Why Encapsulation Does Not Equal Delivery:
Assessing Payload Compatibility
A payload that loads well into lipid-based formulations is not always a payload that releases, escapes the endosome, or drives functional intracellular activity. Learn how to evaluate delivery competence before over-investing in the wrong formulation path.
The Encapsulation Illusion in Formulation Science
High Encapsulation Efficiency (EE%) is widely regarded as a primary milestone in lipid nanoparticle (LNP) and liposome development. For early-stage formulation scientists, achieving an EE% greater than 90% typically signals a "green light" to proceed to functional testing. However, a pervasive pitfall exists in this paradigm: treating a high encapsulation metric as definitive proof of formulation success.
Encapsulation confirms association with the carrier; it does not confirm productive release, cytosolic access, or biological function.
A payload that can be successfully loaded into a lipid carrier may still fail during subsequent phases of the delivery journey, such as cellular internalization, endosomal escape, and intracellular release. Thus, knowing a candidate cargo is merely "encapsulatable" is insufficient; researchers must determine whether it is truly "deliverable" by evaluating structural readiness and release kinetics.
For Teams Facing the Encapsulation–Delivery Gap
This resource is intended for early-stage formulation scientists, RNA delivery researchers, and biotech project teams developing liposomal or LNP-based payloads.
If a formulation shows strong loading but inconsistent release, weak endosomal escape, or limited functional output, payload compatibility should be assessed before additional optimization or in vivo investment.
Quick Payload Compatibility Checklist
Navigating the Intracellular Bottleneck
Functional delivery requires a lipid-based carrier to execute a complex sequence of biological events. While surviving systemic circulation and reaching target cells are vital, the most critical bottleneck occurs within the endosome. To prevent degradation by lysosomal enzymes, the carrier must facilitate rapid endosomal escape.
For nucleic acids like mRNA or siRNA, this is heavily reliant on the pH-responsive nature of ionizable lipids interacting with the anionic endosomal membrane to induce membrane destabilization. If the internal interactions between the payload and the carrier are overly stabilized—a common byproduct of optimizing solely for encapsulation efficiency—the complex may fail to disassemble.
The key compatibility question is therefore not only whether the payload can be loaded, but whether the lipid-payload complex can disassemble under biologically relevant conditions and support functional release.
