The Ultimate Guide to Liposome Preparation: Thin-Film Hydration vs. Ethanol Injection
A comprehensive resource for beginners and lab-scale researchers seeking the most effective methods for liposomal formulation.
Introduction to Liposome Preparation
Liposomes, spherical vesicles composed of one or more lipid bilayers, have revolutionized the field of drug delivery. Their unique ability to encapsulate both hydrophilic and hydrophobic compounds makes them invaluable tools for increasing the therapeutic index of drugs, reducing toxicity, and enabling targeted delivery. For beginners in the laboratory, selecting the right preparation method is the first and most critical step in establishing a reliable formulation workflow.
The journey from a laboratory concept to clinical application relies heavily on the reproducibility and scalability of these initial methods. As noted by Senjab et al. in their recent review "Advances in liposomal nanotechnology: From concept to clinics"1, the field has seen a significant shift towards methods that offer better control over size and polydispersity. While numerous techniques exist—ranging from reverse-phase evaporation to detergent depletion—two methods stand out for their accessibility and foundational importance in lab-scale environments: Thin-Film Hydration (TFH) and Ethanol Injection (EI).
This guide provides a detailed comparison of these two fundamental techniques, helping you navigate the trade-offs between equipment requirements, encapsulation efficiency, and scalability to find the best fit for your research needs.
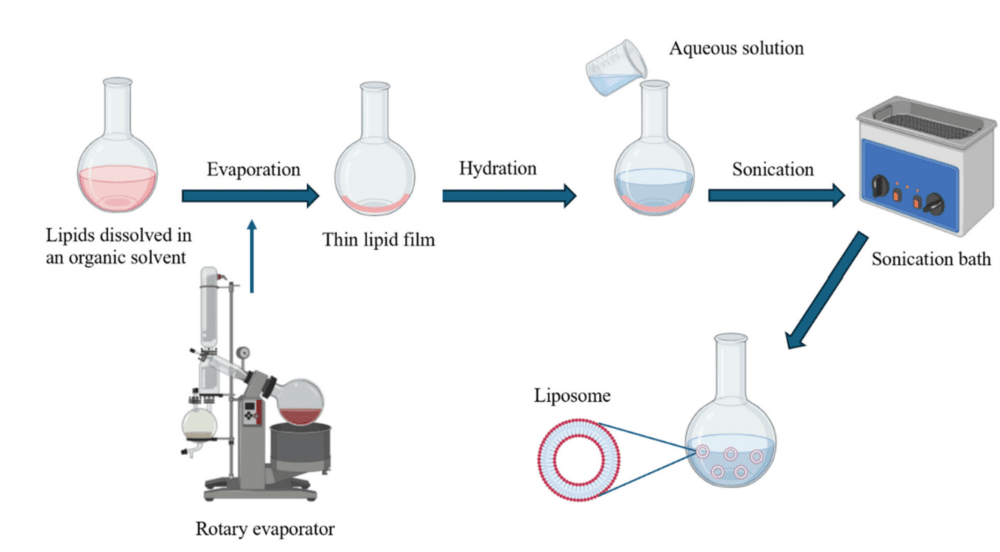
Thin-Film Hydration: The Classical Approach
Mechanism and Process
The Thin-Film Hydration (TFH) method, also known as the Bangham method, was the first technique developed for liposome preparation and remains the gold standard for small-scale production in academic labs. The process begins with dissolving lipids in an organic solvent (typically chloroform or methanol) in a round-bottom flask. The solvent is then evaporated using a rotary evaporator, leaving a thin, homogenous lipid film on the flask wall.
Upon addition of an aqueous buffer (hydration phase) at a temperature above the phase transition temperature (Tc) of the lipids, the film swells and detaches, spontaneously forming large multilamellar vesicles (MLVs). As depicted in Fig.1, these initial MLVs are often heterogeneous in size and require downstream processing—such as sonication or extrusion—to produce uniform unilamellar vesicles desirable for most biological applications.
Pros and Cons
- Advantages: Simple equipment requirements (rotary evaporator), typically high encapsulation efficiency for hydrophobic drugs (lipid bilayer), and moderate-to-high efficiency for hydrophilic drugs (aqueous core) depending on the hydration volume. Compatibility with a wide range of lipid compositions.
- Limitations: Difficult to scale up, potential for residual solvent, and the formation of heterogeneous populations requiring vigorous sizing steps.
