Prodrug Liposomes: Translating Chemical Design into Delivery Advantages
A practical framework for converting prodrug chemistry into higher loading, lower premature leakage, and more controllable tumor-site activation.
The Delivery Bottleneck in High-Potency Therapeutics
Who may benefit from this strategy?
This strategy is particularly relevant for teams working on highly potent cytotoxics, difficult-to-retain small molecules, or payloads that require tighter control over systemic exposure.
For teams developing difficult-to-deliver anti-cancer small molecules or highly potent payloads, identifying an active compound is only the beginning of the development challenge. The main barrier is often no longer whether liposomes can be formulated, but whether molecular design can be translated into reliable in vivo delivery performance.
The bottleneck usually emerges at three connected levels: the intrinsic liabilities of the native payload, the formulation limits of conventional liposomes, and the disconnect between prodrug synthesis and delivery design.
Native Payload Liabilities
During preclinical translation, many potent small molecules still face the same historical problems: high systemic toxicity, poor solubility, limited formulation compatibility, and a critically narrow therapeutic window.
Limits of Conventional Liposomes
Even when a native drug can be encapsulated, conventional liposomes may still show limited loading efficiency, poor retention during circulation, or insufficient control over when and where the payload is released in the tumor microenvironment.
The Chemistry-Delivery Disconnect
In many projects, prodrug synthesis and nanocarrier formulation are still optimized separately. As a result, the prodrug may be chemically elegant but poorly aligned with the biophysical requirements of stable liposomal delivery.
Why Prodrug Liposomes Can Outperform Conventional Formulations
Prodrug liposomes offer a versatile solution—provided that the prodrug is designed not only for chemical conversion, but also for carrier compatibility. When chemical design is matched to carrier requirements, prodrug structure can be translated into practical delivery advantages.
Improve Bilayer Compatibility
By synthetically masking hydrophilic groups or appending lipophilic tails, prodrugs can achieve a higher partition coefficient (LogP), supporting more stable incorporation within the lipid bilayer.
Reduce Premature Drug Leakage
The covalent linker strategy may reduce "burst release" and serum-driven dissociation during systemic circulation, helping to widen the therapeutic window of toxic payloads.
Enable Conditional Activation
Liposomal formulations provide a vehicle to transport stimuli-responsive prodrugs, enabling targeted cleavage and conditional activation under specific tumor conditions or exogenous triggers.
Translating Chemical Design into Delivery Outcomes
Chemical design matters only when it is translated into measurable delivery outcomes. To build a successful prodrug liposome, researchers must approach the problem through a continuous causal chain, from initial lipid-drug compatibility to final pharmacokinetic behavior.
Design for Bilayer Compatibility
Partitioning & Loading
By introducing a lipophilic anchor (e.g., cholesterol derivatives, long-chain fatty acids) or an amphiphilic motif, prodrug design can increase the tendency of the payload to partition into the hydrophobic acyl core of the lipid bilayer. This often improves bilayer retention and loading behavior compared to standard gradient loading of the native drug.
Translating this chemistry requires expertise in lipid selection to avoid phase separation. To help navigate this, researchers can utilize our liposome-encapsulated prodrug development services, which are designed to support the screening of stable co-assembly parameters and structure and composition analysis.
Design for Circulation Stability
Reducing Premature Leakage
Hydrophilic or moderately amphiphilic native drugs can easily diffuse out of the liposome when exposed to dynamic shear forces and protein-rich environments. Stronger hydrophobic association between the lipophilic prodrug moiety and the lipid bilayer can improve retention during circulation and reduce premature dissociation in protein-rich biological environments.
Prolonged circulation and appropriate formulation properties can support accumulation at tumor sites; additional targeting ligands may further improve localization in selected designs.
Design for Conditional Activation
Triggerable Linkers
Chemical linkers joining the active payload to the lipophilic anchor are evaluated based on their stability in the bloodstream versus their susceptibility to distinct stimuli. Endogenous triggers rely on conditions like high ROS, acidic pH, or specific proteases (e.g., Cathepsin B). Exogenous triggers (like light, magnetic fields, or ultrasound) may offer supplementary spatial control.
