Gradient Loading and Formulation Design
for High-Performance Small Molecule Liposomes
A practical guide to selecting transmembrane gradients and membrane compositions for higher encapsulation efficiency, stronger payload retention, and more stable small molecule liposome formulations.
Overcoming Limitations in Small Molecule Encapsulation
For formulation scientists and CMC teams, encapsulating small molecule therapeutics presents a unique set of challenges that differ fundamentally from the entrapment of large macromolecules like mRNA or proteins. Formulators driving IND-enabling studies frequently encounter critical bottlenecks: capture-volume limited encapsulation, premature payload leakage in serum environments, and the complex balancing of variables—such as lipid ratios, internal salt systems, and loading temperatures—to achieve batch-to-batch reproducibility during scale-up.
For many hydrophilic or amphipathic small molecules, passive loading is capture-volume limited and often yields low encapsulation efficiency. Furthermore, small amphipathic molecules possess high membrane permeability. Even if initially captured, these molecules can rapidly equilibrate across the lipid bilayer, diffusing out of the liposome during extended storage or immediately upon intravenous injection.
To translate a promising active pharmaceutical ingredient (API) into a viable clinical product, researchers must transition from passive entrapment to advanced remote loading technologies. By leveraging active gradient systems, it is possible to transform the liposome from a simple passive container into a dynamic, chemical "trap" that actively draws and sequesters the drug. The central development question is therefore not simply whether a small molecule can be encapsulated, but which gradient chemistry and membrane architecture can convert transient loading into durable retention. Partnering with experts through a specialized Liposome Encapsulated Small Molecule Drugs Development Service can significantly accelerate this complex transition, ensuring that the selected loading mechanism aligns perfectly with the physicochemical profile of the API.
Mechanisms of Active Remote Loading
Remote loading, also known as active loading, relies on generating a transmembrane chemical gradient. This gradient acts as the thermodynamic driving force to pump amphipathic weak acids or bases from the external aqueous medium into the aqueous core of preformed liposomes.
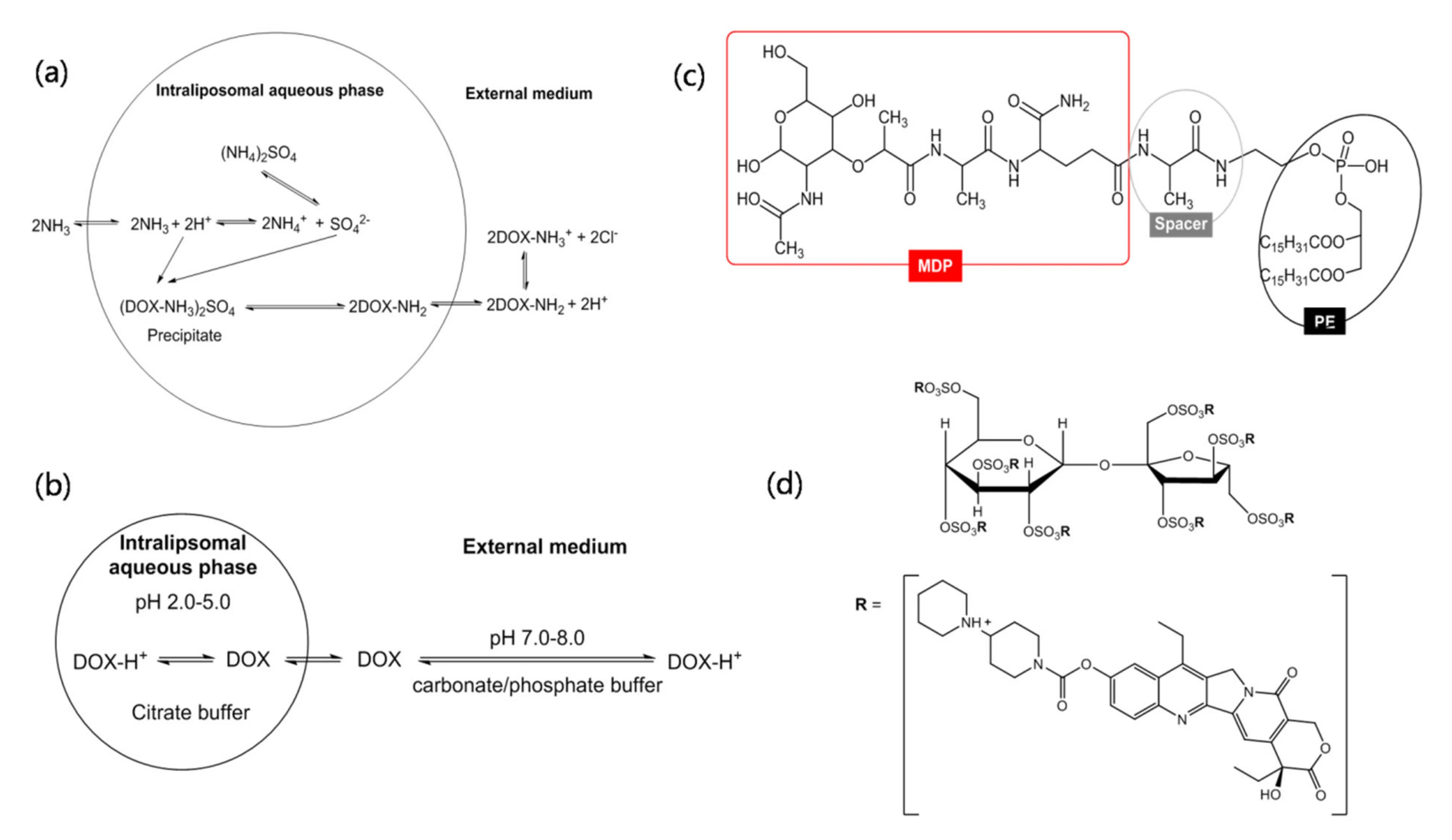
The fundamental principles underlying these techniques are critical for optimizing nanomedicines. Different remote loading mechanisms are used in clinically relevant liposomal small-molecule formulations. As structurally represented, transmembrane ion or pH gradients drive neutral/permeable drug species across the lipid bilayer. Once inside the liposome's aqueous core, the microenvironment triggers intraliposomal protonation, complexation, gelation, or precipitation. This phase-change or ionization event traps the molecule inside, effectively converting a highly permeable neutral species into a membrane-impermeable ionized or crystalline state.
