Key CQAs for Liposomal Skin Delivery:
Stability, Loading and Irritation
A practical guide to defining particle size, PDI, zeta potential, encapsulation and irritation-related acceptance criteria for liposomal dermal and transdermal formulations.
The Challenge of Dermal Liposome Development
Liposomal skin delivery sits at the critical intersection of barrier penetration, physical stability, and biological tolerability.
Many teams can generate basic liposomal suspensions, but frequently struggle to define defendable Critical Quality Attributes (CQAs) for size distribution, surface charge, loading efficiency, and irritation risk. Transitioning from an exploratory bench-top formulation to a reproducible clinical candidate demands rigorous analytical targets.
This resource outlines how to set fit-for-purpose CQA ranges and which analytical methods support development decisions for dermal and transdermal products, empowering formulators to navigate the scaling and validation processes seamlessly.
Target Audience
This resource is designed for dermal and transdermal formulation scientists, CMC/formulation development managers, preclinical translation teams, and cosmeceutical R&D groups developing liposomal products that require reproducible performance, defensible specifications, and low irritation risk.
Core Pain Points Addressed
Resolving common bottlenecks including unstable size distribution during storage, inconsistent encapsulation across batches, unclear zeta potential targets, lacking target ranges/release logic, and the difficulty of balancing deeper penetration with acceptable skin tolerability.
Why Skin Delivery Requires Project-Specific CQAs
The stratum corneum acts as a highly effective, impermeable biological barrier. While liposomes can facilitate drug penetration via intercellular, transcellular, and transappendageal pathways, their success depends entirely on meticulous formulation engineering. You cannot apply generic parenteral liposome specifications to a topical gel.
A robust Quality by Design (QbD) framework is imperative to ensure physical stability, colloidal integrity, and storage robustness throughout development and shelf-life. Formulators must establish criteria that are uniquely tailored to their clinical intent:
- For local dermal deposition: prioritize physical stability, narrow size distribution, moderate skin retention, and low irritation risk.
- For deeper transdermal flux: smaller vesicles and stronger permeation may be beneficial, but irritation thresholds and systemic exposure risk should be strictly tightened.
Transforming formulation data into defendable CQA specifications requires rigorous analytical strategy. Creative Biolabs provides comprehensive Formulation Analysis and Characterization services to support data-driven decision-making.
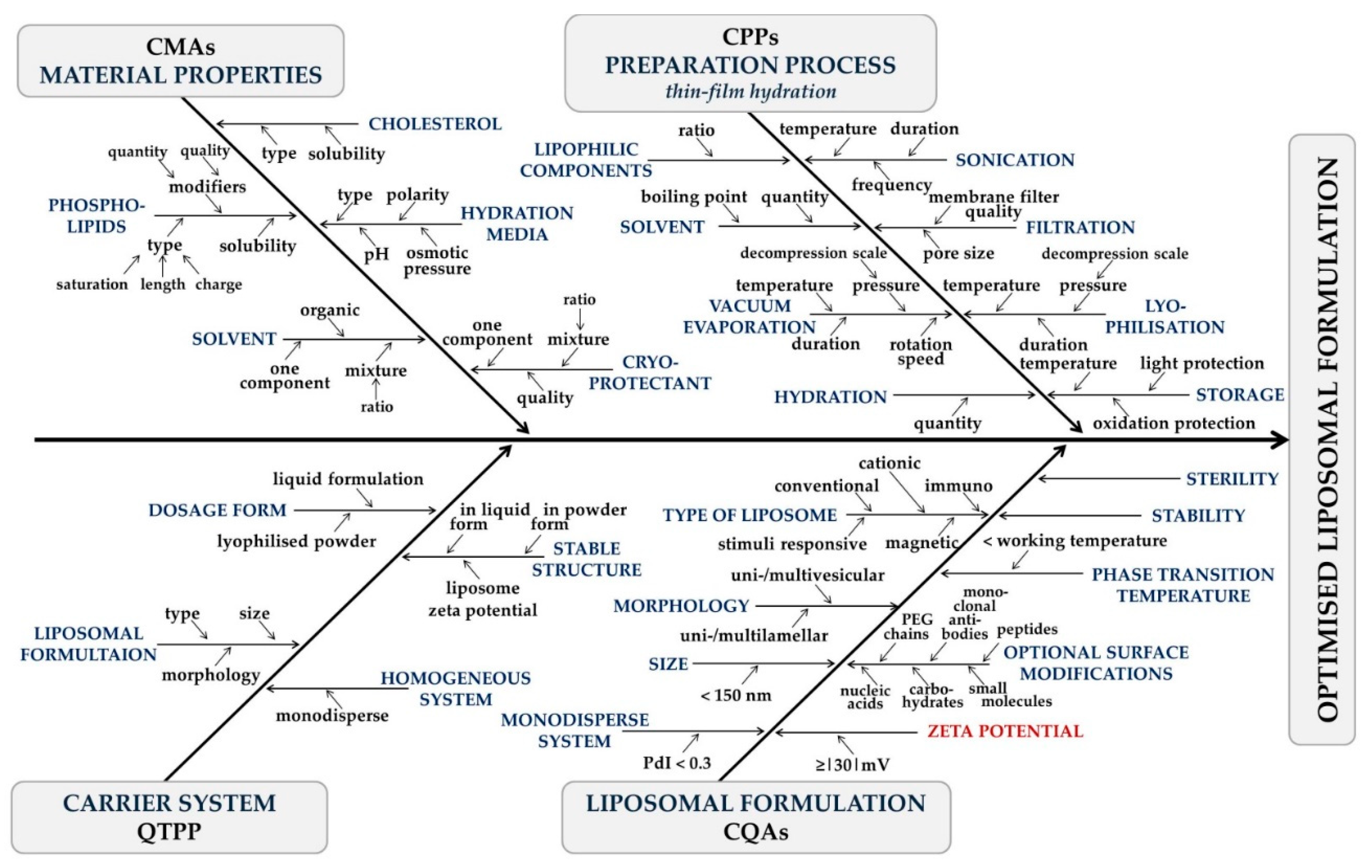
How to Set Particle Size and PDI Targets
