PEGylation Is Not Always Better:
When to Use Cleavable PEG in Lipid-Based Delivery
PEGylation is widely used to extend circulation and stabilize lipid-based carriers, but more PEG is not always better. In programs that depend on tumor penetration, intracellular delivery, or repeated administration, persistent PEG shielding can become a barrier rather than a benefit. This page explains when a cleavable PEG strategy may offer a better balance between systemic stability and biological activity.
What Is the PEG Dilemma in Liposome and LNP Delivery?
Surface modification with polyethylene glycol (PEG) is foundational to modern nanomedicine. A robust PEG hydration layer masks lipid nanoparticles (LNPs) from serum opsonization and extends in vivo half-life. This "stealth" strategy is central to standard long-circulating liposome development.
However, at the target site, the very property that protected the carrier becomes a barrier. The steric hindrance of the PEG corona can block close interaction between the nanoparticle and target cell membranes. This trade-off is the "PEG Dilemma".
Benefits of PEGylation
- ✓ Prevents particle aggregation
- ✓ Reduces MPS/RES clearance
- ✓ Extends circulation half-life
Constraints of Permanent PEG
- ⚠ Can reduce targeted cellular uptake
- ⚠ Impedes endosomal escape fusion
- ⚠ May trigger ABC phenomenon in repeated dosing
Permanent PEG vs. Cleavable PEG: Which Fits Your Program?
In candidate screening, choosing between standard PEGylation and stimulus-responsive (cleavable) PEG requires balancing biological needs with manufacturing complexity.
| Key Parameters | Permanent PEG | Cleavable PEG |
|---|---|---|
| Circulation Stability | High & consistent | High initially, programmed to shed |
| Cellular Uptake | Often restricted (steric barrier) | Restored upon target-site trigger |
| Endosomal Escape | Poor (hinders membrane fusion) | Significantly improved |
| Repeated Dosing | High risk of ABC phenomenon | May reduce immune recognition |
| Formulation Complexity | Standard & scalable | Requires advanced analytical validation |
| Best-Fit Applications | Passive systemic delivery, simple liposomal small molecules | Nucleic acids (mRNA/siRNA), tumor-targeted payloads, multi-dose regimens |
When Should You Use Cleavable PEG?
A Practical Decision Framework for Formulation Design Teams
Cleavable PEG is not a universal upgrade. It is most valuable when a program needs both early-stage stealth and later-stage surface exposure, and when a relevant biological trigger is available. If your program aligns with the following conditions, it may be time to pivot your PEG strategy:
1. The Payload Needs Cytosolic Access
If your LNP carries mRNA, siRNA, or CRISPR components, endosomal escape is mandatory. Permanent PEG prevents the cationic lipids from interacting with the endosomal membrane, resulting in lysosomal degradation rather than transfection.
2. Repeated Administration is Required
For translational programs requiring multi-dose regimens (e.g., enzyme replacement, continuous oncology therapies), reducing persistent PEG exposure can be a strategic move to manage immunogenicity and the ABC phenomenon.
3. Shielding Active Targeting Ligands
When engineering targeted liposomes, exposing targeting ligands (like peptides or antibodies) too early can lead to off-target binding or rapid clearance. Cleavable PEG acts as a "smart cover" that exposes the ligand only at the disease site.
4. Exploiting a Defined Microenvironment
The strategy requires a reliable biological trigger. If the target tissue possesses a distinct gradient—such as the acidic tumor microenvironment (pH ~6.5) or highly reductive cytosol (high GSH)—stimuli-responsive PEG can be successfully deployed.
5. Uptake Data Indicates the "PEG Dilemma"
If your in vitro cell uptake assays show poor internalization despite excellent stability, or if in vivo data shows high tumor accumulation but poor therapeutic efficacy, the particles might be trapped in the extracellular matrix. DePEGylation is a targeted rescue strategy for these exact symptomatic profiles.
How Cleavable PEG Improves Tumor, Intracellular Delivery, and Repeated Dosing
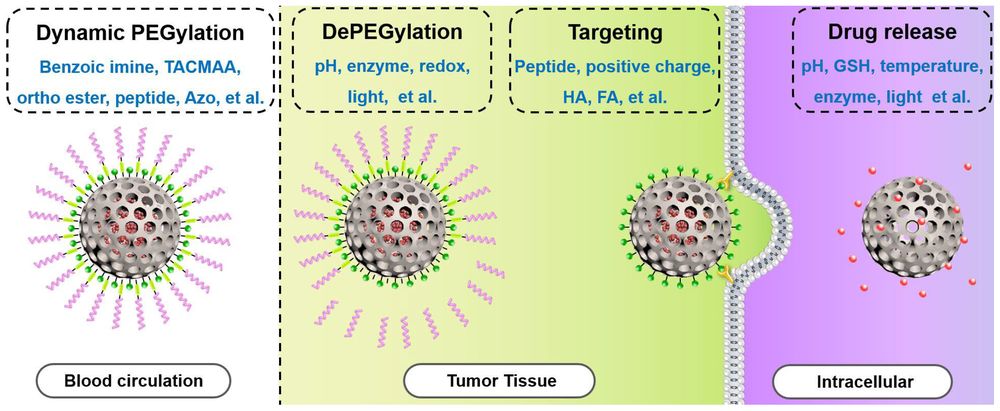
Fig.1 Schematic illustration of DePEGylation for “tumor-triggered” targeting or intracellular drug release. 1,2
The DePEGylation Rationale
This scheme illustrates the rationale of dePEGylation-based delivery systems: PEG provides stealth and prolonged circulation during systemic transport, while stimulus-triggered PEG shedding at the tumor site or intracellular environment helps restore cellular interaction and promote payload release.
