Neutral Liposome Development: How to Design Formulations for Stability and Long Circulation
Empowering researchers and biopharma developers with advanced formulation strategies to achieve high serum stability, prolonged circulation, and translational manufacturability for systemic drug delivery.
Navigating the Formulation Challenges of Systemic Drug Delivery
The successful systemic delivery of therapeutics—ranging from small molecule chemotherapeutics to complex biologics—relies heavily on the nanocarrier's ability to navigate a hostile physiological environment. Among various lipid-based nanosystems, neutral liposome development has proven indispensable for improving the pharmacokinetics and biodistribution of encapsulated active pharmaceutical ingredients (APIs). However, researchers and biopharma developers face a persistent core challenge: engineering a rational formulation that flawlessly balances serum stability, prolonged circulation, robust drug loading, and translational manufacturability.
Unlike cationic liposomes, which readily interact with negatively charged cell membranes but suffer from rapid clearance and potential systemic toxicity, neutral liposomes inherently avoid non-specific protein binding. Yet, a formulation composed solely of basic neutral phospholipids is highly susceptible to premature degradation by serum lipoproteins and rapid clearance by the mononuclear phagocyte system (MPS). Overcoming this limitation requires a highly sophisticated approach to lipid selection, where the interplay between neutral phospholipids, cholesterol, and PEGylated lipids determines the ultimate fate of the liposome in vivo.
The Structural Basis of Neutral Liposomes
Understanding the precise architecture of a liposome and the chemical properties of its constituent lipids is the foundational step in rational design. The mechanical strength of the lipid bilayer and its interaction with the surrounding biological fluid depend directly on these structural parameters.
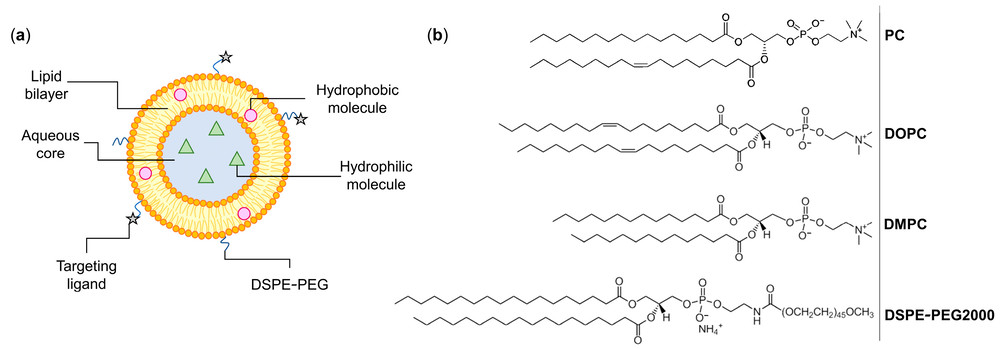
This figure illustrates the structural basis of neutral liposome development. In long-circulating and stable formulations, neutral phospholipids form the bilayer framework, higher-transition-temperature lipids help reinforce membrane rigidity, and PEG-lipids such as DSPE-PEG reduce opsonization and prolong systemic circulation. The physical chemistry of the selected phospholipids, particularly the acyl chain length and degree of saturation, profoundly influences the bilayer's permeability.
To ensure that the structural integrity of your designed formulation meets translational standards, deep analytical characterization is required. Proper validation guarantees that the liposomes will perform predictably from in vitro cell culture testing to in vivo pharmacokinetic profiling.
Strategic Lipid Selection for Optimal Membrane Rigidity
The backbone of any neutral liposome is the primary phospholipid, most commonly a phosphatidylcholine (PC) derivative. The critical parameter here is the lipid's phase transition temperature (Tm)—the temperature at which the lipid bilayer transitions from a solid gel phase to a fluid liquid-crystalline phase.
Lipids with saturated, long hydrocarbon chains, such as DSPC (1,2-distearoyl-sn-glycero-3-phosphocholine, Tm ~55°C) and DPPC (1,2-dipalmitoyl-sn-glycero-3-phosphocholine, Tm ~41°C), create highly ordered, rigid bilayers at physiological body temperature (37°C). This rigidity restricts the mobility of the lipid chains, drastically minimizing the premature leakage of encapsulated hydrophilic drugs. Conversely, unsaturated lipids like DOPC (1,2-dioleoyl-sn-glycero-3-phosphocholine, Tm ~-17°C) yield highly fluid membranes that are generally unsuitable for prolonged systemic circulation, though they may be useful for specific topical or rapid-release applications.
Selecting the right matrix lipid is a nuanced decision. A formulation intended for intravenous chemotherapy demands high retention until it reaches the tumor microenvironment. Through specialized neutral liposome development services, formulators can systematically screen different lipid combinations to identify the exact Tm and hydrocarbon chain architecture required to match the API's release profile and the targeted disease state.
The Essential Role of Cholesterol in Serum Stability
While high-Tm phospholipids provide rigidity, they can also make the bilayer overly brittle, leading to poor drug loading and unpredictable crystallization during storage. Cholesterol acts as the crucial "fluidity buffer" in neutral liposome formulations.