The spatial dimensions of a liposome dictate its interaction with the complex architecture of the skin. However, strict universal cutoffs do not exist; specifications must be driven by the intended penetration pathway.
Particle Size Specification Logic
-
Larger Vesicles (>300 nm): Commonly preferred for forming a localized, protective depot on the stratum corneum, supporting sustained superficial drug release rather than deep penetration.
-
Sub-200 nm Range: Vesicles in this range are often explored for deeper epidermal or follicular delivery, although actual penetration behavior remains formulation- and skin-model-dependent.
Polydispersity Index (PDI)
A high PDI indicates a heterogeneous population, leading to erratic release kinetics and dramatically reducing formulation stability due to Ostwald ripening.
A PDI below 0.3 is commonly used as a development target for acceptable size homogeneity, while narrower distributions (closer to 0.1) may be preferred when reproducibility and stringent storage stability are critical.
How to Define Zeta Potential for Colloidal Stability
Zeta potential—the electrostatic potential at the hydrodynamic shear plane—dictates the balance between attractive Van der Waals forces and repulsive electrostatic forces within colloidal dispersions (DLVO theory).
As a development heuristic, absolute zeta potential values around or above 30 mV are often associated with improved electrostatic stabilization. However, acceptable ranges should be justified against formulation composition, ionic strength, steric stabilization strategy (e.g., PEGylation), and intended storage medium, rather than treated as an absolute universal rule.
A pivotal study by Németh et al. utilized a QbD-driven approach to map zeta potential leveraging charge-imparting membrane additives. Their research established that optimizing molar ratios of anionic/cationic lipids directly modulates both macroscopic shelf-life and microscopic interactions with the skin barrier. For instance, mildly cationic liposomes frequently exhibit enhanced electrostatic adhesion to the negatively charged stratum corneum, promoting prolonged residence time, though excessive positive charge must be constrained to limit cellular toxicity.
To decode the phase behavior and structural nuances of your lipid bilayers under diverse environmental stresses, explore our Structure and Composition Analysis services.
Encapsulation Efficiency vs Drug Loading: What Should Be Controlled?
Setting a generic "EE > 80%" benchmark is a common pitfall. Targets must be fundamentally justified by the Active Pharmaceutical Ingredient (API) properties.
For Hydrophilic APIs
Hydrophilic compounds sequestered within the aqueous core inherently face lower EE% limits due to the constrained internal volume of small vesicles. Loading targets should be justified against dose feasibility and therapeutic requirement rather than generic high-percentage benchmarks. Active loading techniques (e.g., pH gradients) may be necessitated.
