Proliposomes are dry and free flowing particles consisting of phospholipids coatings on water soluble powder particles. They solve the problem associated with sterilization of liposomes as they have a high storage stability and provide simple large-scale production. They have successfully been used for enhancing bioavailability of anticancer drugs and other therapeutic agents. Creative Biolabs assembles a strong team with experienced Ph.D. level scientists, well trained technicians and responsible project managers just in order to deliver the most suitable service according to your budget, timeline, and specific requirements. Our professional team is optimized to help you get the customized services for your drug delivery project.
Proliposomes (PLs) are dry, free-flowing granular products that on hydration or on contact with biological fluids in the body, form liposomal dispersion. They are composed of water soluble porous powder and phospholipid and have provided a major breakthrough in solving the stability issues associated with liposomes. Liposomes can either be formed in vivo by the influence of biological fluids in the body or in vitro using a suitable hydrating fluid before the administration. Solubility and bioavailability problems of many drugs can be overcome by developing PL formulations. PLs are phospholipid based drug delivery systems that are finding important applications in the field of pharmaceutics. For producing commercial liposome products, PL is one of the most widely used and cost-effective methods. As they are available in dry powder form, it’s easy to distribute, transfer, measure and store, making it a diverse system. They are prepared by many methods such as film-deposition on carrier method, spray drying method, fluidized-bed method and supercritical anti-solvent method.
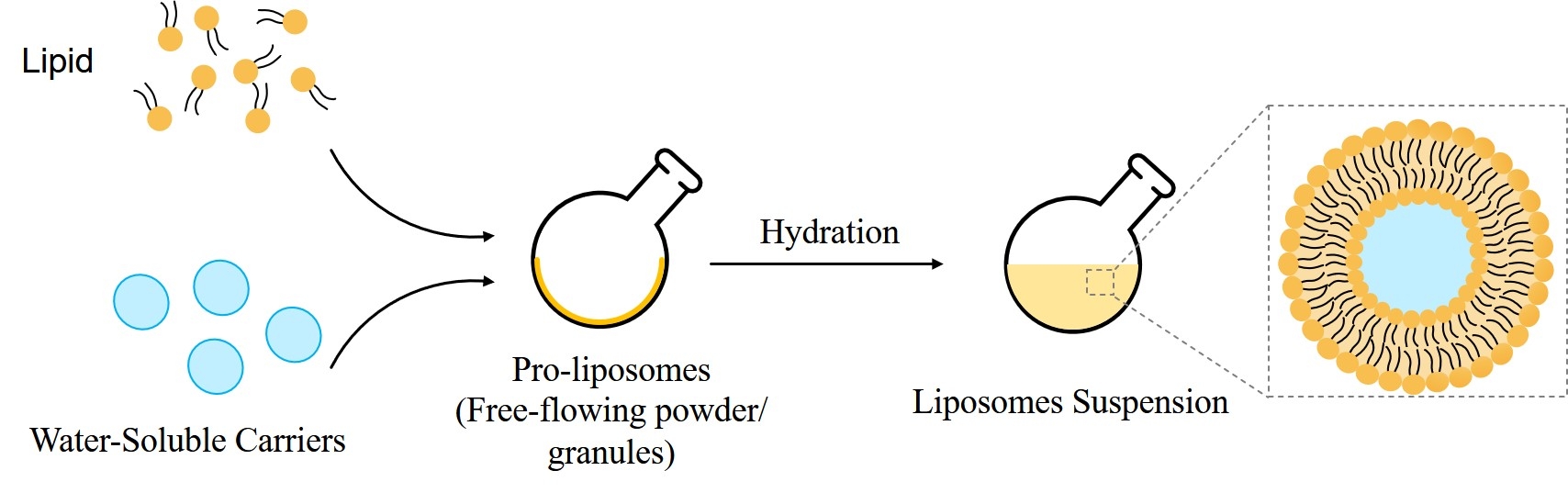 Fig.1 Synthesis of Proliposomes.
Fig.1 Synthesis of Proliposomes.
Oral delivery has been the most convenient route of delivery due to its ease of administration, high patient adherence, cost effectiveness, less invasive and flexibility in the design of dosage form. However, the major problem associated with development of an oral dosage form is the poor bioavailability of the drug. The stability of PLs is far superior to liposomes making them more suitable for the delivery of drugs with poor oral bioavailability. In pro-liposomal technology, the drug is usually incorporated either into the hydrophilic region or encapsulated in the lipid bilayers depending on the hydrophilicity or lipophilicity of the drug. Besides, PLs demonstrated controlled drug release, improved stability and increased solubility relatively to conventional liposomes. Preparing liposomes by conversion from PLs allows the encapsulation of a wide variety of drugs with different solubility in water and organic solvents and presents high encapsulation efficiencies when compared to other methods based on passive entrapment. Based on these, PLs have definitely established their position in modern delivery system.
PLs hold great promise for oral delivery of drugs with poor bioavailability. They have also opened newer areas of liposome application. Aided by our well-established platforms and experienced scientists, A wide spectrum of PLs is available for your choice. Moreover, we can provide customized services of PLs and also provide more LDD formulations for drug delivery.
At Creative Biolabs, we provide the largest and diversiform portfolio of LDD products and services. Our service can be designed to meet your special needs if you have any requirements. If you are interested in our service, please contact us by e-mail and our team will get back to you as soon as possible.
 For Research Use Only. Not For Clinical Use
For Research Use Only. Not For Clinical UseSupports
Online Inquiry