Incorporated typically at 30-45 mol%, cholesterol intercalates between the phospholipid molecules. At temperatures below the lipid's Tm, cholesterol disrupts the tight packing of the acyl chains, preventing crystallization. Above the Tm, it restricts chain motion, reducing membrane permeability. Most importantly, in vivo, adequate cholesterol content prevents high-density lipoproteins (HDL) in the blood from extracting phospholipids from the liposome, thereby conferring essential serum stability and preventing the rapid dismantling of the nanocarrier.
PEGylation: Engineering the Stealth Effect for Prolonged Circulation
Even a rigid, cholesterol-stabilized neutral liposome will eventually be recognized by the immune system as a foreign entity. To achieve true longevity in the bloodstream, formulators turn to surface modification via PEGylation.
Mechanism of Steric Hindrance
The inclusion of PEGylated lipids, predominantly DSPE-PEG2000, generates a dense, hydrophilic hydration sphere around the liposome. This polymeric brush layer provides robust steric hindrance, effectively masking the liposome surface. As a result, the adsorption of serum opsonins (such as immunoglobulins and complement proteins) is drastically reduced. Without opsonization, the liposomes successfully evade phagocytosis by the macrophages of the MPS, particularly in the liver and spleen.
This "stealth" attribute translates to an extended circulation half-life, allowing the nanoparticles to pass through the fenestrated vasculature of tumors or inflamed tissues via the Enhanced Permeability and Retention (EPR) effect. However, over-PEGylation (typically >10 mol%) can disrupt the bilayer structure, causing the transition from vesicular liposomes to mixed micelles, leading to catastrophic loss of the encapsulated payload.
Optimizing Molar Ratios for Translational Success
The art of neutral liposome formulation lies in establishing the optimal molar ratio of these three core components: the neutral phospholipid, cholesterol, and the PEG-lipid. Minor adjustments can lead to profound differences in ex vivo stability and in vivo performance.
| Formulation Component | Typical Molar Ratio | Primary Function & Impact |
|---|---|---|
| Phospholipid (e.g., DSPC, DPPC) | 50 - 65 mol% | Forms the primary structural bilayer; high Tm lipids reduce permeability and drug leakage. |
| Cholesterol | 30 - 45 mol% | Modulates membrane fluidity, prevents phase separation, and inhibits lipid extraction by serum lipoproteins. |
| PEG-lipid (e.g., DSPE-PEG2000) | 2 - 5 mol% | Provides steric stabilization, minimizes opsonization, and extends circulation half-life in vivo. |
Analytical Validation and Stability Tracking
Beyond theoretical design, practical scale-up and rigorous quality control are mandatory. Formulations must undergo techniques like thin-film hydration followed by sequential extrusion or microfluidic mixing to achieve a uniform size distribution (typically 80-120 nm) with a low polydispersity index (PDI < 0.2).
Translational manufacturability requires continuous monitoring. Developers must track parameters like vesicle fusion, drug leakage, and lipid degradation under various storage conditions. Leveraging a specialized formulation stability monitoring service provides the necessary analytical insight to confirm that the designed neutral liposome maintains its physical and chemical integrity from the bench to the clinic. By combining advanced lipid chemistry with rigorous analytical testing, researchers can successfully bridge the gap between initial discovery and viable systemic therapies.
Frequently Asked Questions
Neutral liposomes offer superior biocompatibility and drastically reduced systemic toxicity compared to cationic formulations. Because they lack a strong positive surface charge, neutral liposomes do not non-specifically bind to negatively charged serum proteins and cell membranes, which helps prevent rapid clearance by the immune system and reduces off-target effects in vivo.
Cholesterol is essential for maintaining the structural integrity of the lipid bilayer in biological fluids. It modulates membrane fluidity—preventing crystallization below the phase transition temperature and reducing permeability above it. Crucially, in vivo, adequate cholesterol levels (typically 30-45 mol%) prevent high-density lipoproteins from stripping phospholipids from the liposome, thereby enhancing serum stability.
For long-circulating "stealth" liposomes, the optimal concentration of a PEG-lipid (like DSPE-PEG2000) is generally between 2 to 5 mol%. This ratio provides sufficient dense steric hindrance to prevent opsonization and evade macrophage clearance. Exceeding 8-10 mol% can compromise the structural integrity of the liposome, potentially leading to micelle formation rather than stable vesicles.
The Tm determines whether the lipid bilayer is in a solid gel phase or a fluid liquid-crystalline phase at body temperature (37°C). High-Tm lipids like DSPC form rigid bilayers that drastically slow down the diffusion of encapsulated drugs, ensuring the cargo is safely retained during systemic circulation until the liposome reaches its target site.
Stability monitoring involves tracking physical and chemical parameters over time and under specific storage or serum conditions. Techniques include Dynamic Light Scattering (DLS) to measure changes in particle size and polydispersity (indicating aggregation or fusion), zeta potential analysis, and HPLC methods to quantify lipid degradation (oxidation/hydrolysis) and the precise rate of drug leakage.
References
- Luiz, Hugo, Jacinta Oliveira Pinho, and Maria Manuela Gaspar. "Advancing medicine with lipid-based nanosystems—the successful case of liposomes." Biomedicines 11.2 (2023): 435. https://doi.org/10.3390/biomedicines11020435
- Under Open Access license CC BY 4.0, without modification.
Online Inquiry
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.


