Protecting Enzymatic Activity:
Liposomal Encapsulation Strategies for Enzymes
Advancing enzyme replacement therapies and catalytic nanomedicine by shielding biocatalysts from rapid in vivo degradation through precision liposomal engineering.
The Challenge: Enzymatic Instability In Vivo
Enzymes, as biocatalysts, possess exquisite specificity and catalytic efficiency, making them potent therapeutic agents for conditions ranging from lysosomal storage disorders to cancer. However, the successful clinical translation of enzyme therapies is severely hampered by their intrinsic fragility within the biological environment. When administered as "naked" proteins, enzymes face immediate threats that compromise their therapeutic utility.
The primary obstacle is rapid proteolytic degradation. Systemic circulation is replete with proteases that quickly hydrolyze therapeutic enzymes, leading to a drastically short half-life often measured in minutes. Furthermore, exogenous enzymes are frequently recognized as foreign antigens by the immune system, triggering the production of neutralizing antibodies (ADAs) which not only nullify efficacy but can also cause severe allergic reactions or anaphylaxis.
Additionally, enzymes typically possess poor membrane permeability, limiting their ability to reach intracellular targets. To overcome these barriers, robust delivery vehicles are essential. Liposomes, owing to their biocompatibility and versatility, have emerged as the gold standard for encapsulating enzymes, effectively isolating them from the hostile in vivo environment while preserving their catalytic function.
Mechanisms of Liposomal Protection
Steric Shielding and Stability
The lipid bilayer of the liposome acts as a physical barrier, preventing interaction between the encapsulated enzyme and serum proteases. This steric shielding significantly extends the enzyme's circulatory half-life. Furthermore, the aqueous core of the liposome provides a stabilizing microenvironment that can be buffered to the enzyme's optimal pH, protecting it from denaturation or conformational changes associated with physiological pH fluctuations.
Immune Evasion
By encapsulating the enzyme, liposomes mask the protein's antigenic epitopes from the host immune system. This "stealth" effect, often augmented by surface modification with polyethylene glycol (PEG), reduces opsonization by plasma proteins and delays clearance by the reticuloendothelial system (RES).
- ✓ Prevention of Proteolysis: Physical isolation from trypsin, chymotrypsin, and other serum proteases.
- ✓ Reduced Immunogenicity: Preventing the formation of anti-drug antibodies (ADAs).
- ✓ Controlled Release: Modulating lipid composition to trigger release at specific sites (e.g., tumor microenvironment).
- ✓ Conformational Stability: Maintaining the tertiary structure essential for catalytic activity.
Advanced Encapsulation Strategies
Achieving high encapsulation efficiency and maintaining enzyme activity requires a tailored approach to liposome preparation.
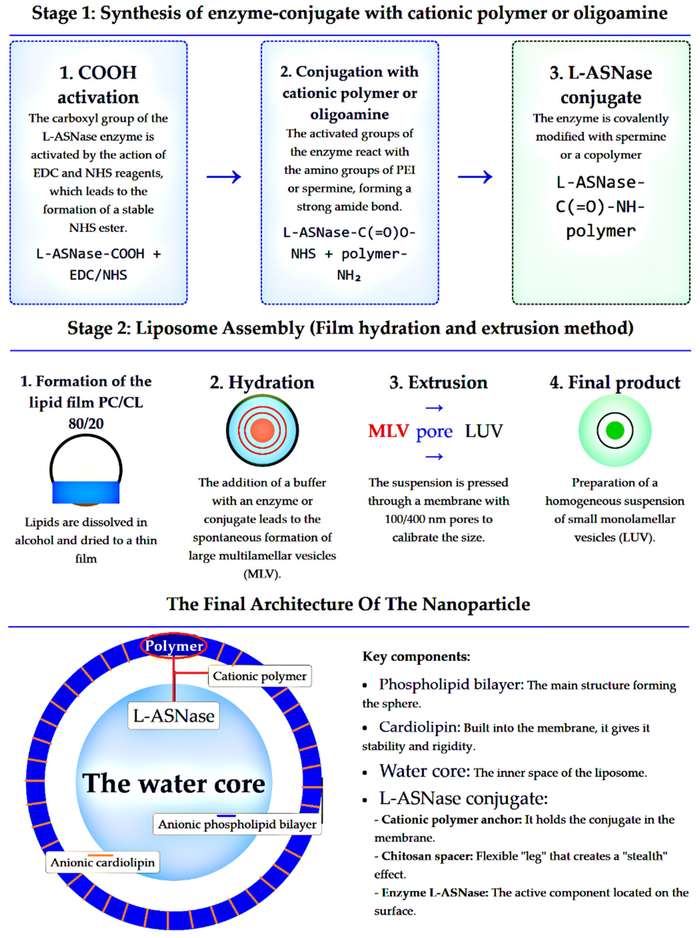
Passive vs. Active Loading
While small molecules can be actively loaded via pH gradients, enzymes, being macromolecules, are typically encapsulated via passive loading techniques such as thin-film hydration followed by extrusion, or reverse-phase evaporation. However, these methods often suffer from low encapsulation efficiency. Advanced strategies, such as the dehydration-rehydration vesicle (DRV) method or freeze-thaw cycles, are employed to increase the internal aqueous volume and thus enzyme entrapment.
