How to Develop Targeted Liposomes:
From Target Selection to Cellular Validation
This resource outlines a practical workflow for targeted liposome development, helping researchers move from target selection and ligand choice to conjugation design and cellular uptake/colocalization validation.
Bridging the Gap: Why This Integrated Workflow Matters
Developing a targeted liposome requires more than simply attaching a targeting ligand to a lipid surface. The success of targeted delivery depends fundamentally on the seamless integration of target biology, ligand format selection, surface conjugation chemistry, and the rigorous design of subsequent cellular validations.
When these critical phases are disconnected, drug developers frequently encounter perplexing setbacks. Particles may bind effectively but fail to internalize; they might internalize but demonstrate poor target specificity; or they may yield imaging data that is difficult to interpret logically. The true power of active targeting lies in the synergistic principles of ligand-mediated targeting and receptor-mediated endocytosis. If a ligand simply anchors the nanoparticle to the cell without triggering uptake, the therapeutic payload remains stranded extracellularly.
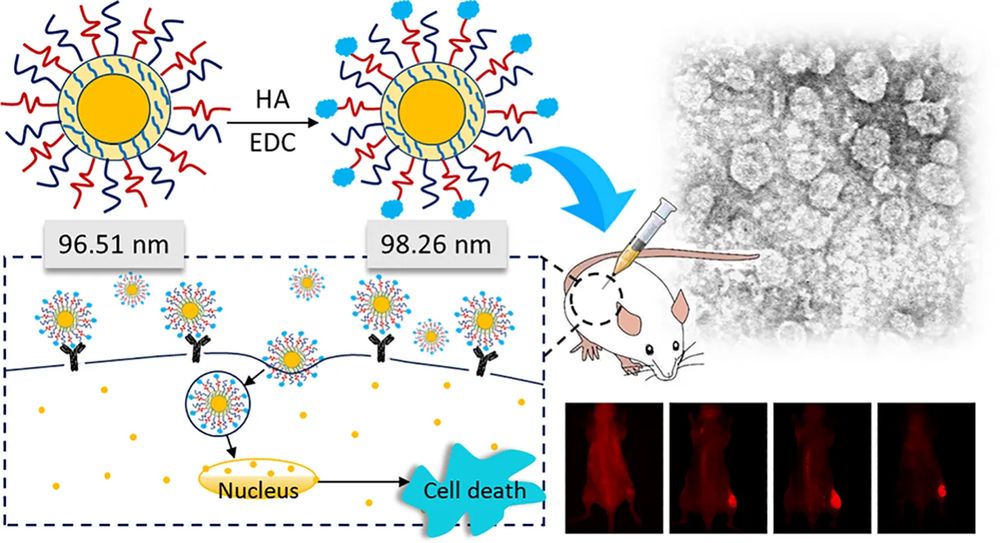
The image illustrates a typical targeted-liposome logic: selecting a disease-relevant receptor, choosing a matching ligand, conjugating that ligand onto the liposome surface, and then confirming whether the modification translates into stronger cellular uptake.
Choosing the Right Target for Liposome Targeting
To bypass off-target accumulation, identifying an appropriate biological target is the foundational step. Not every overexpressed receptor serves as a viable gateway for nanocarrier entry. A target must not only distinguish healthy cells from diseased cells but also function as a molecular turnstile that actively transports the liposome across the cellular membrane. The evaluation of a target must encompass several critical dimensions:
| Evaluation Question | Why It Matters for Liposome Delivery |
|---|---|
| Is the receptor abundantly expressed on target cells? | High receptor density dramatically improves the probability of ligand-receptor interaction, compensating for the brief contact time during circulation. |
| Is it properly exposed on the cell surface? | Steric hindrance by the cellular glycocalyx or extracellular matrix can block access. The target epitope must be accessible to a bulky liposomal structure. |
| Does receptor binding trigger endocytosis? | This is the most critical factor. Non-internalizing receptors will tether the liposome to the cell surface, but internalizing receptors (like transferrin or folate receptors) ensure intracellular payload delivery. |
| Is background expression limited in healthy tissues? | Limits "on-target, off-tumor" toxicity. Ideal targets exhibit a strong expression contrast between the intended delivery site and systemic tissues. |
Matching the Target with the Right Ligand
Once a robust internalizing target is validated, selecting the optimal ligand format is paramount. The decision should not be driven merely by what is available in the literature, but rather by a systematic evaluation of the target's biological properties, the intended administrative route, and translational constraints.
Antibodies and Antibody Fragments
Monoclonal antibodies offer unparalleled specificity and high binding affinity, making them excellent for high-value targets. However, fully intact antibodies are massive (~150 kDa), which can induce an unwanted immune response and significantly increase the overall hydrodynamic size of the liposome, potentially triggering rapid Reticuloendothelial System (RES) clearance. Consequently, antibody fragments (like Fab or scFv) are increasingly preferred, as they provide high specificity with a lower immunogenic profile and favorable pharmacokinetic properties.
Peptides
Peptides (such as the RGD sequence targeting integrins) are ideal when researchers aim to reduce molecular weight, enhance tissue penetration, and simplify chemical conjugation. Their smaller size translates to lower immunogenicity and easier large-scale synthesis. The primary challenge lies in maintaining their conformational stability and ensuring sufficient binding avidity once tethered to the lipid membrane.
Aptamers and Small Molecules
Aptamers provide a highly modifiable, synthetic alternative with low biological toxicity. Alternatively, small-molecule ligands, vitamins (e.g., folic acid), and carbohydrates (e.g., mannose or galactose) are perfect for transporter- or lectin-mediated targeting. Their simple chemistry and high stability make them exceptionally robust for manufacturing.
