A Guide to Transfersome Development: Edge Activators, Size Optimization, and Permeation Testing
A practical development framework for formulation scientists working on deformable vesicle systems for topical and transdermal delivery.
What Are Transfersomes, and Why Does Their Development Require a Structured Approach?
Note: On this page, "transfersome" is used as a general term for ultra-deformable phospholipid vesicles containing edge activators.
Transfersomes are ultra-deformable phospholipid vesicles formulated with edge activators to improve flexibility relative to conventional liposomes. They are widely investigated for topical and transdermal delivery, especially where skin penetration, payload retention, and formulation adaptability must be balanced across a range of hydrophilic and lipophilic actives.
In practice, transfersome performance is shaped not by a single parameter but by the interaction among surfactant selection, vesicle size distribution, colloidal stability, drug loading, and membrane permeation behavior. In some cases, a smaller vesicle population does not outperform a slightly larger but better-balanced formulation when membrane flexibility, loading, and release behavior are not optimized together.
This guide outlines a practical development framework covering three interconnected areas: edge activator screening, size and PDI optimization, and Franz diffusion cell permeation validation.
Explore Our Transfersome Development ServiceKey Formulation Variables in Transfersome Development
| Variable | Impact on Performance |
|---|---|
| Edge activator type | Membrane flexibility, drug release profile |
| EA molar ratio | Deformability, vesicle stability |
| Vesicle diameter (Z-avg) | Skin penetration pathway |
| PDI | Batch reproducibility and shelf-life |
| Gel/carrier viscosity | Drug release rate, spreadability |
| Encapsulation efficiency | Dose accuracy, payload retention |
Who This Guide Is For
Pharmaceutical formulation scientists, dermal delivery researchers, and CRO teams building or troubleshooting transfersome systems who need technical clarity from lipid selection through permeation validation.
Edge Activators: Selection Criteria and Comparative Screening
The choice of edge activator is among the most consequential formulation decisions. Different surfactants produce significantly different biophysical outcomes—even when vesicle size is held constant.
How Edge Activators Affect Membrane Behavior
Edge activators intercalate into the phospholipid bilayer and reduce membrane bending rigidity, enabling vesicle deformation under the osmotic gradient between the hydrated skin surface and deeper dermal layers. The specific surfactant identity, its hydrophile-lipophile balance (HLB), and its molar concentration each shape encapsulation efficiency, physical stability, and in vitro drug permeation behavior.
Commonly studied edge activators include Span 80 (Sorbitan monooleate), Tween 80 (Polysorbate 80), sodium deoxycholate (SDC), sodium cholate, and sucrose laurate. The optimal candidate is drug- and system-dependent. Bile salt-based agents like SDC may enhance permeation at lower concentrations for hydrophilic drugs, while nonionic surfactants like Span 80 are often screened across a broader molar range for lipophilic payloads.
EA levels are typically screened across a defined range rather than fixed at a universal optimum. Regardless of which candidates are evaluated, all should be compared under identical experimental conditions to ensure interpretable results.
| Edge Activator | Type | HLB | Key Characteristic |
|---|---|---|---|
| Span 80 | Nonionic | 4.3 | High deformability potential; often used for lipophilic drugs |
| Tween 80 | Nonionic | 15.0 | Good skin tolerability; hydrophilic drug compatibility |
| Sodium Deoxycholate | Anionic | >20 | Strong membrane disruption; high reported flux in some models |
| Sodium Cholate | Anionic | >20 | Milder disruption than SDC; generally favorable stability |
| Sucrose Laurate | Nonionic | ~15 | Biocompatible; emerging interest in cosmetic applications |
HLB values are approximate. Selection should be guided by the specific drug-lipid-surfactant system.
The Deformability Index: A Comparative Screening Parameter
The Deformability Index (DI) is one of the most commonly used comparative parameters during early transfersome screening. It is measured by extruding a vesicle suspension through a defined membrane under controlled pressure; the resulting DI reflects the ratio of extruded material to vesicle size change. A higher DI suggests greater membrane elasticity—but because DI values depend strongly on test setup, membrane pore size, and calculation method, internal benchmarking within the same experimental system is generally more informative than comparison against a fixed absolute cutoff.
DI should always be evaluated alongside encapsulation efficiency, size, PDI, and zeta potential. A formulation showing favorable deformability but poor EE or elevated PDI may not deliver the expected permeation advantage. Creative Biolabs' lipid-based characterization services combine these assessments into a single, streamlined package for multivariate screening.