High EE% Can Hide Three Delivery Risks
-
1Strong loading, weak release Robust electrostatic forces trap the payload, preventing necessary dissociation from the lipid matrix within the cytoplasm.
-
2Stable particles, poor membrane remodeling Over-optimization for stability can restrict the carrier's ability to undergo the phase transitions required for endosomal fusion.
-
3Good uptake, low functional output High intracellular fluorescence might simply reflect intact carriers trapped and accumulating in lysosomes.
Internal Nanostructures and Structural Competence
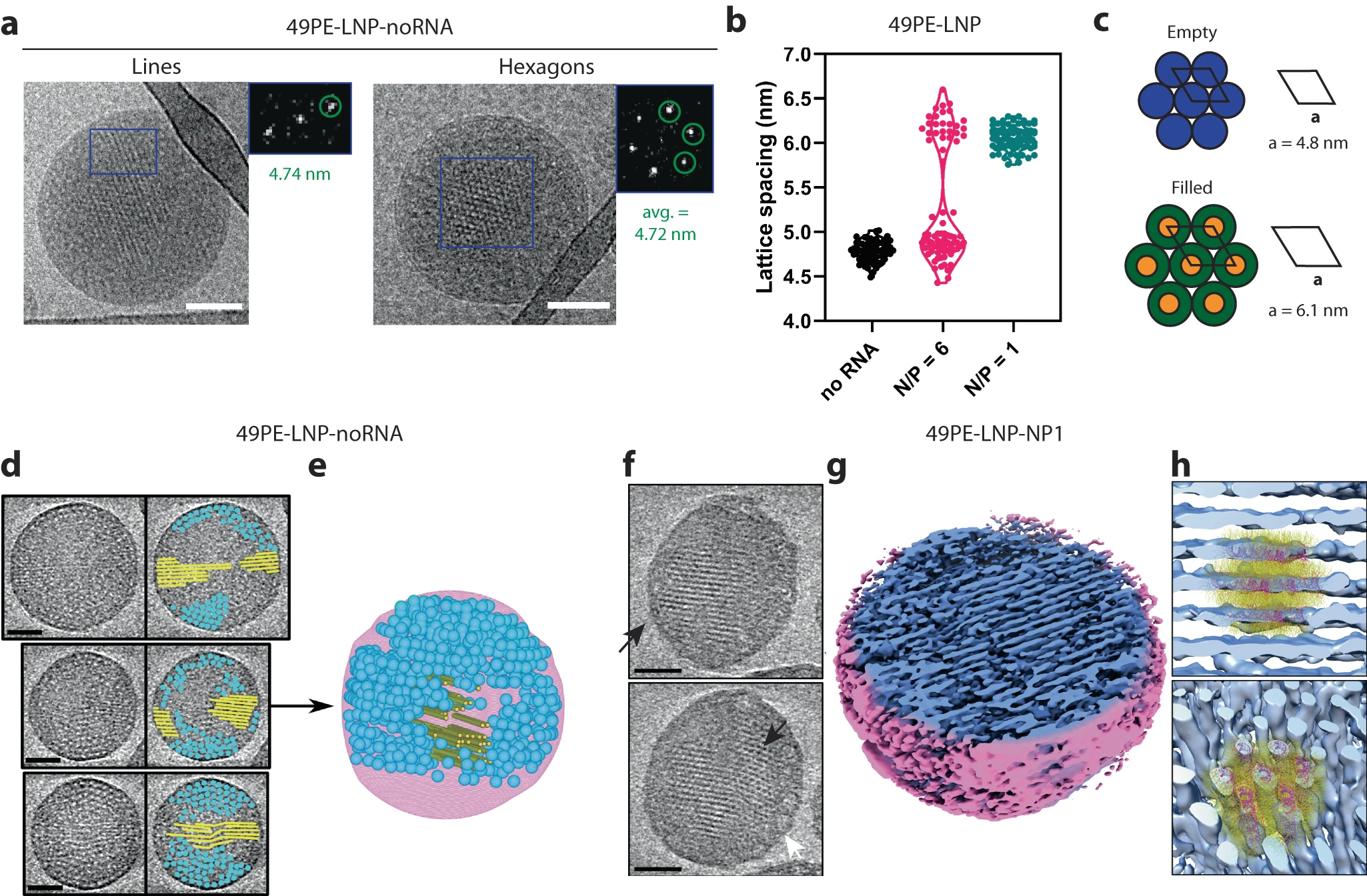
Recent structural studies suggest that macroscopic readouts such as DLS size and PDI are often insufficient to explain why apparently similar formulations perform differently in delivery assays. A pivotal study published in Nature Communications revealed that not all lipid nanoparticles are equally delivery-competent, even when RNA appears to be perfectly encapsulated.
By distinguishing empty from filled inverse hexagonal structures, the study highlights that internal nanostructure and true payload incorporation jointly shape the transfection outcome. Successful encapsulation is only one checkpoint, while intracellular release can also be shaped by internal nanostructure, phase behavior, and lipid-payload organization.
Before advancing to in vivo models, teams should assess whether a given payload-lipid combination forms structurally competent phases associated with productive delivery.
Advanced Analytical Paradigms for Delivery Prediction
Moving beyond superficial metrics requires an upgraded analytical toolkit. Standard sizing and basic dye-exclusion assays must be supplemented with techniques that probe the internal architecture and functional behaviors of the lipid carrier.
| Assessment Goal | Common Early Readout (Limited) | More Informative Readout |
|---|---|---|
| Payload Loading | Bulk Encapsulation Efficiency (EE%) | Internalized vs. surface-associated cargo distinction |
| Carrier Structure | DLS sizing and PDI only | Cryo-EM / SAXS / Internal morphology mapping |
| Release Kinetics | Assessed only at physiological pH 7.4 | Endosome-relevant release and disassembly (pH 5.5) |
| Cellular Delivery | Basic intracellular fluorescence uptake | Endosomal escape tracking + functional reporter expression |
A Practical Diagnostic Workflow
A practical evaluation workflow typically starts with basic physicochemical characterization, then moves to release/disassembly profiling and functional delivery readouts to prevent developmental dead-ends.
