Custom Cationic Lipid Synthesis Service
Background Our Services Workflow Applications Why Choose Us Related Services FAQs
In the era of precision medicine, lipid-based drug delivery systems have revolutionized the therapeutic landscape, enabling the intracellular delivery of genetic payloads that were once considered undeliverable. At the heart of these breakthroughs lies the chemistry of the delivery vehicle itself. Creative Biolabs introduces our premier cationic lipids synthesis service, designed to overcome the critical bottlenecks of stability, toxicity, and endosomal escape efficiency.
Understanding Cationic Lipids in Gene Therapy
Cationic Liposomes vs. Lipid Nanoparticles (LNPs)
While both systems rely on cationic lipids to bind nucleic acids, they differ fundamentally in structure, assembly, and application. Understanding these distinctions is crucial for vector design.
|
Feature
|
Cationic Liposomes
|
Lipid Nanoparticles (LNPs)
|
|
Microstructure
|
Contains an aqueous core; resembles a traditional cell membrane structure.
|
Lipids and nucleic acids form a dense, electron-dense complex in the core.
|
|
Primary Lipid Type
|
Permanently Charged Cationic Lipids (e.g., DOTAP, DOTMA): Positive charge at all pH levels.
|
Ionizable Cationic Lipids: Neutral in circulation (physiologic pH), positive in acidic endosomes.
|
|
Primary Payload
|
Ideal for larger DNA molecules; common in in vitro transfection.
|
RNA (mRNA, siRNA): The gold standard for COVID-19 vaccines and therapeutic RNA delivery.
|
|
Preparation Method
|
Thin-film hydration, extrusion.
|
Microfluidic mixing, ethanol dilution.
|
|
Key Advantage
|
Simple formulation; easy to prepare in a lab setting for cell culture studies.
|
Long circulation half-life; high encapsulation efficiency; superior endosomal escape in vivo.
|
Structure-Function Relationship
The chemical architecture of a cationic lipid dictates the performance of the delivery system. Our synthesis services focus on optimizing three critical structural domains:
-
Hydrophilic Head Group: For ionizable lipids, the head group defines the pKa (Acid Dissociation Constant). An optimal pKa (typically 6.0–7.0) ensures the lipid is neutral in the bloodstream (reducing toxicity) but becomes protonated in the endosome to trigger release.
-
Linker Region: We incorporate specific chemical bonds (e.g., esters, disulfides) to control biodegradability. Ester-linked lipids can be hydrolyzed by intracellular enzymes, preventing lipid accumulation and reducing long-term toxicity.
-
Hydrophobic Tail: The length and saturation of the tail influence membrane fluidity. Unsaturated tails (e.g., linoleyl chains) enhance fusogenicity, facilitating the disruption of the endosomal membrane during the escape process.
Overcoming the Endosomal Barrier
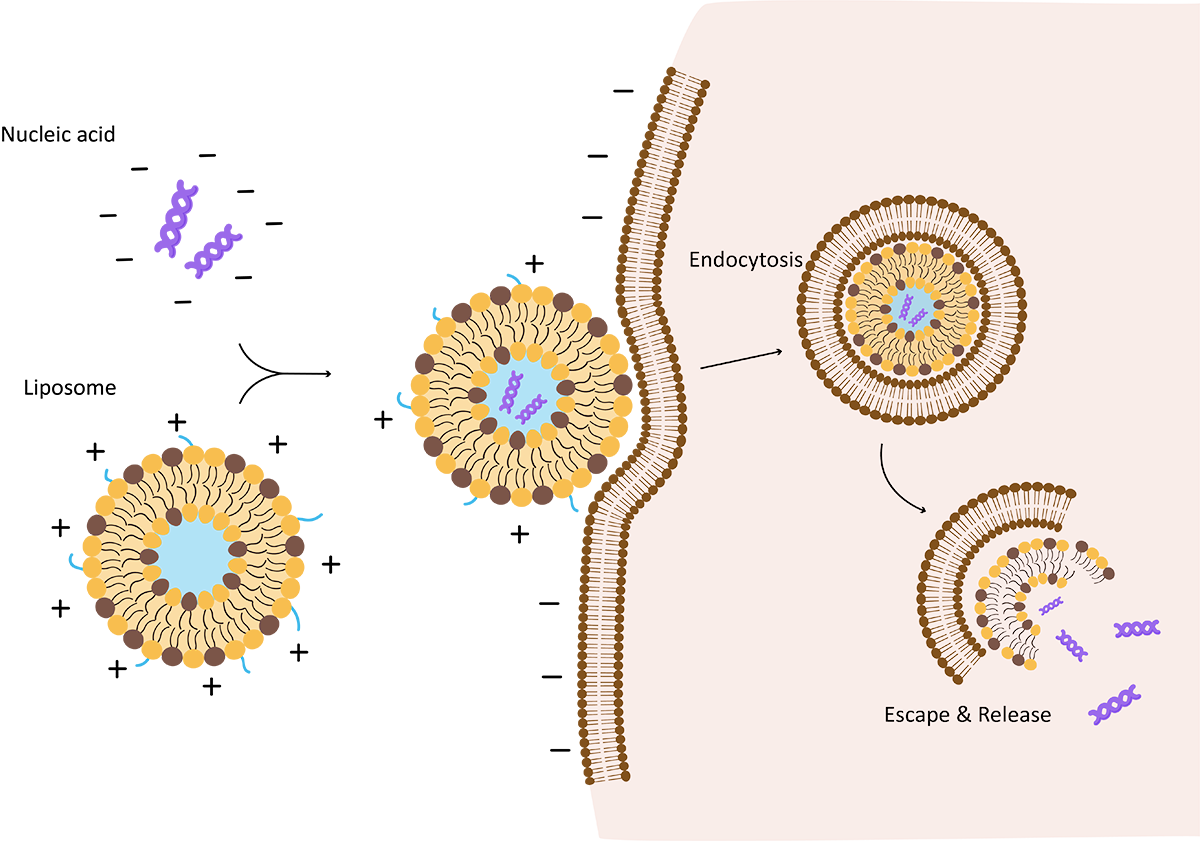 Fig.1 Proposed mechanisms of cationic liposome condensation and uptake