Struggling to identify the perfect ligand for your specific cell target?
Explore our Targeting Ligand Selection and Design expertise to accelerate your screening phase.Conjugation Strategy: Turning Ligand Choice into a Functional Liposome
A brilliantly chosen ligand is useless if the conjugation chemistry destroys its active binding site, causes the liposomes to aggregate, or induces premature payload leakage. The conjugation strategy dictates the physical architecture of the final particle.
Pre-insertion vs. Post-insertion
Pre-insertion involves conjugating the ligand to a lipid anchor (like DSPE-PEG) forming a micelle, which is then mixed with other lipids prior to liposome extrusion. While straightforward, it exposes the ligand to harsh organic solvents or mechanical stress during assembly. Post-insertion, conversely, involves creating a stable, drug-loaded liposome first, and subsequently incubating it with ligand-lipid micelles that spontaneously insert into the outer bilayer. This method is preferred for fragile proteins or sensitive drug payloads.
Common Coupling Chemistries
The chemical bridge must be stable in circulation but reactive enough to ensure efficient coupling. Maleimide-thiol chemistry is the gold standard for coupling peptides or antibodies bearing free sulfhydryl groups, ensuring a stable thioether bond. NHS-amine reactions are ubiquitous for primary amines but offer less site-specific control. Advanced platforms increasingly employ Click chemistry for its rapid, bio-orthogonal efficiency, while Biotin-streptavidin linkages remain a flexible tool for early-stage in vitro screening formats.
Critical Post-Conjugation Quality Checks
Modification of the liposome surface fundamentally alters its physical state. Before proceeding to cellular assays, the construct must be rigorously tested to ensure:
- Particle Size & PDI: Conjugation must not induce severe aggregation.
- Zeta Potential: Changes in surface charge dictate serum protein interactions.
- Encapsulation Retention: The chemical reaction must not compromise bilayer integrity.
- Ligand Density & Orientation: Confirming the ligand is facing outward and retains biological activity.
How to Prove the Targeted Liposome Really Works
Generating convincing evidence of active targeting requires a tiered validation architecture. It is insufficient to merely demonstrate increased fluorescence in a generalized assay; the data must explicitly confirm receptor specificity and map the intracellular journey of the payload.
Level 1: Uptake Specificity Evidence
Initial validation must prove that the liposome is utilizing the intended receptor. This is achieved through competitive inhibition assays, where the addition of excess free ligand should block the uptake of the targeted liposome.
Furthermore, quantitative readouts via flow cytometry must compare the uptake profiles across receptor-positive cell lines versus receptor-low or negative control cell lines. Comparing the targeted construct directly against an identically formulated "blank" (non-targeted) liposome establishes the definitive baseline.
Level 2: Intracellular Colocalization
Once entry is confirmed, spatial tracking is required. Using high-resolution confocal imaging, researchers map the fluorescently tagged liposomes against specific organelle markers.
Demonstrating colocalization with early endosomes or lysosomes validates the receptor-mediated endocytosis pathway. Time-course imaging is vital here to visualize the kinetics of internalization and subsequent endosomal escape, ensuring the payload reaches the cytosol or specific organelle (e.g., mitochondria or nucleus).
Discover our Functionalized Delivery System DevelopmentCommon Failure Points Across the Workflow
Recognizing common pitfalls early in the formulation phase can save substantial time and resources. Even with a theoretically sound design, practical execution can falter due to subtle chemical or biological mismatches.
Target selection based strictly on expression.
Selecting a target solely because it is highly expressed in transcriptomic data, without verifying its capacity for internalization, leads to liposomes that stick to the cell membrane without delivering their cargo.
Overcrowded ligand density.
More is not always better. An excessively high ligand density can cause steric hindrance, preventing proper receptor engagement, or it can induce rapid macrophage clearance in vivo.
Destructive conjugation chemistry.
Using non-specific amine coupling on an antibody may block its complementarity-determining regions (CDRs), rendering the targeting moiety completely inert post-conjugation.
Missing baseline controls in validation.
Presenting uptake data without corresponding free-ligand blocking assays or receptor-negative cell line data makes it impossible to differentiate specific targeting from non-specific endocytosis.
Step-by-Step Targeted Liposome Development Workflow
Define Model
Clearly define the disease model, affected cell types, and the ultimate therapeutic or diagnostic delivery objective.
Screen Targets
Screen target candidates focusing on surface accessibility, high contrast expression, and proven internalization pathways.
Select Ligand Format
Choose the optimal ligand format (antibody, peptide, small molecule) based on target structure and in vivo translation constraints.
Design Conjugation
Formulate the appropriate conjugation route (pre vs. post-insertion) and optimize the liposome base formulation.
Verify Properties
Conduct stringent physicochemical verifications post-modification to ensure size, PDI, and payload integrity remain intact.
Validate Specificity
Execute multi-tiered cellular assays to validate uptake specificity, mechanism of entry, and intracellular colocalization.
Frequently Asked Questions
Passive targeting relies entirely on the physical properties of the liposome (like size and PEGylation) to exploit anatomical differences in diseased tissues, such as the leaky vasculature in tumors (the EPR effect). Active targeting, however, involves the deliberate conjugation of a specific affinity ligand to the liposome surface to actively bind and trigger receptor-mediated endocytosis by the target cells, independent of passive accumulation alone.
References
- Xue, Gangqiang, et al. "Hyaluronic acid–targeted topotecan liposomes improve therapeutic efficacy against lung cancer in animals." Frontiers in Oncology 14 (2024): 1520274. https://doi.org/10.3389/fonc.2024.1520274
- Under Open Access license CC BY 4.0, without modification.
Online Inquiry
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.