Edge Activator Choice Drives Permeation Outcomes: Evidence from In Vitro Data
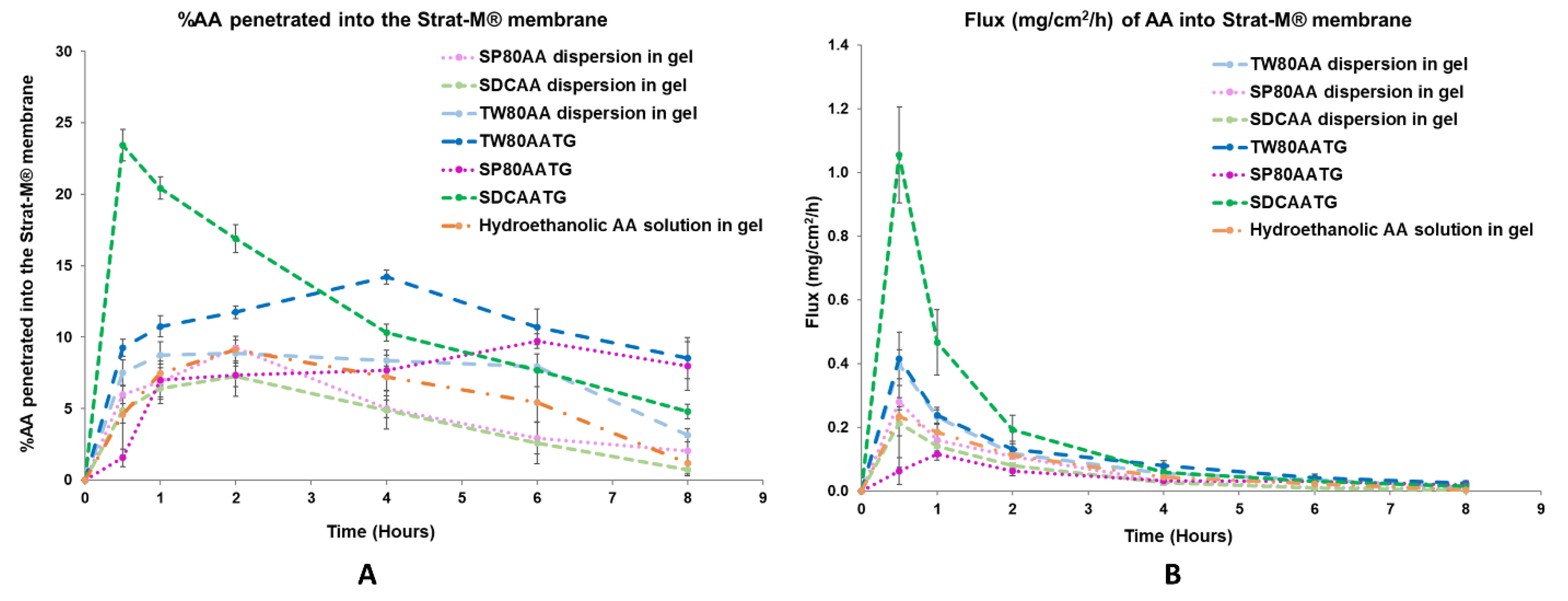
Fig.1 (A) In vitro permeation profile and (B) flux profile of the prepared AATGs and controls. Error bars indicate ± SD with n = 3. 1,2
What This Data Shows
This figure compares the in vitro membrane permeation performance of transfersomal gels prepared with different edge activators. The cumulative drug permeation percentage and steady-state flux vary markedly between formulations—even when vesicle diameters are comparable.
This illustrates a core principle: transfersome development is not simply about minimizing particle size. The edge activator governs membrane interaction and drug partitioning in ways that DLS size metrics alone cannot capture.
Accordingly, edge activator screening, size and PDI control, and Franz diffusion cell validation should be treated as a continuous, integrated pipeline—not isolated development checkpoints.
Transfersome Size Optimization and PDI Control
Size and polydispersity are formulation design levers—not just characterization endpoints. They influence penetration pathway, batch consistency, and interpretability of permeation data.
Why Size Matters Differently for Transfersomes
For rigid nanoparticles, smaller size is a straightforward advantage. Transfersomes operate differently: their access to the stratum corneum is thought to involve deformation-driven permeation, which means that vesicles significantly larger than the pore need sufficient membrane elasticity rather than just small diameter.
