How to Design Stealth Liposomes for Long Circulation:
PEGylation Principles & Formulation Strategies
A comprehensive development guide to mastering PEGylation: control shedding kinetics, avoid rapid clearance, and optimize pharmacokinetics for your liposomal formulation.
Principles of Stealth Technology: Why Modification is Necessary
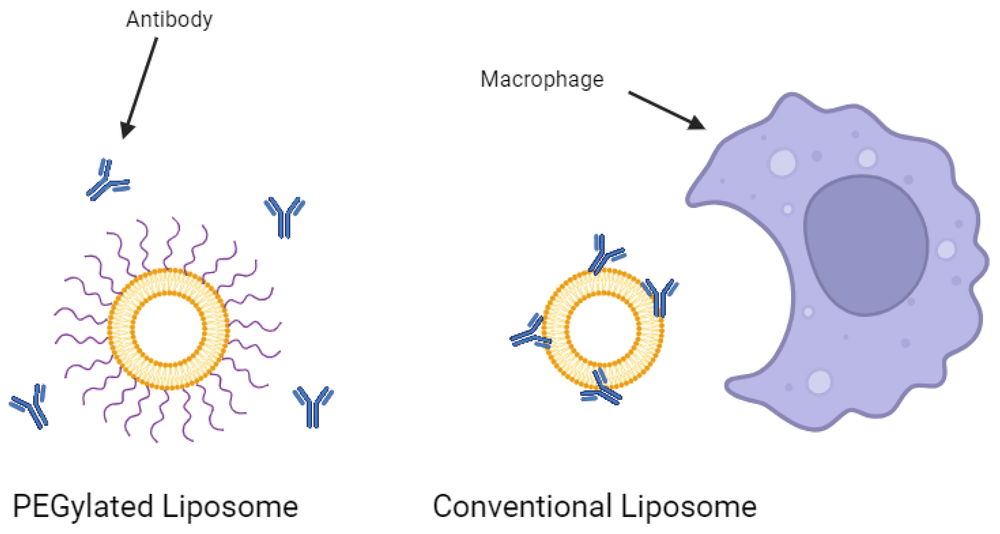
Conventional liposomes, largely composed of natural phospholipids and cholesterol, face a fundamental biological barrier: rapid clearance by the Reticuloendothelial System (RES). Upon intravenous administration, unmodified liposomes are almost immediately opsonized by plasma proteins (opsonins), marking them for phagocytosis by Kupffer cells in the liver and macrophages in the spleen. This results in a circulation half-life often measured in minutes, severely limiting their potential for systemic drug delivery.
The solution lies in surface modification. By grafting hydrophilic polymers—most commonly Polyethylene Glycol (PEG)—onto the lipid bilayer, we create "Stealth Liposomes." This polymer coating provides a hydrated steric barrier that repels opsonins, effectively rendering the liposomes invisible to the immune surveillance system.
Conventional Liposomes
- • Surface: Naked phospholipid bilayer.
- • Interaction: Rapid opsonization by blood proteins.
- • Fate: Quick uptake by MPS (Liver/Spleen).
- • Half-life: Minutes to < 1 hour.
Stealth (PEGylated) Liposomes
- • Surface: Hydrated PEG polymer brush layer.
- • Interaction: Steric repulsion prevents protein binding.
- • Fate: Evades MPS; accumulates in tumors (EPR effect).
- • Half-life: Up to 48+ hours (e.g., PEGylated liposomal doxorubicin).
Mechanism of Steric Stabilization
The "Stealth" effect is not merely a coating; it is a thermodynamic barrier. PEG chains are highly hydrophilic and flexible. When grafted onto the liposome surface, they recruit a dense "cloud" of water molecules. If a protein (opsonin) attempts to approach the liposome surface, it must compress these hydrated PEG chains. This compression reduces the conformational entropy of the PEG, generating a repulsive force that physically blocks the protein from contacting the lipid bilayer.
Critical Design Variables for PEGylated Liposomes
Successful stealth liposome development requires strategic control of PEG anchor stability, polymer length, and surface density. Simply adding PEG-lipid without optimization often leads to suboptimal PK performance or instability.
1. The Lipid Anchor: Stability vs. Shedding
The hydrophobic anchor determines the residence time of the PEG chain on the liposome. For long-circulating formulations, a stable anchor is non-negotiable. However, for applications requiring intracellular delivery, a "sheddable" PEG may be advantageous to facilitate endosomal escape.
| Anchor Type | Characteristics | Strategic Application |
|---|---|---|
| DSPE (C18 Saturated) | High phase transition temperature; extremely stable insertion. | The Gold Standard for Long Circulation. |
| DMPE / DPPE (C14/C16) | Shorter chains; faster exchange rate with serum components. | Used when faster PEG shedding is required to expose the liposome surface. |
| Ceramides (C8-C20) | Rate of diffusion out of bilayer depends heavily on chain length. | "Programmable" stealth; enables time-dependent de-shielding. |
2. Polymer Length & Molecular Weight
PEG-2000 is empirically the optimal molecular weight. Shorter chains (PEG < 1000) fail to form a thick enough steric barrier to mask surface charges effectively. Longer chains (PEG > 5000) occupy excessive volume, potentially inducing micelle formation or hindering cellular uptake at the target site.
