Validating In Vitro Release Methods:
Dialysis vs. Sample Separation Techniques
Establishing accurate release profiles for liposomal formulations through robust methodological comparison and validation strategies in pharmacology.
Introduction to In Vitro Release Testing (IVRT)
In the realm of pharmacology and nanomedicine development, establishing accurate release profiles for colloidal drug carriers—specifically liposomes—is a critical quality attribute (CQA). Unlike conventional dosage forms, liposomes act as reservoirs that control the rate of drug availability. Consequently, validating methods to distinguish between encapsulated drug and free drug released into the media is paramount for predicting in vivo performance.
The complexity of IVRT lies in the physical separation of the released drug from the carrier without disrupting the equilibrium or integrity of the liposome. Currently, two primary methodological categories dominate the field: Dialysis (membrane diffusion-based) and Sample and Separate (physical separation based). Each method presents unique advantages and limitations regarding sink conditions, temporal resolution, and potential artifacts like the "burst release" effect or membrane adsorption.
For pharmacologists and formulation scientists, selecting the appropriate method is not merely a procedural choice but a strategic decision that impacts the reliability of pharmacokinetic (PK) predictions. Creative Biolabs provides comprehensive support in this domain, offering advanced Liposome In Vitro Release Analysis services to ensure your data meets rigorous regulatory standards.
Dialysis Techniques: The Diffusion Standard
Dialysis remains the most widely utilized technique for evaluating drug release from nanocarriers. The fundamental principle involves sequestering the liposomal formulation within a donor compartment separated from an acceptor (sink) compartment by a semi-permeable membrane. The Molecular Weight Cut-Off (MWCO) of the membrane is selected to retain the liposomes while allowing the free drug to diffuse passively into the acceptor media.
Key Variants
- Standard Dialysis Sac: The simplest form, where the formulation is placed in a dialysis bag floating in bulk media. While accessible, it often suffers from poor agitation and violation of sink conditions.
- Reverse Dialysis: The setup is inverted to increase the surface area-to-volume ratio, potentially accelerating the equilibrium process.
- Side-by-Side Diffusion Cells (Franz Cells): Offers precise temperature and agitation control with a defined membrane area, significantly improving reproducibility and minimizing the unstirred water layer effect.
However, pharmacologists must be wary of the "membrane effect"—where drug adsorption to the dialysis membrane or slow diffusion across it becomes the rate-limiting step, artificially masking the true release kinetics of the liposome.
Technical Insight: Sink Conditions
Maintaing sink conditions is non-negotiable for accurate IVRT. The concentration of the drug in the acceptor phase should not exceed 10-20% of its saturation solubility. Failure to maintain this gradient leads to back-diffusion and underestimation of release rates.
Mechanisms of Sample Separation
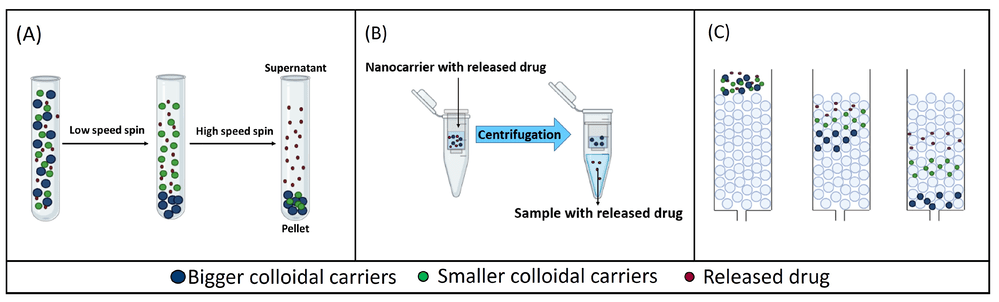
Recent assessments of in vitro release testing methods for colloidal drug carriers have highlighted a critical lack of standardized protocols, leading to variability in data interpretation across different studies. While dialysis remains a prevalent method due to its simplicity, it is often criticized for poor temporal resolution and potential violations of sink conditions due to membrane equilibration lag. Conversely, sample and separate techniques (Fig. 1) offer the advantage of direct quantification of the released drug but introduce their own set of challenges. The choice of separation force—whether it be high-speed centrifugation (A), pressurized filtration (B), or chromatographic separation (C)—can inadvertently alter the equilibrium state of the formulation. For instance, high shear forces in centrifugation may induce leakage in fragile vesicles, while ultrafiltration membranes can suffer from clogging or drug adsorption. Therefore, establishing a robust IVRT profile requires not just method selection, but a thorough validation that accounts for the specific physicochemical properties of the carrier system to avoid experimental artifacts.