That said, size control remains important. Vesicles above a certain diameter range are unlikely to penetrate effectively regardless of deformability, particularly under non-occlusive application conditions. Size also affects drug loading capacity and release kinetics through its influence on the surface-area-to-volume ratio.
A PDI below 0.30 is commonly used as an early development target for relatively uniform vesicle populations. Values in the 0.30–0.40 range may be acceptable during exploratory screening. Higher PDI generally indicates multiple size populations, which complicates both permeation data interpretation and batch reproducibility—and should be addressed before advancing into characterization or regulatory-facing studies.
Process Variables That Govern Size and PDI
Hydration Method and Rate
Thin-film hydration followed by sonication tends to yield smaller initial populations than slow vortex hydration, but the relationship is system-specific.
Extrusion Parameters
Sequential extrusion through polycarbonate membranes (e.g., 400 → 200 → 100 nm) is a common size-reduction approach. Pass number, temperature, and pore sequence each influence the final distribution.
EA Concentration
Increasing EA concentration often reduces vesicle size, but concentrations above a system-specific threshold can increase PDI through partial micellar contamination or vesicle fragmentation.
Hydration Temperature
Hydration above the phase transition temperature (Tm) of the dominant phospholipid improves size uniformity. Hydrating below Tm typically produces more polydisperse populations.
DLS Characterization: What to Report at Each Formulation Iteration
Dynamic light scattering (DLS) remains the primary tool for size and PDI determination. The following parameters should be collected and documented consistently across the development campaign:
Our Lipid-Based Characterization Service provides DLS, zeta potential, encapsulation efficiency, and morphological assessment (TEM/cryo-TEM) in a consolidated package to support complete physicochemical profiling.
Franz Diffusion Cell Testing for Transfersomes: Protocol and Interpretation
The Franz diffusion cell is the standard in vitro method for evaluating topical and transdermal drug permeation. For transfersomes, correct experimental design is essential to generate data that meaningfully differentiates between formulation candidates.
Membrane Selection and Experimental Design
The Franz diffusion cell separates a donor compartment (formulation applied) from a receptor compartment (drug collected over time) by a membrane. Three membrane types are commonly used for transfersome evaluation:
-
▸
Synthetic membranes (cellulose acetate, polysulfone) — Suitable for rapid comparative screening. Do not capture biological skin complexity but allow reproducible, controlled head-to-head comparisons.
-
▸
Ex vivo skin (porcine ear or abdominal) — A widely used physiological model. Ex vivo skin integrity should be verified before each experiment using a predefined laboratory method such as TEWL, electrical resistance, or another validated acceptance criterion, depending on platform and tissue source.
-
▸
Artificial skin equivalents (e.g., Strat-M) — Multilayer membranes designed to mimic human skin diffusion resistance. Offer batch-to-batch consistency, reducing biological variability.
Receptor fluid composition must maintain sink conditions throughout the experiment. Temperature is typically held at 32°C ± 0.5°C to reflect skin surface temperature. All candidates within a screen must be tested under identical membrane conditions to allow valid comparisons.
Sampling intervals spanning the lag phase and steady-state phase (commonly 0.5 through 24 hours) are needed to calculate both cumulative permeation (Qt) and steady-state flux (Jss). Reporting only Q24 without flux and lag time data limits the mechanistic interpretation of results.
Key Permeation Metrics
| Metric | Meaning |
|---|---|
| Qt (µg/cm²) | Cumulative drug permeated per unit area over time t |
| Jss (µg/cm²/h) | Steady-state flux; slope of linear permeation phase |
| Tlag (h) | Lag time before linear permeation begins |
| EE (%) | Fraction of drug retained within vesicles |
| ER | Enhancement ratio vs. plain gel or free drug control |
Common Pitfalls to Avoid
- ✗ Using occlusive conditions with intact transfersomes (suppresses osmotic gradient mechanism)
- ✗ Failing to verify sink conditions throughout sampling
- ✗ Comparing formulations across different membrane models without cross-validation
- ✗ Omitting a plain gel comparator for enhancement ratio calculation
- ✗ Reporting only Q24 without flux and lag time analysis
Looking for ex vivo or in vitro permeation testing support?
Creative Biolabs offers dermal delivery liposome development services including formulation screening and Franz diffusion cell validation using synthetic and ex vivo skin models.