Quick Start: TFH Protocol Basics
- Target Size: 100–200 nm (post-extrusion)
- Recommended Lipid Conc: 5–20 mg/mL
- Key Equipment: Rotary Evaporator, Extruder or Probe Sonicator
- Success Criteria: PDI < 0.2, Translucent appearance (for SUVs)
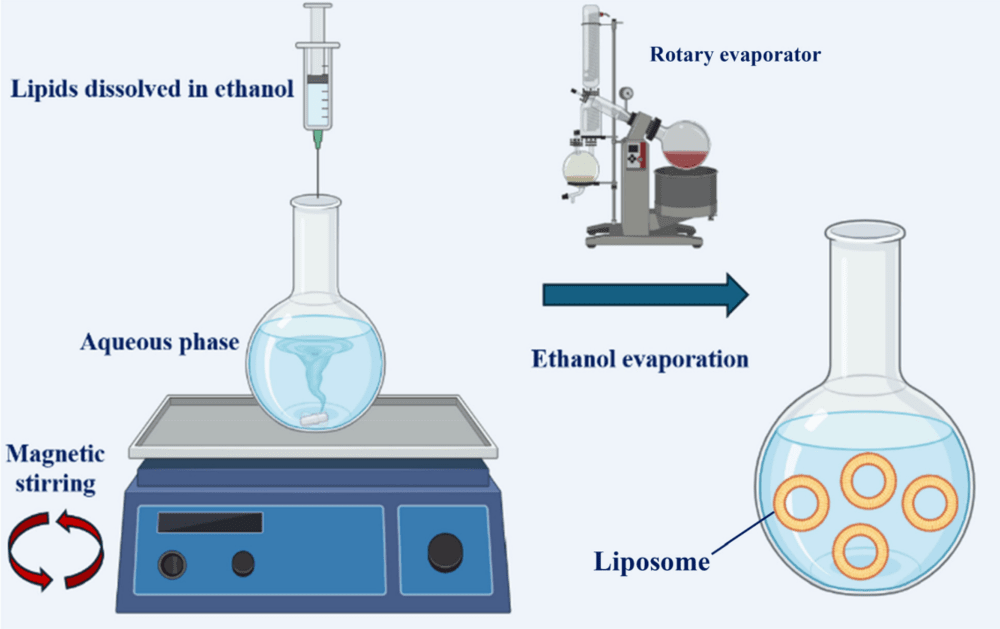
Ethanol Injection: The Scalable Alternative
Mechanism and Process
Ethanol Injection offers a more streamlined approach that is inherently better suited for scale-up. In this method, lipids are dissolved in ethanol and then injected rapidly into a stirring aqueous phase. The rapid dilution of ethanol causes the lipids to precipitate and self-assemble into vesicles.
As illustrated in Fig.2, this technique typically yields small unilamellar vesicles (SUVs) directly, often bypassing the need for extensive downsizing techniques like sonication. The critical parameters controlling size include the injection speed, lipid concentration, and stirring rate. This method is particularly favored in industrial settings due to its reproducibility and the ease of removing ethanol via dialysis or diafiltration.
Pros and Cons
- Advantages: Scalable, produces smaller and more uniform vesicles spontaneously, simpler process control, and reduced risk of lipid degradation compared to harsh sonication.
- Limitations: Limited lipid solubility in ethanol for some species, necessity of ethanol removal step, and typically moderate encapsulation efficiency for hydrophilic cargos due to the dilution effect in the aqueous phase.
Quick Start: EI Protocol Basics
- Target Size: 80–150 nm (spontaneous)
- Recommended Lipid Conc: 5–10 mg/mL (final aqueous conc)
- Key Equipment: Syringe Pump (for reproducibility), Magnetic Stirrer, Dialysis Tubing
- Success Criteria: PDI < 0.2, Minimal aggregates
Comparative Analysis: Decision Matrix
| Feature | Thin-Film Hydration | Ethanol Injection | Best When You... | Watch-outs |
|---|---|---|---|---|
| Primary Vesicle Type | Multilamellar Vesicles (MLVs) | Small Unilamellar Vesicles (SUVs) | Need specific lamellarity control | MLVs require sizing steps |
| Particle Size Control | Requires extrusion/sonication | Controlled by injection speed & mixing | Want tight size distribution via extrusion | Manual injection can cause variability |
| Scalability | Low (Batch limited by surface area) | High (Continuous process feasible) | Developing industrial process | Solvent removal capacity limits scale |
| Encapsulation (Hydrophilic) | Generally Higher (Variable) | Moderate (Dilution effect) | Have expensive/scarce cargo | May need active loading for high EE |
| Organic Solvent | Chloroform/Methanol (Toxic) | Ethanol (Less toxic) | Prioritize green chemistry | Ensure complete removal (dialysis) |
Troubleshooting Guide for Beginners
Problem: High PDI (>0.3)
- Cause: Lipid concentration too high, insufficient mixing speed (EI), or too few passes through extruder (TFH).