Representative Case: Translating Prodrug Chemistry into Ultrasound-Triggered Delivery
To illustrate how chemical design can be translated into delivery performance, a representative exogenous-triggered liposomal system can be considered. In this type of platform, prodrug structure, lipid composition, and activation mechanism are designed together rather than optimized in isolation.
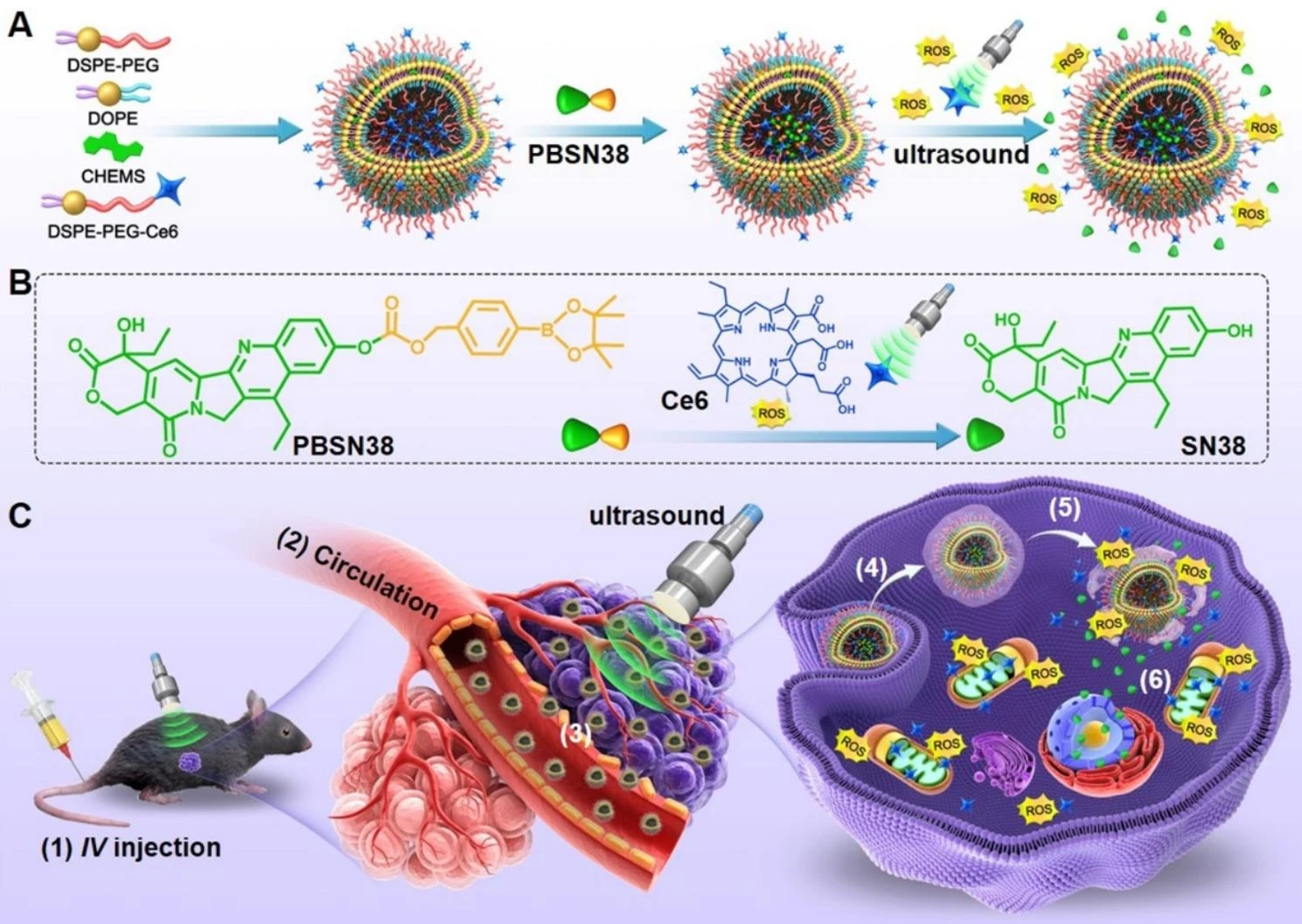
Recently, scientists reported an ultrasound-activated prodrug-loaded liposome composed of chlorin e6 (Ce6)-modified lipids and pinacol boronic ester-conjugated SN38 (PBSN38). In this design, the liposomal carrier provides a transport framework for the chemically modified payload, while the Ce6 component functions as a sonosensitizing module. Upon ultrasound irradiation, ROS generation is enhanced and promotes cleavage of the prodrug linker, enabling conditional release of active SN38 at the tumor site.