An Integrated Transfersome Development Workflow
Edge activator screening, size control, and permeation testing should be designed as interdependent stages, not sequential checkpoints. Here is how a structured, evidence-based workflow is typically organized.
Lipid System Design
Select phospholipid backbone, define lipid-to-drug ratio, and identify EA candidates for parallel screening
Preparation & Size Reduction
Thin-film hydration, sonication, then sequential extrusion toward target Z-average and PDI
Physicochemical Characterization
Measure Z-avg, PDI, zeta potential, EE%, and DI for each candidate; eliminate outliers before advancing
Franz Cell Permeation Testing
Run in vitro permeation with consistent membrane model; compare Jss, Q24, and Tlag to select lead
Gel Formulation
Incorporate optimized vesicles into a gel matrix; assess rheology and its effect on drug release
Scale-Up & Stability
Evaluate batch size scalability; assess physical and chemical stability across defined storage conditions
Structured Documentation and Pharmaceutical Development Principles
Creative Biolabs provides structured development documentation designed to support formulation understanding, method traceability, and downstream CMC communication. Where appropriate, study designs can be aligned with broader pharmaceutical development principles described in ICH Q8, including characterization of critical quality attributes and the design space. This supports transparency in formulation decision-making and provides a foundation for downstream regulatory discussion.
Related Services from Creative Biolabs
From lipid formulation design through physicochemical characterization and permeation testing, Creative Biolabs supports every stage of the transfersome development program.
Frequently Asked Questions
Commonly investigated edge activators include Span 80 (Sorbitan monooleate), Tween 80 (Polysorbate 80), sodium deoxycholate (SDC), sodium cholate, and sucrose laurate. Each carries a distinct HLB value and membrane interaction profile that influences vesicle deformability, encapsulation efficiency, and in vitro permeation behavior. The optimal edge activator is drug- and system-specific. EA candidates are typically screened across a defined concentration range, and comparative data—including deformability index, PDI, zeta potential, and permeation flux—is used to identify the most suitable option for a given formulation.
A PDI below 0.30 is commonly used as an early development benchmark for relatively uniform transfersome populations. Values in the 0.30–0.40 range may be tolerated during initial exploratory screening, but a PDI above 0.40 typically indicates a heterogeneous population that complicates both permeation data interpretation and batch reproducibility. This should generally be resolved before advancing to formal characterization or regulatory-facing studies. The acceptable window may vary depending on the specific formulation, drug, and analytical platform in use.
The deformability index is measured by extruding a fixed volume of vesicle suspension through a defined membrane (typically a polycarbonate membrane with 50–200 nm pore size) under controlled pressure. The DI is calculated based on the weight of extruded material and the size change observed before and after extrusion—a higher DI reflects greater membrane elasticity. Because DI values depend strongly on test setup, membrane pore size, and calculation method, internal benchmarking within the same experimental system is generally more informative than reference to a single external cutoff. DI should always be interpreted alongside EE%, PDI, and zeta potential, not as an isolated metric.
The most appropriate membrane depends on the stage and purpose of testing. Synthetic cellulose acetate or polysulfone membranes are well-suited for rapid, reproducible head-to-head screening of edge activator candidates. Ex vivo porcine ear or abdominal skin—or commercially available multilayer membranes such as Strat-M—provide more physiologically relevant permeation data and are often preferred when the goal is mechanistic understanding or formulation selection for regulatory purposes. Regardless of model, all candidates within a comparison should be tested under identical conditions, and skin integrity should be verified before each ex vivo experiment using a defined laboratory method.
Yes. Well-documented transfersome development data—covering particle size, PDI, zeta potential, encapsulation efficiency, deformability index, drug release, and in vitro permeation—can form part of the formulation understanding and CMC documentation required for pharmaceutical development. Creative Biolabs provides structured development reports designed to support formulation decision-making, method traceability, and downstream regulatory communication. Where appropriate, study designs can be aligned with pharmaceutical development principles described in ICH Q8, including critical quality attribute identification and design space exploration.
References
- Opatha, Shakthi Apsara Thejani, et al. "Preparation, characterization and permeation study of topical gel loaded with transfersomes containing asiatic acid." Molecules 27.15 (2022): 4865. https://doi.org/10.3390/molecules27154865
- Under Open Access license CC BY 4.0, without modification.
Online Inquiry
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.