- Solution: Reduce lipid concentration, increase stirring rate during injection, or increase extrusion passes (min 11 times).
Problem: Size Drift / Batch Variability
- Cause: Process temperature below Phase Transition Temperature (Tc), inconsistent manual injection speed.
- Solution: Ensure all steps are performed >10°C above Tc. Use a syringe pump for EI to standardize flow rate.
Problem: Low Encapsulation Efficiency
- Cause: Passive loading limitations for hydrophilic drugs (especially in EI).
- Solution: Switch to Thin-Film Hydration for higher passive loading, or implement active (remote) loading techniques (e.g., pH gradient).
Problem: Residual Solvent / Toxicity
- Cause: Insufficient evaporation or dialysis time.
- Solution: Use extended vacuum drying (overnight) for TFH. Perform thorough dialysis with multiple buffer changes for EI. Verify with GC.
Characterization: From Lab Bench to Publication
Regardless of the method chosen, the final liposomal product must be rigorously characterized. As highlighted in recent literature discussing the assessment of in vitro release testing methods for colloidal carriers, standardized protocols are essential for reproducibility.
Different preparation methods can lead to variations in bilayer packing density and lamellarity, which directly impact how a drug is released in vitro and in vivo. Researchers must establish robust QC gates early in the process.
Need a Control for Your Experiments?
Establishing a baseline is crucial for validation. Creative Biolabs offers high-quality plain liposomes as negative controls.
View Plain Liposome Products →Essential Characterization (The "Release Gate")
- Size (Z-average) & PDI: Measured via Dynamic Light Scattering (DLS). Target PDI < 0.2 for homogeneity.
- Zeta Potential: Indicates surface charge and colloidal stability.
- Appearance: Visual check for turbidity or precipitates.
- Encapsulation Efficiency (EE%): Quantification of free vs. encapsulated drug.
Advanced Characterization (For Publication/Scale-up)
- Morphology: Cryo-TEM to confirm unilamellar structure.
- Lamellarity: NMR or SAXS analysis.
- Residual Solvent: Gas Chromatography (GC) to ensure safety.
- In Vitro Release: Dialysis or sample-and-separate methods to determine release kinetics.
Frequently Asked Questions
The main difference lies in the size and lamellarity of the vesicles formed. Thin-Film Hydration (TFH) typically yields large, multilamellar vesicles (MLVs) that require downsizing (e.g., extrusion) to become uniform. Ethanol Injection usually produces small unilamellar vesicles (SUVs) spontaneously due to the rapid dilution of the solvent, making it more suitable for generating smaller particles without extensive mechanical processing.
For Thin-Film Hydration, the bulk solvent is removed via rotary evaporation, often followed by overnight storage under vacuum to remove trace residues. For Ethanol Injection, the ethanol is typically removed via dialysis against a buffer or by using diafiltration (tangential flow filtration) if working with larger volumes.
The phase transition temperature (Tc) is the temperature at which the lipid bilayer changes from a rigid gel phase to a fluid liquid-crystalline phase. Hydration must be performed above the Tc of the lipid with the highest melting point in your mixture to ensure the lipids are fluid enough to form sealed, flexible vesicles.
Yes, both methods can encapsulate hydrophilic drugs. In TFH, the drug is dissolved in the aqueous hydration buffer. In Ethanol Injection, the drug is also in the aqueous phase, but the encapsulation efficiency is often lower compared to TFH because the large volume of aqueous buffer dilutes the drug relative to the lipid volume.
Ethanol Injection is generally superior for scaling up. It is a continuous process that is easier to control using automated pumps and mixers, whereas TFH is a batch process limited by the surface area of the flask used for evaporation.
References
- Senjab, Reem M., et al. "Advances in liposomal nanotechnology: From concept to clinics." RSC Pharmaceutics 1.5 (2024): 928-948. https://doi.org/10.1039/D4PM00176A
- Under Open Access license CC BY 4.0, without modification.
Online Inquiry
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.