Surface Engineering (PEGylation)
To prevent rapid clearance by the RES, "stealth" liposomes are created by grafting PEG chains onto the lipid surface. This hydration layer repels opsonins. However, excessive PEGylation can hinder cellular uptake (the "PEG dilemma"). Strategies such as cleavable PEG linkers or the use of semi-shielding configurations are critical for balancing circulation time with cellular internalization.
Stimuli-Responsive Release
For enzymes that need to act intracellularly or within specific microenvironments (e.g., lysosomes or tumors), stimuli-responsive liposomes are designed. These formulations utilize pH-sensitive lipids or enzyme-cleavable peptides to destabilize the membrane and release the payload upon specific triggers.
Optimization and Safety Evaluation
Lipid Composition
The choice of lipids determines membrane fluidity and stability. High transition temperature (Tm) lipids like DSPC are often combined with cholesterol to create rigid bilayers that resist leakage of the enzyme payload in vivo.
Physicochemical Characterization
Critical quality attributes (CQAs) such as particle size (typically 100-150 nm for tumor targeting), Polydispersity Index (PDI), and Zeta potential must be rigorously controlled to ensure batch-to-batch consistency and predictable pharmacokinetics.
Safety & Biocompatibility
Liposomes must be evaluated for potential cytotoxicity, hemocompatibility, and sterility. Ensuring the formulation does not induce complement activation pseudo-allergy (CARPA) is a key safety checkpoint.
Therapeutic Applications
Cancer Therapy (L-Asparaginase)
L-Asparaginase is a cornerstone in the treatment of acute lymphoblastic leukemia (ALL). However, its clinical use is limited by hypersensitivity reactions and a short half-life. Liposomal encapsulation has been shown to significantly reduce allergic reactions and extend the circulation time of L-Asparaginase, maintaining therapeutic blood levels with less frequent dosing.
Lysosomal Storage Diseases
In diseases like Gaucher's or Fabry's disease, enzymes are deficient within lysosomes. Liposomes can be surface-modified with ligands (e.g., mannose) to target specific cell receptors, facilitating endocytosis and subsequent delivery of the enzyme directly to the lysosome, replacing the missing function.
Antioxidant Therapy (SOD/Catalase)
Superoxide dismutase (SOD) and catalase rapidly degrade reactive oxygen species (ROS) but have poor cellular uptake. Liposomal co-encapsulation of these enzymes protects them from degradation and facilitates their delivery to sites of inflammation or oxidative stress, showing promise in treating conditions like rheumatoid arthritis.
Gout Management (Uricase)
Uricase converts uric acid to allantoin but is highly immunogenic in humans. Liposomal formulations, particularly PEGylated ones, shield uricase from immune detection, allowing for sustained reduction of plasma uric acid levels in refractory gout patients.
Frequently Asked Questions
Liposomes encapsulate enzymes within their aqueous core or lipid bilayer, creating a physical barrier that isolates the enzyme from the external environment. This steric shielding prevents interaction with serum proteases (such as trypsin and chymotrypsin) and neutralizing antibodies, thereby preserving the enzyme's structural integrity and catalytic activity while extending its circulation half-life.
Passive loading involves encapsulating the enzyme during the formation of the liposomes (e.g., during hydration of the lipid film). It is the most common method for macromolecules but can have variable efficiency. Active loading typically uses pH gradients to draw small molecules into pre-formed liposomes; however, for large enzymes, advanced passive techniques like Dehydration-Rehydration Vesicles (DRV) are often required to achieve high encapsulation efficiency without denaturing the protein.
Yes. One of the significant advantages of liposomal encapsulation is the masking of antigenic epitopes on the enzyme's surface. By hiding the enzyme from the host's immune surveillance, liposomes reduce the likelihood of Anti-Drug Antibody (ADA) formation and allergic reactions. PEGylation of the liposome surface further enhances this "stealth" effect by repelling opsonins.
Key parameters include lipid composition (using high-transition temperature lipids for stability), cholesterol content (for membrane rigidity), vesicle size (typically 100-200 nm for improved pharmacokinetics), surface charge (zeta potential), and the method of preparation to ensure high Encapsulation Efficiency (EE) without compromising enzyme activity.
Creative Biolabs offers comprehensive services ranging from custom lipid synthesis and formulation development to physicochemical characterization, in vitro release studies, and safety evaluations. We specialize in designing tailored liposomal systems (PEGylated, targeted, or stimuli-responsive) to maximize the therapeutic potential of your specific enzyme.
References
- Zlotnikov, Igor D., et al. "Liposomal Formulations of L-Asparaginase Conjugated with Cationic Polymers for Enhanced Internalization into Cancer Cells." Macromol 5.4 (2025): 54. https://doi.org/10.20944/preprints202509.0521.v1
- Under Open Access license CC BY 4.0, without modification.
Online Inquiry
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.