Fig.1 Proposed mechanisms of cationic liposome condensation and uptake
The "Endosomal Escape" paradox is the primary bottleneck in gene delivery. Advanced ionizable lipids utilize the Proton Sponge Effect or membrane fusion mechanisms:
-
Cellular Uptake: The LNP enters the cell via endocytosis.
-
Acidification: As the endosome matures, the internal pH drops (pH ~5.0–6.0).
-
Protonation & Disruption: The ionizable lipids become protonated (positively charged). They interact with the anionic lipids of the endosomal membrane, destabilizing the bilayer structure and releasing the genetic cargo into the cytosol.
Representative Cationic Lipids
In the rigorous pursuit of efficient gene delivery, choosing the right lipid is paramount. The field has transitioned from first-generation lipids, which focused primarily on electrostatic condensation, to sophisticated third-generation ionizable lipids designed for intracellular trafficking. Understanding the distinct physicochemical profiles of these molecules—from the stable ether linkages of DOTMA to the pH-responsive domains of DLin-MC3-DMA—is essential for rational experimental design.
|
Lipid Name
|
Primary Research Utility
|
Scientific Rationale & Mechanism
|
|
DOTAP
|
In vitro transfection (DNA/RNA); Vaccine adjuvants.
|
Biodegradable ester linkage facilitates metabolic clearance. Constitutive positive charge ensures robust DNA condensation but limits systemic tolerability.
|
|
DOTMA
|
In vitro gene delivery; Liposomal stability studies.
|
Features an ether linkage conferring high chemical stability against hydrolysis. Forms rigid liposomes; non-biodegradable nature restricts in vivo application.
|
|
DC-Chol
|
Clinical gene therapy trials; Non-viral vectors.
|
Cholesterol backbone mimics bio-membrane components, enhancing biocompatibility. Cleavable ester bond reduces cytotoxicity compared to aliphatic analogs.
|
|
DLin-MC3-DMA
|
Hepatic siRNA delivery
|
Optimized pKa (~6.44) enables endosomal escape via the proton sponge effect while maintaining neutral circulation pharmacokinetics.
|
|
SM-102
|
mRNA vaccines; Systemic RNA delivery.
|
Tertiary amine allows pH-dependent ionization. Branched tail structure enhances membrane fusogenicity; ester linkages promote rapid biodegradation.
|
|
DODAP
|
Helper lipid in pH-sensitive liposomes.
|
Unsaturated ionizable lipid. Reduces surface charge at physiological pH to extend circulation; facilitates hexagonal phase transition at acidic pH.
|
Comprehensive Custom Synthesis Solutions
Creative Biolabs offers a comprehensive synthesis platform designed to support researchers from early-stage discovery through to clinical development. We do not just synthesize lipids; we optimize them for your specific therapeutic targets to convince you of our capabilities and professionalism.
