PG Anionic Liposome: Optimizing Membrane Stability and Payload Compatibility
Advanced formulation strategies for resolving lipid-cargo mismatch and achieving predictable release profiles across dynamic physiological environments.
What Is a PG Anionic Liposome and Why Is It Used?
PG anionic liposome systems are widely used in lipid-based drug delivery because phosphatidylglycerol provides a negative surface charge that can support colloidal stability, membrane organization, and formulation flexibility. However, optimizing a PG anionic liposome for both membrane stability and payload compatibility remains challenging across small molecules, proteins, and nucleic acids.
Under suitable formulation conditions, PG can help reduce aggregation and improve dispersion behavior. In physiological environments, however, electrolyte-rich media may weaken this benefit and increase the risk of instability. As a result, researchers and formulation scientists often struggle to balance structural integrity, encapsulation efficiency, and reproducible release performance in a single PG-based system.
Common development bottlenecks include lipid-cargo mismatch, insufficient loading, colloidal instability, and variable behavior after administration. Addressing these issues requires systematic optimization of lipid composition, buffer conditions, and loading strategy based on both payload type and target application. For projects that require deeper screening and formulation refinement, specialized Anionic Liposome Development Services can help align liposome surface properties with payload and application goals.
How Membrane Stability Affects PG Anionic Liposome Performance
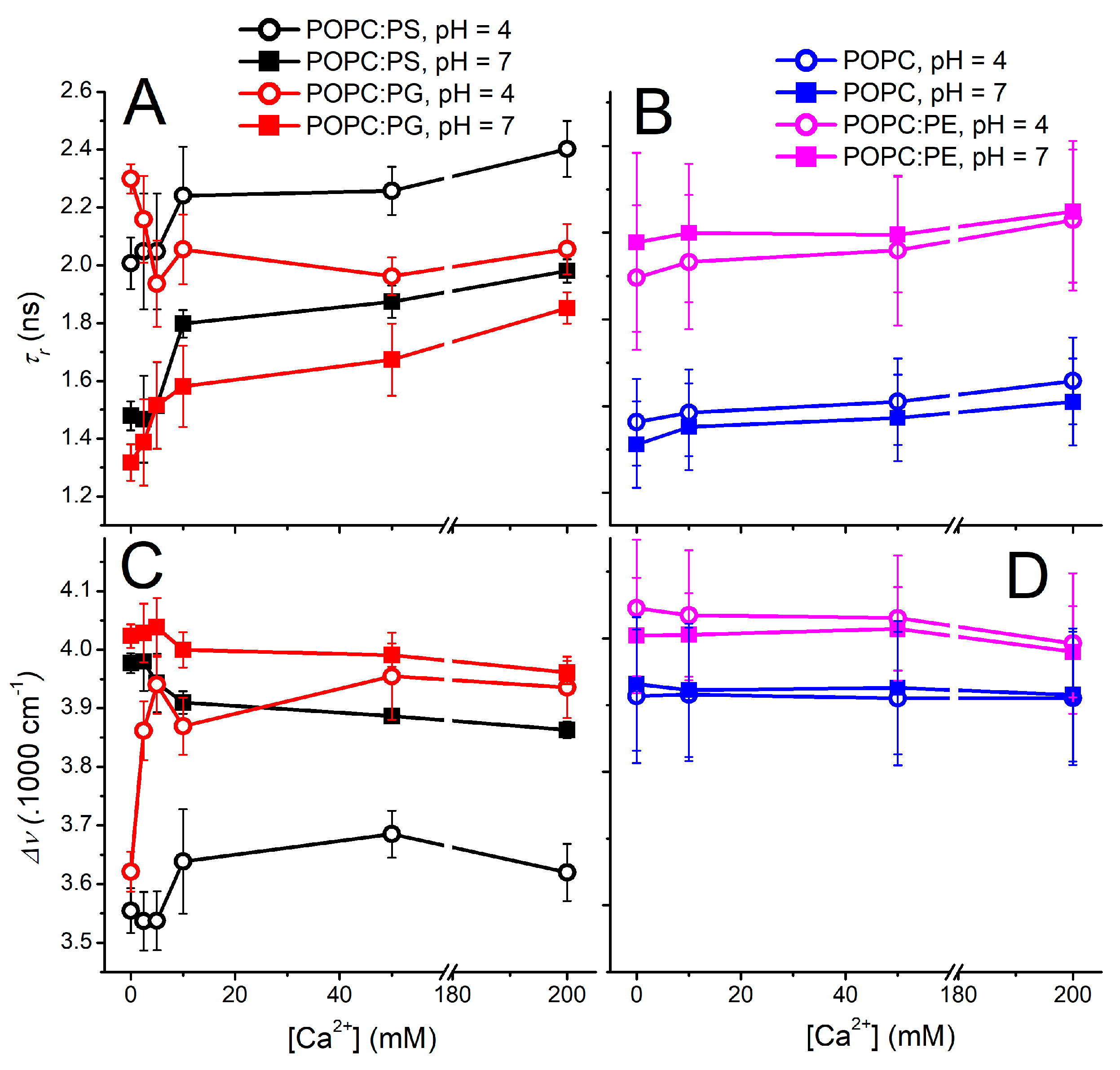
The stability of a PG anionic liposome is not an isolated attribute; it is a highly dynamic property governed by the surrounding microenvironment. Once administered in vivo, liposomes encounter varying pH levels and high concentrations of divalent cations, such as calcium, which can radically alter the structural integrity of the lipid bilayer.
