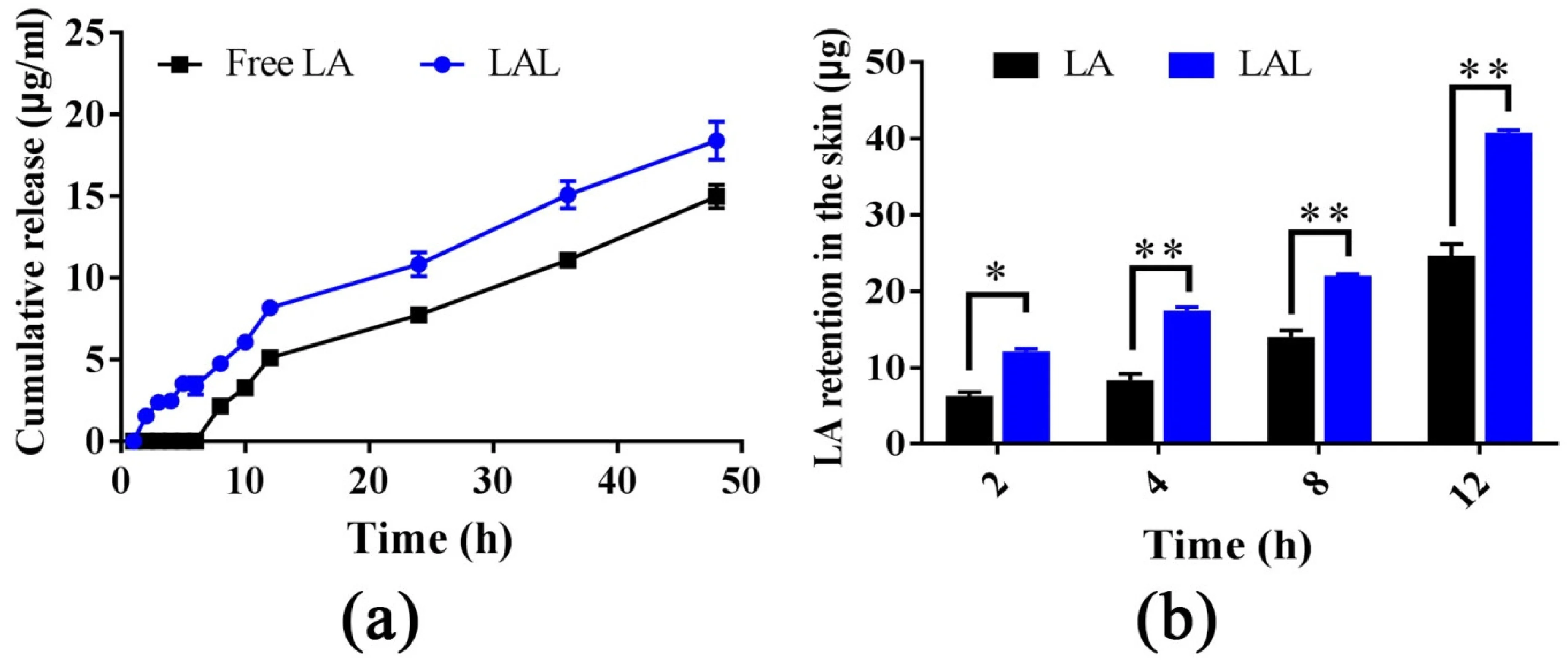
Figure 1a showcases the release profile of LA-loaded skin keratin liposomes mapped under dialysis conditions, explicitly defining the sustained release capability of the lipid architecture. Conversely, Figure 1b visualizes the *skin retention* results of the identical formulation utilizing Franz diffusion cells equipped with an actual dermal barrier.
Placing these panels side-by-side validates a foundational principle: how a formulation releases its payload in a simple dialysis setup does not automatically equate to how it will deposit within the skin. A formulation might exhibit rapid dialysis release but fail to penetrate the stratum corneum, remaining useless on the skin surface. Conversely, a formulation with ultra-slow dialysis release might exhibit superior skin interaction, fluidizing the epidermal lipids to facilitate deep dermal deposition.
Mapping isolated release (dialysis) against functional permeation (Franz cells) is mandatory. Furthermore, understanding how these variables shift over time is critical for formulation stability monitoring services, ensuring the final product remains therapeutically active.
Method Selection Guide
Quickly identify the correct analytical approach based on your specific formulation development question.
| Your Key Question | Primary Readout | Recommended Method |
|---|---|---|
| Is the API released from the carrier over time? | Release profile | Dialysis |
| Does the formulation improve passage across the skin barrier? | Permeation / flux | Franz diffusion cells |
| Does the drug remain within the skin layers? | Skin retention / deposition | Franz + skin extraction analysis |
| Do I need mechanistic and application-relevant evidence? | Release + permeation/retention | Dialysis + Franz |
Need help choosing the right dermal delivery evaluation workflow?
Avoid costly data rejection. Our scientists can help you design a fit-for-purpose strategy covering *in vitro* release testing, Franz diffusion cell studies, skin retention analysis, and complete formulation characterization for liposomal topical products.
Frequently Asked Questions
To truly compare efficacy, Franz diffusion cells are superior. While dialysis can show that the free drug diffuses faster than the encapsulated drug (which is expected), Franz cells reveal the biological value of the liposome—specifically, whether the lipid carrier enhances the penetration of the drug into the skin layers compared to a simple free drug solution.
Yes, Franz cells are highly suitable for semisolid formulations. They are routinely used to evaluate gels, creams, and ointments containing nanoparticles. The formulation is applied to the donor chamber (open or occluded), mimicking actual patient application, allowing researchers to measure how the complex rheology of the gel interacts with the release and subsequent skin permeation of the nanocarriers.
For early-stage, high-throughput screening of numerous formulation candidates, synthetic membranes like Strat-M® are highly recommended. They offer excellent reproducibility, no biological variability, and bypass ethical constraints. Once the top 1 or 2 candidate formulations are selected, the study should be repeated using ex vivo porcine ear skin or human cadaver skin for definitive, physiologically relevant data prior to in vivo studies.
Increasingly, yes. High-impact pharmaceutical and nanomaterials journals generally expect researchers to present a complete mechanistic picture. Reviewers often require dialysis data to prove the liposomes successfully and sustainably release the drug, accompanied by Franz diffusion cell data (along with skin retention mapping) to prove the formulation has tangible application value for transdermal or topical delivery. Relying solely on one method frequently leads to major revision requests.
Reference
- Wu, Wenfeng, et al. "Mechanisms of penetration enhancement and transport utilizing skin keratine liposomes for the topical delivery of licochalcone A." Molecules 27.8 (2022): 2504. https://doi.org/10.3390/molecules27082504
- Under Open Access license CC BY 4.0, without modification.
Online Inquiry
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.