3. Surface Density (The "Brush" Regime)
To achieve true stealth, PEG chains must extend outward in a "brush" conformation rather than coiling near the surface ("mushroom" conformation). This is typically achieved at 5-10 mol% PEG-lipid.
Note: Accurate PEG surface density quantification is critical but technically challenging. Creative Biolabs offers advanced analytical services to validate your surface modification.
Explore Formulation Analysis & Characterization Services »Common Development Challenges in PEGylated Liposomes
Why does my stealth liposome fail in vivo despite looking perfect in vitro?
PEG Shedding Kinetics
If the lipid anchor is mismatched with the bilayer fluidity, PEG may desorb too quickly in blood, leading to premature clearance.
The ABC Phenomenon
Repeat dosing can trigger "Accelerated Blood Clearance" via anti-PEG antibodies, rendering the second dose ineffective.
Ligand Shielding
Attaching targeting ligands to the liposome surface is futile if the dense PEG layer buries them, preventing receptor binding.
Quantification Difficulty
Determining the exact mol% of PEG incorporated versus free PEG requires specialized analytical methods (e.g., NMR, HPLC-ELSD).
Batch-to-Batch Variability
Inconsistent PEGylation density can lead to drastic differences in PK profiles between production lots.
Unexpected Liver Uptake
Even with PEG, changes in size or zeta potential during storage can trigger recognition by Kupffer cells.
These challenges often require integrated formulation design and rigorous in vivo validation.
Industry Standard Formulation Strategy
Most successful long-circulating liposome projects begin with a proven baseline configuration. We recommend starting here before customizing for specific needs.
- ✔ PEG-Lipid: DSPE-mPEG-2000
- ✔ Density: 5 – 10 mol%
- ✔ Lipid Matrix: HSPC/Cholesterol or DSPC/Cholesterol (High Tm)
Looking for high-quality reagents? Browse our PEGylated Lipids Catalog »
Need Customization?
We synthesize custom lipid conjugates and optimize formulations for unique payloads.
Explore Development ServiceFrom Concept to Clinic: Translating Stealth Technology
While the principles of PEGylation are well-established, translating a stealth liposome from a concept to a viable therapeutic candidate requires rigorous optimization. Factors such as lipid composition, PEG shedding kinetics, drug retention, and manufacturing scalability must be balanced perfectly.
Furthermore, emerging challenges like the ABC phenomenon in repeat dosing regimes require advanced strategies, such as the use of cleavable PEG systems or alternative hydrophilic polymers. Our team evaluates ABC risk in preclinical PK studies and proposes robust mitigation strategies.
Why Choose Creative Biolabs?
- ★ End-to-End Support: From custom lipid synthesis to in vivo PK validation.
- ★ Advanced Characterization: Precise measurement of PEG layer thickness and density.
- ★ Problem-Solving: Expert consultation on shedding, stability, and ABC issues.
Frequently Asked Questions
The Accelerated Blood Clearance (ABC) phenomenon occurs upon repeated administration of PEGylated liposomes. The first dose stimulates the production of anti-PEG IgM antibodies. When a second dose is administered several days later, these antibodies bind to the PEG layer, triggering rapid complement activation and uptake by Kupffer cells in the liver, drastically reducing circulation time. Researchers are actively developing alternative polymers (like poly(sarcosine) or zwitterionic materials) to mitigate this issue.
DSPE-PEG2000 is favored because the DSPE (distearoyl) anchor has two saturated C18 chains, which provide a high phase transition temperature and strong hydrophobic interaction with the lipid bilayer, preventing premature shedding. The PEG2000 chain length offers the optimal balance between forming a dense steric brush layer and minimizing the total volume of the polymer, which helps maintain liposome stability and reduces micelle formation.
Yes. This is often done by using heterobifunctional PEG-lipids (e.g., DSPE-PEG-Maleimide or DSPE-PEG-NHS). The ligand (antibody, peptide, or aptamer) is conjugated to the distal end of the PEG chain. This allows the ligand to extend beyond the steric barrier of the PEG layer ("cloud") to interact with cell surface receptors, a strategy known as active targeting.
Generally, yes. The presence of a hydrated PEG layer can reduce the permeability of the lipid bilayer, thereby slowing down the leakage of encapsulated hydrophilic drugs. However, for drugs that need to be released rapidly at the target site (e.g., inside a tumor cell), this stability can be a hindrance. Strategies such as using "sheddable" PEG-lipids or pH-sensitive lipids are employed to trigger release specifically within the target environment.
References
- Gatto, Matthew S., McNeely P. Johnson, and Wided Najahi-Missaoui. "Targeted liposomal drug delivery: Overview of the current applications and challenges." Life 14.6 (2024): 672. https://doi.org/10.3390/life14060672
- Under Open Access license CC BY 4.0, without modification.
Online Inquiry
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.