This sophisticated physicochemical manipulation can enable very high loading in suitable drug/gradient pairs—and crucially helps retain the payload during in vivo circulation until the liposome reaches the target tissue. To confirm the success of these gradients, rigorous analytical validation is required. Utilizing a robust Liposome Encapsulation Efficiency Analysis ensures that the intra-vesicular precipitation is uniform and the unencapsulated drug fraction is thoroughly quantified and removed.
| Gradient Type | Core Mechanism | Ideal Drug Profile | Clinical / Industry Example |
|---|---|---|---|
| Ammonium Sulfate Gradient | Exchange of uncharged drug with ammonia. Intraliposomal precipitation as drug-sulfate complex. | Amphipathic weak bases (pKa < 11). | Doxorubicin liposomes |
| Transmembrane pH Gradient (Citrate) | Acidic core (pH ~4.0) vs neutral exterior (pH ~7.4). Drug protonates and is trapped. | Weakly basic drugs requiring mildly acidic stabilization. | Vincristine liposomes, Daunorubicin |
| Calcium Acetate Gradient | Acetate diffuses out carrying protons; interior pH rises. Drug forms complex with intra-vesicular Calcium. | Amphipathic weak acids. | Topotecan, Glucocorticoids |
| Transition Metal Ion Gradients (Cu2+, Mn2+) | Drug coordinates with transition metals inside the core, often accompanied by a secondary ionophore. | Molecules capable of forming stable coordination complexes. | Representative investigational systems |
Synergizing Formulations with Drug Properties
The choice of gradient chemistry cannot be made in isolation; it must be precisely matched to the drug’s ionization behavior, lipophilicity (LogP), and desired release profile. An optimal formulation requires a holistic approach that simultaneously addresses the internal aqueous phase chemistry and the external lipid membrane composition.
Lipid bilayer dynamics play a critical role in controlling the rate of drug loading and the subsequent rate of release in vivo. Formulators must carefully select bulk lipids (e.g., HSPC, DSPC, DMPC) with appropriate phase transition temperatures (Tm). Loading is typically performed at temperatures above the Tm, where the membrane is in a fluid liquid-crystalline phase, allowing rapid drug diffusion. Upon cooling below the Tm, the membrane solidifies into a rigid gel phase, locking the payload inside.
Cholesterol is universally added to these formulations. It eliminates the phase transition peak, tightens the packing of phospholipid acyl chains, and significantly reduces the membrane's permeability to both the entrapped drug and the gradient-forming ions. Balancing these components requires extensive screening, often facilitated by expert Liposome Formulation Optimization services, to ensure that the liposome remains intact in the harsh environment of the bloodstream while releasing its payload appropriately at the target site.
Key Physicochemical Considerations
-
1Drug pKa, Ionization State, and Membrane-Permeable Fraction Dictates the required pH of the external medium during loading to ensure a sufficient uncharged fraction is available to permeate the membrane.
-
2Lipid Phase Transition (Tm) Loading temperature must be calibrated above the Tm of the primary phospholipid to facilitate rapid transmembrane flux.
-
3Intra-vesicular Counter-Ion Selecting sulfate, citrate, or transition metals determines the solubility and crystalline state of the drug inside the core, directly impacting the release kinetics.
