Why Liposomes Fail in Skin Applications:
A Practical Troubleshooting Guide
Learn why dermal liposome formulations separate, penetrate poorly, or show dramatic size growth after lyophilization—and how to troubleshoot the likely root causes.
If your liposomal gel separates within days, your API remains trapped in the stratum corneum, or your vesicle size jumps after freeze-drying, the issue is often not a single parameter—it is the interaction between composition, deformability, process conditions, and analytical controls. This guide highlights three common failure patterns in skin liposome development and practical ways to troubleshoot them.
Beyond the Beaker: Designing for the Stratum Corneum
Formulating liposomes for topical or transdermal delivery is a complex challenge. Dermal delivery is strictly limited by the stratum corneum—the outermost layer of the skin that acts as an efficient barrier against foreign substances. For a liposomal formulation, success depends heavily on vesicle size, membrane flexibility, and thermodynamic stability.
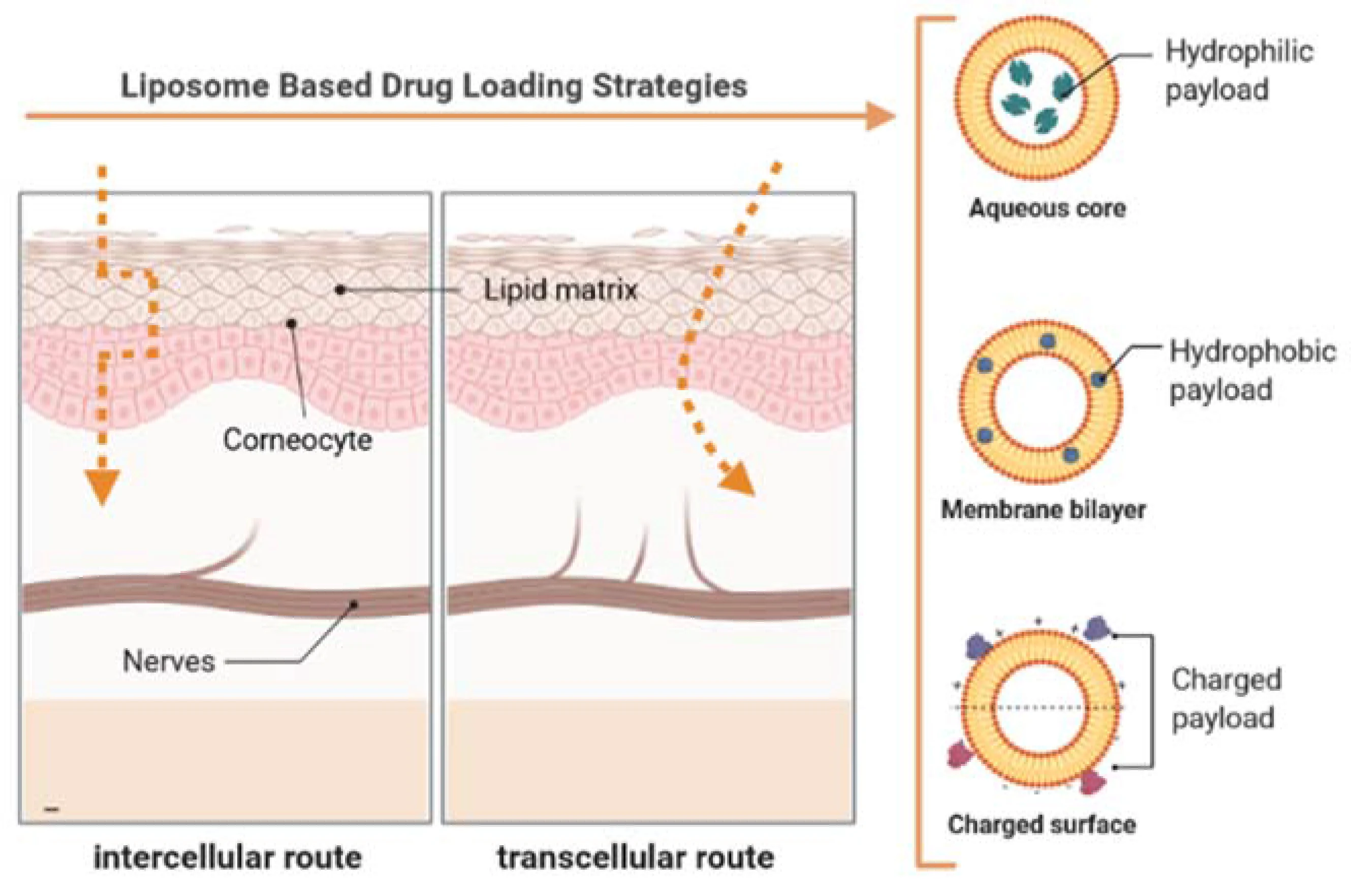
When physical instability occurs in your formulation—such as phase separation, aggregation, or severe particle growth—it often translates directly to poor permeation performance in in vitro, in vivo, and ex vivo models. The formulation must be inherently designed to remain stable while migrating along the restrictive intercellular lipid pathways shown above.
To ensure your vesicles are inherently designed for this challenging environment, explore our comprehensive Liposomes for Dermal Delivery services, tailored to cross the stratum corneum efficiently.
Troubleshooting Phase Separation
1 What users usually observe
- Clear supernatant forming above a cloudy lipid layer, or visible sedimentation.
- Gel thinning or local clumping after incorporating liposomes into a cream or hydrogel base.
- Consistently rising particle size and Polydispersity Index (PDI) during stability storage.
2 Likely root causes
- Insufficient stabilization: Lack of electrostatic repulsion (zeta potential too close to neutral) or inadequate steric hindrance.