-
Rational Lipid Design & Library Synthesis: We assist in the design of novel lipid libraries. By systematically varying key structural domains—such as tuning charge density in the head group or adjusting linker biodegradability—we help you identify the lead candidate with optimal pKa and fusogenicity.
-
Custom Synthesis of Specialized Lipids: Whether you need a known reference standard (e.g., DLin-MC3-DMA analogs) or a proprietary novel molecule, our synthetic chemistry team delivers with speed and precision.
-
Process Development & Scale-Up: We transition seamlessly from milligram-scale research batches to kilogram-scale production, designing synthetic routes that are safe, cost-effective, and robust for scale-up.
Workflow
Applications of Cationic Lipid in Modern Research
Our high-purity lipids are currently empowering research across the most dynamic fields of biotechnology:
-
Liposome & LNP Delivery System Development: Accelerating the formulation and optimization of novel lipid carriers to ensure structural stability, high encapsulation efficiency, and enhanced bioavailability for diverse therapeutic payloads.
-
mRNA Vaccine Development: Utilizing high-efficiency ionizable lipids crucial for the stable encapsulation and cytosolic delivery of mRNA antigens for infectious diseases and cancer vaccines.
-
CRISPR/Cas9 Gene Editing: Enabling the co-delivery of Cas9 mRNA and sgRNA to specific tissues by optimizing lipids for organ-specific accumulation.
-
Gene Silencing (RNAi): Providing potent lipids for siRNA/miRNA therapies that require silencing of disease-causing genes in the liver and extra-hepatic tissues.
-
Ex Vivo Cell Therapy: Supporting CAR-T and stem cell engineering with reagents designed for high transfection rates and high cell viability.
Why Choose Creative Biolabs?
-
Unrivaled Purity (>98%): We understand that impurities can trigger immune responses. Our lipids undergo rigorous purification (HPLC, prep-chromatography) to ensure the highest quality.
-
Expertise in LNP Formulation: Unlike standard chemical vendors, we understand the biology. Our team provides insights into how synthesis choices impact LNP formulation and stability.
-
Rapid Turnaround: We offer some of the fastest synthesis timelines in the industry, accelerating your "Design-Make-Test" cycles.
-
Analytical Excellence: Every batch comes with a comprehensive Certificate of Analysis (CoA) including MS, and HPLC analysis.
-
Intellectual Property Protection: We adhere to strict confidentiality agreements to protect your proprietary structures and novel lipid designs.
Creative Biolabs is your premier partner for overcoming the chemical and biological challenges of gene delivery. From the precision synthesis of novel cationic lipids to the scale-up of proven standards, we provide the essential building blocks for the next generation of genetic medicines. Our commitment to quality, speed, and scientific collaboration ensures your research moves forward without delay.
Related Services
FAQs
What is the typical purity level of your synthesized cationic lipids?
We standardly deliver lipids with >95% purity, but we frequently achieve >98% or >99% purity for demanding applications, verified by HPLC and MS.
Can you synthesize lipids that are not in your catalog (custom structures)?
Yes, custom synthesis is our specialty. We can synthesize novel lipids based on your specific structural designs or chemical drawings under strict confidentiality.
What scale can you support?
We are flexible and can support projects ranging from a few milligrams for initial screening to hundreds of grams or kilograms for development and production.
Can you provide advice on the best lipid for my specific RNA payload?
Absolutely. Our team includes formulation scientists who can advise on lipid selection based on your payload type (mRNA vs. siRNA) and target tissue.

For Research Use Only. Not For Clinical Use
 Fig.1 Proposed mechanisms of cationic liposome condensation and uptake
Fig.1 Proposed mechanisms of cationic liposome condensation and uptake
 For Research Use Only. Not For Clinical Use
For Research Use Only. Not For Clinical Use