For Lipophilic APIs
Lipophilic drugs reside within the acyl hydrocarbon chains. Here, the critical control strategy shifts toward defining a maximum drug loading capacity. Overloading the bilayer risks profound membrane destabilization, premature drug crystallization during storage, and unregulated burst release upon topical application.
| Critical Quality Attribute | Target Specification Logic | Analytical Technique |
|---|---|---|
| Particle Size | Formulation-dependent (e.g., sub-200nm for deep penetration) | DLS / NTA / Cryo-TEM for orthogonal confirmation |
| Polydispersity Index (PDI) | < 0.3 (Development target for homogeneity) | Dynamic Light Scattering (DLS) |
| Zeta Potential | Justified by composition, typically around ±30 mV | Electrophoretic Light Scattering / Laser Doppler Electrophoresis |
| Encapsulation & Loading | Project-specific target defined by API properties & dose requirement | HPLC / UPLC / Dialysis Methods |
How to Establish Irritation-Related Screening Thresholds
Biocompatibility represents a paramount safety CQA for any dermatological formulation. While basal liposomal constituents (e.g., phosphatidylcholines) are intrinsically benign, formulation trigger points—such as high ethanol content, cationic lipids, synthetic surfactants, extreme pH, or potent penetration enhancers—can readily induce erythema and edema.
For early screening, irritation-related CQAs should be translated into measurable decision gates, rather than being treated as a purely descriptive safety endpoint at the end of development.
- Primary Screen (RhE Viability Threshold): In vitro Reconstructed Human Epidermis (RhE) models, guided by OECD TG 439, use tissue viability (often ≤50%) as a hazard-screening threshold. Formulations causing viability to drop below this screening gate are flagged as potential irritants early on.
- Supportive Markers: Beyond binary viability, monitoring pro-inflammatory cytokine release (e.g., IL-1α) and integrating TEER/TEWL (Transepidermal Water Loss) assessments in ex vivo compatible workflows provides a nuanced tolerability profile.
- Project-Specific Threshold Setting: For cosmetics and chronic topical therapeutics, internal acceptance criteria are often far stricter than a generic "not-classified" OECD cutoff.
Analytical Toolbox for CQA Establishment
Achieving a compliant, effective, and stable liposomal skin product demands holistic analysis. Trust the experts to provide precise analytical data spanning physical, chemical, and biological attributes to refine your candidate prior to scale-up.
Lipid-based Basic CharacterizationFrequently Asked Questions
Initial specifications should be driven by the target deposition site. If your goal is deep epidermal or follicular delivery, target a sub-200 nm range. If you intend to form a surface depot, larger multilamellar vesicles (>300 nm) are appropriate. For PDI, a value below 0.3 is a standard development target to ensure population homogeneity and mitigate Ostwald ripening, though stricter limits (closer to 0.1) may be required depending on scale-up robustness and release kinetics.
No, it is not an absolute rule. While absolute zeta potential values around or above 30 mV are often used as an initial stability heuristic indicating strong electrostatic repulsion, stability is multifaceted. Acceptable ranges must be justified against the specific formulation composition, ionic strength of the buffer, presence of steric stabilizers (like PEGylated lipids), and the intended storage medium.
Yes, distinguishing between the two is critical. Encapsulation Efficiency (EE%) indicates the proportion of API successfully entrapped versus the total input, which is vital for process economics. Drug loading capacity defines the mass of API relative to the lipid matrix. For lipophilic drugs, defining a maximum drug loading capacity is crucial to prevent bilayer destabilization and crystallization, even if EE% appears exceptionally high.
For early nonclinical decision-making, in vitro Reconstructed Human Epidermis (RhE) models are employed. A common hazard-screening threshold (e.g., OECD TG 439) flags formulations causing tissue viability to drop to ≤50% as potential irritants. However, for chronic topical products or cosmetics, internal acceptance criteria should integrate supportive markers (like IL-1α release) and be set much stricter than generic regulatory cutoffs.
A robust candidate comparison requires an integrated analytical package that assesses physical stability (DLS for Size/PDI, Electrophoretic Light Scattering for Zeta Potential), chemical loading (HPLC/UPLC for EE% and Drug Loading), and structural integrity. A comprehensive Lipid-based Basic Characterization service enables data-driven down-selection before advancing to expensive in vivo or ex vivo skin permeation studies.
Reference
- Németh, Zsófia, et al. "Quality by design-driven zeta potential optimisation study of liposomes with charge imparting membrane additives." Pharmaceutics 14.9 (2022): 1798. https://doi.org/10.3390/pharmaceutics14091798
- Under Open Access license CC BY 4.0, without modification.
Online Inquiry
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.