As demonstrated in recent biophysical studies, PG-containing anionic lipid membranes are highly responsive to acidic and calcium-rich environments. Under acidic conditions, PG bilayers become less hydrated and more rigid, while calcium addition partially restores hydration and membrane mobility. For PG anionic liposome design, this suggests that membrane stability is not fixed by lipid identity alone, but is strongly shaped by the ionic microenvironment, making formulation optimization critical for maintaining structural integrity and drug compatibility.
Such conformational shifts have profound implications for drug delivery. Ion- and pH-induced changes in bilayer packing can alter permeability, interfacial hydration, and defect formation, which may lead to either improved retention or accelerated leakage depending on lipid composition and payload type. To overcome these dynamic challenges, targeted chemical engineering and rigorous testing are required to ensure the vesicle survives transit to the target site.
Improving Payload Compatibility in PG Anionic Liposome Formulations
A major hurdle in PG liposome development is ensuring payload compatibility across diverse molecular classes. The electrostatic nature of PG lipids can lead to either unwanted repulsions or excessively strong non-specific binding, both of which drastically reduce encapsulation efficiency. Formulation scientists must utilize advanced Liposome Formulation Optimization protocols to resolve these mismatches before progressing to in vitro or in vivo evaluations.
| Payload Class | Primary Interaction Challenge | Optimization Strategy |
|---|---|---|
| Small Hydrophobic Molecules | Membrane fluidization leading to premature leakage. The molecule partitions into the hydrophobic tail region, disrupting packing. | Tuning the cholesterol-to-PG ratio to increase acyl chain packing density and membrane rigidity, thereby retarding passive diffusion. |
| Therapeutic Proteins (e.g., mAbs) | Protein denaturation due to strong electrostatic binding to the anionic surface; orientation disruption. | Optimizing buffer composition, ionic strength, and pH within the protein’s conformational stability window to minimize adsorption, unfolding, or aggregation. |
| Nucleic Acids (RNA/DNA) | Severe electrostatic repulsion between the anionic phosphate backbone of the nucleic acid and the anionic PG headgroup. | Because nucleic acids are also negatively charged, PG-rich liposomes often require indirect loading strategies, such as complex-assisted encapsulation, helper lipids, ion-mediated condensation, or hybrid formulations that temporarily reduce electrostatic repulsion during assembly. |
By methodically tuning parameters such as the lipid molar ratio, phase transition temperatures, and zeta potential, researchers can dictate how a payload interacts with the liposome. A granular understanding of these interactions ensures the resulting nanomedicine functions efficiently without sacrificing vesicular architecture.
Key Formulation Variables for PG Anionic Liposome Optimization
Achieving a balance between structural integrity and high encapsulation demands tight control over multiple physicochemical parameters. To build a robust PG liposome formulation, developers must focus on the following core variables:
PG mol% and Helper Lipid Selection
PG content affects charge density, membrane curvature, and sensitivity to ionic shielding. Carefully selecting the ratio of PG to structural helper lipids (such as DSPC or DPPC) allows formulators to tune the phase transition temperature and overall rigidity of the bilayer. This precision is essential for managing the balance between fluidity and stability in complex biological fluids.