How to Match Gradient Strategy to API Properties
A practical decision framework for formulation scientists:
- Is the API a weak base, weak acid, or neutral compound? This fundamental property determines if a gradient can be applied.
- What is the pKa range? Ensures the active species can exist in a membrane-permeable state at physiological or tunable external pH.
- Does it form an insoluble salt/complex in the core? Crystallization within the liposome dramatically limits osmotic swelling and leakage.
- Is the desired product profile fast release, sustained retention, or long-circulating stability? Influences the choice of bulk lipid saturation and counter-ion selection.
Maximizing Payload Retention and Shelf-Life Stability
Achieving high initial encapsulation is merely the first milestone. The ultimate success of a liposomal small-molecule therapeutic depends on its stability over time. Premature leakage destroys the therapeutic index, increasing off-target toxicity and reducing efficacy at the disease site.
Morphological Stability
Intraliposomal crystallization (e.g., Doxorubicin-sulfate forming 'coffee-bean' like crystals) drastically limits osmotic swelling and membrane rupture. Controlling the crystal lattice structure is paramount for extending shelf-life.
In Vivo Steric Shielding
Incorporation of PEGylated lipids (e.g., DSPE-mPEG2000) prevents plasma protein opsonization and RES clearance. This shielding ensures the liposome can prolong circulation and improve the opportunity for site exposure.
Thermodynamic Analysis
Continuous assessment of leakage kinetics under various physiological conditions (pH, temperature, serum presence) is required. Accelerated stress testing predicts long-term ex vivo storage capabilities.
To guarantee that a rationally designed formulation maintains its integrity from manufacturing to bedside, comprehensive longitudinal studies are vital. Employing a dedicated Formulation Stability Monitoring Service provides the precise analytical tracking necessary to detect trace leakage, lipid degradation, and aggregate formation, ensuring regulatory compliance and clinical safety.
Frequently Asked Questions
Passive loading relies on the drug being dissolved in the aqueous buffer during liposome formation. Because liposomes have a very small internal captured volume (typically 1-5 µL/µmol lipid), they physically encapsulate a tiny fraction of the surrounding fluid. Consequently, the maximum encapsulation efficiency for passive loading rarely exceeds 10%. Furthermore, highly permeable small molecules will rapidly diffuse back out until concentration equilibrium is reached. Active gradient loading circumvents this by using a chemical driving force to concentrate the drug inside the liposome up to 1000-fold higher than the external medium.
The most successful candidates are amphipathic weak bases or weak acids. They need to possess an appropriate LogP to permeate the lipid bilayer in their un-ionized state, yet have a pKa that allows them to become highly ionized (and thus membrane-impermeable) once they encounter the altered pH or ion environment inside the liposome core. Molecules that can form low-solubility salts or complexes with the chosen trapping agent (like sulfate or transition metals) will yield the best retention profiles.
While remote loading requires the drug to be solubilized in the external buffer, many potent APIs are highly hydrophobic and poorly water-soluble. In these cases, formulation scientists may employ solvent-assisted techniques, adding carefully controlled amounts of water-miscible solvents (e.g., ethanol or DMSO) to the external loading buffer or utilizing cyclodextrin complexes to keep the drug transiently dissolved long enough to permeate the bilayer and interact with the internal gradient.
The choice heavily depends on the target drug's pKa and its tendency to precipitate. Ammonium sulfate gradients are generally preferred for weak bases because the combination of internal acidification (as ammonia escapes) and the presence of sulfate ions drives the drug to form highly stable, insoluble gel-like crystals inside the liposome. A simple pH gradient (e.g., utilizing a citrate buffer) is utilized when the drug is susceptible to degradation by sulfate or when an intermediate release rate is required, relying purely on protonation rather than crystallization to hold the drug inside.
Absolutely. The loading process must be conducted at a temperature above the Tm of the main lipid component. At temperatures above the Tm, the lipid bilayer is in a fluid, liquid-crystalline state, making it highly permeable to the uncharged form of the drug. Once loading is complete, the liposomes are rapidly cooled below the Tm. The membrane transitions into a rigid gel state, drastically reducing permeability and securely locking the ionized/crystallized payload inside the core.
Validation requires quantifying both the internal structure and the loading metrics. High-Performance Liquid Chromatography (HPLC) coupled with ultrafiltration or size-exclusion chromatography (SEC) is used to separate and quantify the free versus encapsulated drug, determining the precise EE%. Dynamic Light Scattering (DLS) ensures that loading hasn't caused aggregation. Most importantly, Cryo-Transmission Electron Microscopy (Cryo-TEM) is frequently utilized to visually confirm the presence of intra-vesicular drug crystals, which is the hallmark of a successful active loading process.
Online Inquiry
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.