- Polymer incompatibility: Formulating oppositely charged liposomes with gelling polymers or electrolytes (e.g., cationic vesicles in an anionic carbomer matrix).
- Chemical degradation: Lipid hydrolysis or oxidation during storage alters the bilayer geometry, prompting precipitation.
What to check first
- Size and PDI trend: Confirm if flocculation happens immediately during mixing or gradually over time.
- Zeta potential: Evaluate the charge profile across different buffer ionic strengths.
- pH drift: A dropping pH often indicates active lipid hydrolysis.
- Polymer/buffer compatibility: Check for interactions between the liposomal charge and the secondary formulation base.
Troubleshooting Poor Permeation
A common frustration during ex vivo Franz diffusion cell assays is discovering that the active pharmaceutical ingredient (API) has remained entirely on the surface of the skin or within the upper layers of the stratum corneum, completely failing to reach the targeted deeper dermal tissues.
Conventional liposomes may show limited penetration beyond the upper skin layers, especially when vesicle rigidity, composition, and formulation architecture are not optimized for dermal delivery. Because the intercellular spaces of the stratum corneum are exceedingly narrow, rigid vesicles often cannot squeeze through. Instead, they may dehydrate and fuse with the skin surface, releasing the API prematurely.
For some applications, more deformable systems such as transferosomes or ethosomes may improve delivery performance, but the optimal strategy depends on the API, target skin depth, and formulation context. In many cases, incorporating specific edge activators—single-chain surfactants like sodium cholate or polysorbates—can lower the bending modulus of the lipid bilayer. This elasticity often allows the vesicle to squeeze through pores much smaller than its own diameter, driven by the natural transdermal hydration gradient.
Mitigating Lyophilization Stress
Because aqueous liposome dispersions are prone to hydrolysis over extended shelf lives, lyophilization (freeze-drying) is a standard preservation technique. However, it is a high-stress process for lipid membranes. A formulation that measured a pristine 100 nm with a low PDI before freezing may emerge from reconstitution with major particle growth or a substantial loss of vesicle integrity.
This severe destabilization is primarily caused by ice crystal formation and the removal of the hydration shell during the freezing and primary drying phases. Without their protective water layer, lipid bilayers are forced into close proximity, frequently leading to massive vesicle fusion and the leakage of encapsulated water-soluble drugs.
Suggested checks after reconstitution
- Compare size, PDI, and zeta potential before freezing vs. after reconstitution.
- Check encapsulation retention / assess API leakage rates.
- Review cryoprotectant type (e.g., trehalose, sucrose) and the lipid-to-sugar mass ratio.
- Review freezing rate, annealing time, and primary drying conditions.
- Confirm whether the reconstitution medium matches the intended ionic environment.
Identify exactly where the physical changes occur using advanced dynamic light scattering (DLS) and thermal analysis.
View Lipid-based Basic Characterization ServicesFormulation Troubleshooting Matrix
| Observed issue | What it may indicate | What to review first | Potential optimization direction |
|---|---|---|---|
| Rapid Phase Separation | Insufficient electrostatic repulsion or lipid hydrolysis. | Zeta potential; pH drift over time. | Incorporate charged lipids; ensure storage buffer is strictly pH controlled. |
| High PDI after loading into gel / cream base | Incompatibility between vesicle charge and polymer network, leading to local flocculation. | Charge matching between liposomes and gelling agents. | Switch to non-ionic polymers or adjust the zeta potential of the vesicles. |
| API Stagnates in Stratum Corneum | Membrane is too rigid to deform and pass intercellular junctions. | Cholesterol ratio; presence of penetration enhancers. | Reduce cholesterol; introduce edge activators or ethanol to increase deformability. |
| Size Spike post-Lyophilization | Vesicle fusion due to loss of hydration shell and ice crystal shearing. | Lipid-to-sugar ratio; freezing profile. | Increase cryoprotectant concentration; optimize the annealing step in the freeze-drying cycle. |
Frequently Asked Questions
Phase separation upon gel incorporation often results from a charge incompatibility between the liposome surface and the gelling polymer network. For instance, mixing cationic liposomes with an anionic carbomer matrix can cause instant complexation and precipitation. Poor initial zeta potential can also lead to flocculation under the stress of mixing.
If your API is trapped in the upper layers, it may indicate that your vesicles are too rigid. Conventional liposomes with high cholesterol content often behave as inflexible spheres and cannot squeeze through the narrow intercellular spaces of the stratum corneum, causing them to dehydrate and release the API prematurely on the surface.
Not always. It heavily depends on your therapeutic goal. If you aim for local depot formation within the upper skin layers, conventional liposomes may be sufficient. However, if deep dermal or transdermal delivery is required, ultradeformable systems (like transferosomes) or ethanol-containing systems (ethosomes) often provide superior penetration by increasing membrane flexibility and fluidizing skin lipids.
Dramatic size increases usually result from the removal of the lipid hydration shell and the physical shearing caused by ice crystals during lyophilization. Without adequate cryoprotectants (like trehalose or sucrose) to stabilize the membrane via hydrogen bonding, vesicles are forced into close proximity, leading to widespread fusion and aggregation.
A combination of dynamic light scattering (DLS) for particle size and PDI, zeta potential analysis for colloidal stability, and ex vivo Franz diffusion studies for permeation profiling is essential. Creative Biolabs can leverage these tools alongside formulation optimization services to identify exactly where your system fails and help re-engineer the lipid composition.
Reference
1. Souto, Eliana B., et al. "Elastic and ultradeformable liposomes for transdermal delivery of active pharmaceutical ingredients (APIs)." International Journal of Molecular Sciences 22.18 (2021): 9743. https://doi.org/10.3390/ijms22189743
2. Under Open Access license CC BY 4.0, without modification.
Online Inquiry
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.