This example is informative not because ultrasound activation is required in all prodrug liposome systems, but because it clearly shows how molecular design can be aligned with delivery goals. The prodrug is engineered not only for chemical masking, but also for carrier compatibility, reduced premature leakage, and externally controlled activation. For teams developing potent oncology payloads, this type of integrated design provides a practical framework for linking chemistry decisions to measurable delivery outcomes.
Analytical Checkpoints for Translating Design into Delivery Performance
For teams moving from formulation to in vivo validation, assessing the true benefit of a prodrug liposome requires stringent testing. A rationally designed prodrug should be evaluated through specific analytical checkpoints to confirm that the chemical modifications facilitate expected delivery benefits.
Key Evaluation Questions for Prodrug Liposome Development
- ✓ Bilayer Retention: Is the prodrug retained in the lipid bilayer under physiologically relevant serum conditions?
- ✓ Linker Stability: Does the linker stability match the intended circulation window, avoiding premature hydrolysis in vivo?
- ✓ Trigger Sensitivity: Is activation measurable and robust under biologically relevant trigger conditions? (Supported by in vitro release kinetics analysis)
- ✓ PK Differentiation: Can pharmacokinetic assays distinguish the intact prodrug-liposome complex from prematurely released free drug?
- ✓ Biodistribution Profile: Does the biodistribution data support lower off-target exposure and appropriate tissue accumulation? (Supported by PK/PD analysis)
Frequently Asked Questions
A prodrug liposome strategy is often worth considering when the native payload exhibits a very narrow therapeutic window, high systemic toxicity, or when it cannot be stably retained in conventional liposomes due to poor partitioning (e.g., highly water-soluble drugs that leak rapidly, or certain challenging lipophilic drugs that crystallize). They are also useful when specific spatial or temporal control of drug release is mandated by the target disease biology.
Carrier compatibility is often improved by modifying the native drug with lipophilic anchors, such as cholesterol derivatives, tocopherol, diacylglycerols, or long-chain aliphatic tails. These features increase the hydrophobicity (LogP) of the molecule, allowing it to insert deeply into the lipid bilayer of the liposome, which generally improves encapsulation efficiency and reduces premature drug leakage.
This balance is critical and often represents the core challenge in prodrug design. The linker must be sufficiently robust to withstand hydrolysis and enzymatic degradation in the systemic circulation to prevent off-target toxicity. Simultaneously, it must be highly sensitive to the chosen trigger (e.g., tumor-specific enzymes, acidic pH, or external stimuli like ultrasound) to ensure the active payload is efficiently released at the pathological site.
Prior to in vivo validation, researchers typically rely on comprehensive formulation characterization (size, zeta potential, encapsulation efficiency), followed by rigorous in vitro release kinetics analysis in biologically relevant media (e.g., human serum) to assess premature leakage. Additionally, trigger-specific cleavage assays (e.g., exposure to specific proteases or varied pH) are essential to confirm the mechanism of conditional activation.
Demonstrating reduced off-target exposure requires precise PK/PD analysis capable of distinguishing the intact liposome-prodrug complex from the free, active drug in systemic circulation. A successful profile will show a high ratio of intact formulation in the plasma and decreased accumulation of the free drug in healthy clearance organs (like the liver, kidneys, or heart) compared to standard formulations, alongside confirmed efficacy at the target site.
Reference
- 1. Jiang, Yifan, et al. "Ultrasound-activated prodrug-loaded liposome for efficient cancer targeting therapy without chemotherapy-induced side effects." Journal of Nanobiotechnology 22.1 (2024): 2. https://doi.org/10.1186/s12951-023-02195-5
- 2. Under Open Access license CC BY 4.0, without modification.
Online Inquiry
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.