Cholesterol Content and Bilayer Packing
Cholesterol level modulates bilayer packing and leakage behavior. Acting as a critical fluidity buffer, the inclusion of cholesterol reduces membrane permeability to water-soluble molecules and prevents phase separation among different lipid components. Optimizing cholesterol levels is vital to maintaining physical stability during extended storage while ensuring the correct release kinetics.
Buffer Composition, pH, and Ionic Strength
Buffer ionic strength strongly affects surface charge screening and colloidal stability. The external and internal aqueous environments dictate the ionization state of the PG headgroups and the payload. Proper selection of salts and pH can minimize undesirable electrostatic phenomena, thereby enhancing loading efficiency and preventing liposomal aggregation during the formulation process.
Particle Size, Lamellarity, and Zeta Potential Control
The physical dimensions and charge profile of the PG anionic liposome directly govern biodistribution. Monodisperse, unilamellar vesicles typically exhibit more predictable in vivo performance. Furthermore, maintaining an optimal zeta potential ensures adequate inter-particle electrostatic repulsion, which is crucial to preventing aggregation prior to administration.
Loading Method Selection by Payload Class
Payload localization determines whether the main risk is leakage, denaturation, or poor loading. Passive loading techniques might suffice for lipophilic small molecules, whereas active gradient loading or pre-complexation strategies are often necessary for amphipathic drugs or biologics. Manufacturing method heavily influences reproducibility, size distribution, and scale-up feasibility.
Scale-Up Considerations for PG Anionic Liposome Development
Translating an optimized PG anionic liposome formula from the laboratory bench to a reproducible scalable process represents a formidable challenge. A formulation that exhibits excellent monodispersity and encapsulation in vitro may fail completely under the sheer stresses of large-scale extrusion or tangential flow filtration.
To ensure clinical and commercial viability, strict Quality Control (QC) metrics must be enforced. Variables such as the Polydispersity Index (PDI), encapsulation efficiency (EE%), and morphological consistency must remain stable across multiple batches. Furthermore, long-term shelf-life stability, often requiring optimization of lyophilization protocols with suitable cryoprotectants, must be evaluated to prevent aggregation of the anionic particles over time.
Ensure Consistency Across All Scales
Avoid the pitfalls of unpredictable release behaviors and post-processing instability. Implementing robust analytical frameworks early in the development cycle is essential for a seamless transition to clinical phases.
Frequently Asked Questions
The main advantage of a PG anionic liposome lies in the negative surface charge imparted by phosphatidylglycerol. This charge helps prevent vesicle aggregation through electrostatic repulsion, improving colloidal stability under specific formulation conditions. It also offers unique tunability for interacting with certain microenvironments and basic drug molecules.
In physiological environments rich in electrolytes (like sodium and calcium ions), the negative charges on the PG headgroups can be shielded or cross-linked by divalent cations. This electrostatic screening diminishes the repulsive forces between liposomes, potentially leading to particle aggregation, changes in membrane hydration, and altered permeability.
Yes, but they require meticulous optimization. Proteins possess complex surface charge distributions that can strongly interact with anionic PG lipids, risking protein unfolding or denaturation. Success relies on optimizing buffer composition, ionic strength, and pH within the protein's conformational stability window to minimize detrimental surface adsorption.
While challenging due to the electrostatic repulsion between the negatively charged nucleic acid backbone and the anionic PG lipids, it is possible. It typically requires indirect loading strategies, such as using pre-complexation agents, specific helper lipids, or ion-mediated condensation techniques to temporarily bridge or shield the charges during formulation.
Improving encapsulation efficiency depends entirely on the payload. For small lipophilic drugs, adjusting the cholesterol-to-PG ratio to optimize bilayer packing is key. For amphipathic or hydrophilic compounds, active loading methods employing transmembrane pH or ion gradients often yield the highest efficiency while preventing lipid-cargo mismatches.
During scale-up, maintaining batch-to-batch consistency is critical. The most vital parameters to monitor include the Polydispersity Index (PDI), mean particle size, zeta potential (to ensure continued electrostatic repulsion), encapsulation efficiency, and morphological integrity under the stress of large-scale manufacturing processes like extrusion.
References
- Abhinav, et al. "Modulation of Anionic Lipid Bilayers by Specific Interplay of Protons and Calcium Ions." Biomolecules 12.12 (2022): 1894. https://doi.org/10.3390/biom12121894
- Under Open Access license CC BY 4.0, without modification.
Online Inquiry
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.