Sample and Separate (S&S): Direct Quantification
The "Sample and Separate" methodology involves incubating the liposomes in release media and periodically withdrawing aliquots, which are then subjected to a physical separation process to isolate the released drug. This approach theoretically offers better temporal resolution and avoids the diffusion lag time associated with dialysis membranes.
Centrifugation
Effective for dense or large liposomes. Requires high-speed ultracentrifugation (e.g., >100,000 g) for small unilamellar vesicles (SUVs), which poses a risk of vesicle rupture (leakage) due to shear stress.
Ultrafiltration
Uses centrifugal concentrators. While fast, localized high concentrations at the membrane surface can cause polarization, clogging, or artificially induced leakage.
SEC (Gel Filtration)
The "gold standard" for gentleness. Using mini-columns allows for the separation of free drug in the void volume. The primary drawback is sample dilution and the time-consuming nature of processing multiple time points.
The choice of separation technique heavily influences the calculated PK profile. For complex formulations requiring precise correlation with biological outcomes, our PK-PD Analysis Services help bridge the gap between in vitro data and in vivo expectations.
Comparative Analysis: Dialysis vs. Sample Separation
| Feature / Parameter | Dialysis Method | Sample and Separate (S&S) |
|---|---|---|
| Principal Mechanism | Passive diffusion across a semi-permeable membrane. | Physical separation (density, size, or filtration). |
| Temporal Resolution | Low; limited by membrane diffusion rates (lag time). | High; "snapshot" of the release state at specific times. |
| Stress on Liposomes | Minimal; gentle environment. | Moderate to High (shear stress, pressure, g-force). |
| Sink Conditions | Easier to maintain with large acceptor volumes. | Challenging; often requires sample dilution or media replacement. |
| Suitability | Slow-release formulations; small drug molecules. | Rapid-release formulations; burst release detection. |
| Main Artifacts | Membrane binding; back-diffusion; osmotic pressure. | Incomplete separation; leakage induction; filter clogging. |
Validation Strategies and Best Practices
Regardless of the chosen method, validation is essential to prove that the IVRT method is discriminatory and reproducible. Key validation parameters include:
- Specificity: Demonstrating that the method measures the drug substance and not excipients or degradation products.
- Linearity and Range: Ensuring the detector response is linear over the range of drug concentrations released.
- Accuracy and Precision: Calculating recovery rates (typically 90-110%) and inter-day/intra-day variability.
- Robustness: Assessing the impact of small variations in pH, temperature (37°C ± 0.5°C), and agitation speed.
Furthermore, distinguishing "burst release" (surface-associated drug) from sustained release requires high-resolution sampling in the first few hours. Creative Biolabs employs a hybrid approach when necessary, utilizing S&S for initial time points to capture burst effects and Dialysis for long-term profiling, ensuring a comprehensive understanding of the formulation's behavior.
Frequently Asked Questions
The primary difference lies in how the released drug is isolated. Dialysis uses a semi-permeable membrane to allow passive diffusion of the free drug into an acceptor compartment, which is gentle but slow. Sample and separation (S&S) techniques actively separate the drug from liposomes using forces like centrifugation or filtration, offering faster results but potentially stressing the liposomes.
Sink conditions are maintained when the volume of the release medium is at least 3 to 10 times the saturation volume of the drug. This ensures that the drug concentration in the bulk media is negligible, preventing back-diffusion and ensuring that the release rate is controlled by the formulation, not the solubility limit.
Yes. High centrifugal forces (g-forces) required to pellet small liposomes can deform the lipid bilayer, leading to induced leakage of the encapsulated drug. This artifact can result in an overestimation of the release rate. Optimization of speed and time is crucial for validation.
Burst release refers to the rapid release of drug immediately upon administration, often due to drug adsorbed on the liposome surface. An accurate measurement is vital for safety (avoiding toxicity spikes) and efficacy (ensuring enough drug remains for sustained delivery). S&S methods are typically better at detecting this than dialysis.
There is no single "better" method; it depends on the administration route and drug properties. However, dialysis is often preferred for simulating the slow clearance in the bloodstream, while specific S&S setups can better mimic rapid dilution or cellular interaction environments. A combination of data often yields the best IVIVC.
References
- Gómez-Lázaro, Laura, et al. "Assessment of in vitro release testing methods for colloidal drug carriers: the lack of standardized protocols." Pharmaceutics 16.1 (2024): 103. https://doi.org/10.3390/pharmaceutics16010103
- Under Open Access license CC BY 4.0, without modification.
Online Inquiry
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.