Begin with thorough basic characterization and rigorous encapsulation efficiency analysis that challenges the particle to distinguish properly internalized cargo from surface-bound payload.
Subject the particles to endosome-mimicking environments to assess pH-dependent membrane disruption and true payload disassembly kinetics.
Utilize cell-based uptake assays, endosomal escape tracking, and specific functional reporter systems to prove the payload is biologically active post-delivery.
Formulation Strategies for True Payload Delivery
When compatibility is weak, the task is not simply to increase loading further, but to redesign the formulation so that loading, release, membrane interaction, and storage stability remain balanced.
- Tuning Helper Lipid Ratios: Fine-tuning components like DOPE vs. DSPC can favor membrane fusion at low pH. DOPE naturally promotes the inverted hexagonal phase (HII), significantly enhancing fusogenicity upon endosomal acidification.
- Managing the "PEG Dilemma": While PEGylated lipids are vital for stability, they sterically hinder cellular uptake. Incorporating sheddable PEG-lipids that detach in response to the target microenvironment activates the carrier's fusogenic potential precisely when needed.
- Maintaining Storage Fitness: Tracking degradation kinetics through formulation stability monitoring ensures that structural competence observed on day one remains intact.
Through iterative refinement provided by formulation optimization services, researchers can align the lipid composition to perfectly match the physicochemical properties of their specific payload.
Ready to Evaluate Whether Your Payload Is Truly Delivery-Compatible?
High loading alone can create false confidence. Creative Biolabs helps teams assess structural loading, release behavior, and functional delivery performance before committing to an unproductive formulation path.
Frequently Asked Questions
High encapsulation efficiency merely indicates that the payload is successfully entrapped within or tightly associated with the lipid carrier during the manufacturing process. However, for successful in vivo delivery, the carrier must also protect the payload in systemic circulation, facilitate cellular uptake, and crucially, release the payload into the cytoplasm. A formulation that binds its cargo too tightly to achieve near 100% encapsulation may fail to release it inside the cell, rendering it biologically inactive.
Several red flags suggest a formulation is trapped in an "encapsulation illusion":
- High cellular uptake but weak downstream functional reporter signal.
- Strong cargo association but demonstrably poor endosomal escape.
- A stable DLS profile but weak payload release under acidic/endosomal conditions.
- Good bulk EE% but a surprisingly large fraction of surface-associated (rather than internalized) cargo upon enzymatic challenge.
Standard fluorescent assays often measure total associated payload, which includes molecules stuck to the exterior surface of the liposome. To differentiate, advanced assays utilize enzymatic digestion (e.g., RNase or DNase for nucleic acids) or quenching agents that cannot penetrate the intact lipid bilayer. Payload that survives this treatment is verified as fully internalized. Additionally, structural techniques like SAXS or Cryo-EM can visually and mathematically confirm internal loading.
The internal lipid phase dictates how the carrier interacts with cellular membranes. Formulations that naturally adopt or can transition into an inverted hexagonal phase (HII) in acidic environments are highly fusogenic. This structural phase destabilizes the endosomal membrane, facilitating the release of RNA into the cytoplasm. If the internal phase is incorrect or "empty," even a fully intact LNP will fail to release its cargo efficiently.
Payload compatibility should be assessed immediately after initial formulation screening, before committing to extensive in vivo studies. Evaluating phase behaviors, stability in serum, and performing robust in vitro or ex vivo endosomal escape assays early in development prevents researchers from wasting resources on formulations that have high EE% but lack delivery competence.
Creative Biolabs utilizes a multi-tiered approach that moves beyond standard DLS and EE% measurements. We deploy advanced morphological analysis via Cryo-EM, SAXS for internal structure validation, pH-dependent membrane disruption assays to simulate endosomal escape, and highly specific reporter-based in vitro functional assays. This integrated workflow helps determine whether a formulation is optimized not only for loading and stability, but also for productive intracellular release.
Reference
- Pattipeiluhu, Roy, et al. "Liquid crystalline inverted lipid phases encapsulating siRNA enhance lipid nanoparticle mediated transfection." Nature Communications 15.1 (2024): 1303. https://doi.org/10.1038/s41467-024-45666-5
- Under Open Access license CC BY 4.0, without modification.
Online Inquiry
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.