Once the environmental trigger cleaves the linker, the PEG corona detaches, revealing a bare formulation surface that can drive rapid endocytosis or expose cationic charges for membrane disruption.
Solid Tumor Microenvironment (TME) Targeting
While standard PEGylated liposomes passively accumulate in tumors via the EPR effect, bulky PEG shells often prevent deep matrix penetration.
Utilizing linkers sensitive to mild extracellular acidity (pH 6.5-6.8) or Matrix Metalloproteinases (MMPs), the PEG layer can shed precisely in the tumor interstitium. This in situ dePEGylation facilitates deeper tissue penetration and direct cellular binding.
Intracellular Nucleic Acid Delivery
For mRNA or siRNA therapies, endosomal escape is the critical bottleneck. Permanent PEGylation suppresses the necessary electrostatic interactions.
Incorporating a cleavable lipid—often accessed via a custom PEGylated lipid synthesis service—that responds to the acidic endosome (pH 5.0-6.0) is designed to enable PEG shedding exactly when the LNP needs to fuse, supporting high transfection efficiency.
Repeated Dosing and Immune Compatibility
Persistent PEG exposure can induce anti-PEG IgM/IgG, triggering the Accelerated Blood Clearance (ABC) phenomenon upon subsequent administrations.
By shedding the PEG layer quickly after accumulating in the target, the systemic exposure time to the PEG epitope is reduced. Cleavable PEG can be considered as one strategy to mitigate immune recognition, though factors like dose interval and PEG density remain critical.
Which Cleavable PEG Linker Should You Choose?
Selecting the correct cleavable bond is highly dependent on the physiological trajectory of the nanocarrier. Different chemistries offer distinct biological triggers and specific manufacturing trade-offs.
| Linker Chemistry | Cleavage Trigger & Location | Primary Application | Key Design Consideration |
|---|---|---|---|
| Hydrazone / Orthoester |
Mild acidity TME (pH ~6.5), Endosomes (pH 5.0-6.0) |
Enhancing endosomal escape, tumor penetration. | Risk of premature cleavage under slightly acidic storage/buffer conditions. |
| Disulfide Bond (-S-S-) |
High reducing potential (GSH) Intracellular Cytosol |
Intracellular release of fragile payloads (mRNA, siRNA). | Extracellular stability and resistance to plasma thiols must be thoroughly validated. |
| Peptide-based (e.g., GPLGIAGQ) |
Specific Enzymatic Cleavage TME matrix (MMP-2/9) |
Exposing shielded targeting ligands exactly at the tissue site. | Trigger specificity depends heavily on disease biology and enzyme heterogeneity. |
How to Balance Triggered Shedding with Formulation Stability
At the formulation design stage, incorporating stimuli-responsive elements significantly improves potential therapeutic outcomes but introduces complexity into manufacturing and CMC (Chemistry, Manufacturing, and Controls). A cleavable PEG lipid must be responsive enough to degrade in vivo under the precise trigger, yet stable enough to survive long-term storage ex vivo and circulation in the blood without premature shedding.
During lead optimization, achieving this delicate balance requires rigorous analytical testing. It is essential to continuously monitor particle size, zeta potential, encapsulation efficiency, and structural integrity under stress conditions. Partnering with a specialized formulation stability monitoring service provides the necessary comprehensive kinetic data to ensure your "smart" nanocarriers remain intact prior to administration.
Optimize Your Smart Liposome FormulationFrequently Asked Questions
The "PEG dilemma" refers to the trade-off inherent in PEGylating nanoparticles. While a dense PEG corona hides the particle from the immune system and extends circulation time, it simultaneously creates a steric barrier. This barrier can prevent the nanoparticle from closely interacting with target cell membranes, potentially hindering cellular uptake and subsequent endosomal escape.
Nucleic acids like mRNA must reach the cell's cytoplasm to function. LNPs use ionizable lipids that become positively charged in the acidic endosome to fuse with the endosomal membrane. Permanent PEG can block this fusion. Cleavable PEG lipids designed to shed in the low pH of the endosome expose the cationic lipids at the critical moment, supporting efficient endosomal escape.
Common triggers leverage the unique characteristics of the tumor microenvironment (TME). These include mild extracellular acidity (pH 6.5-6.8), which can trigger hydrazone or orthoester linkers, and the overexpression of specific enzymes like Matrix Metalloproteinases (MMPs), which can cleave engineered peptide linkers near the tumor site.
Potentially, in some formulations. The ABC phenomenon is often caused by anti-PEG antibodies generated upon repeated administration of PEGylated nanocarriers. By utilizing cleavable PEG architectures, the systemic residence time of the PEG epitope is reduced, which may serve as one strategy to lower the immunogenic profile in multi-dose regimens, though dose intervals and lipid design remain crucial factors.
Formulation stability requires meticulous chemistry selection and buffer optimization to prevent premature cleavage. It is essential to conduct rigorous accelerated stability studies (evaluating dynamic light scattering, zeta potential, and payload retention) to track formulation integrity over time before moving to in vivo studies.
References
- Ren, Zhe, et al. "Drug delivery systems with a “Tumor-Triggered” targeting or intracellular drug release property based on DePEGylation." Materials 15.15 (2022): 5290. https://doi.org/10.3390/ma15155290
- Under Open Access license CC BY 4.0, without modification.
Online Inquiry
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.


