High-Ethanol Ethosomes: Balancing Drug Loading, Stability, and Skin Irritation in Transdermal Delivery
A formulation scientist's guide to optimizing ethanol concentration, co-solvent selection, and phospholipid composition for effective transdermal ethosome systems.
What Are Ethosomes and Why Does Ethanol Matter?
Ethosomes are phospholipid-based vesicular carriers that incorporate a relatively high concentration of ethanol (typically 20–45% v/v) as an integral structural component — not merely a co-solvent. First described by Touitou and colleagues, ethosomes are distinguished from conventional liposomes by their ability to deeply penetrate the stratum corneum, making them a powerful platform for transdermal and topical drug delivery.
The ethanol in ethosome formulations serves multiple roles: it fluidizes the lipid bilayer by interacting with phospholipid head groups, enhances membrane flexibility, and transiently disrupts the highly ordered lipid structure of the skin. These combined effects allow ethosomes to carry both hydrophilic and lipophilic drugs deep into the viable epidermis and dermis — a feat that conventional liposomes rarely achieve.
However, the same ethanol concentration that enables enhanced skin penetration also raises three critical formulation challenges that every developer must confront: (1) how to maximize drug encapsulation efficiency without destabilizing the vesicle, (2) how to maintain physical and chemical stability over a product shelf-life, and (3) how to prevent skin irritation at the application site. Resolving these tensions is the central problem in ethosome development.
Our Ethosome Development Service is specifically designed to help researchers and pharmaceutical teams navigate these formulation trade-offs using systematic screening and analytical characterization.
Who Faces This Formulation Challenge?
Ethosome development bottlenecks are commonly encountered by:
- ✓ Pharmaceutical formulation scientists developing novel transdermal APIs who need to balance high EE with acceptable vesicle integrity.
- ✓ Dermatology researchers working on skin diseases (psoriasis, alopecia, fungal infections) where deep follicular penetration is required.
- ✓ CMC teams in biotech companies who require stability data for IND filings and need rapid formulation screening before scaling up.
- ✓ Academic groups studying in vitro and ex vivo skin permeation who require stable, reproducible ethosome batches for comparative studies.
How Ethanol Drives Skin Penetration — and Why It Also Creates Problems
Understanding the mechanistic roles of ethanol helps explain why optimizing its concentration is a multi-objective problem, not simply a matter of using more solvent.
Membrane Fluidization
Ethanol intercalates between phospholipid acyl chains, reducing the gel-to-liquid phase transition temperature and increasing bilayer fluidity. This fluidization allows vesicles to deform under pressure — a key property enabling permeation through tight intercellular spaces in the stratum corneum.
Skin Lipid Disruption
Upon topical application, ethanol rapidly partitions into the stratum corneum and transiently disrupts the lamellar lipid domains between corneocytes. This creates hydrophilic pathways that the intact ethosome vesicle can exploit. The perturbation is reversible at moderate concentrations, but at very high ethanol levels, prolonged disruption becomes a source of barrier damage and irritation.
Drug Solubilization & Partitioning
Many APIs with limited aqueous solubility require ethanol for adequate dissolution during vesicle formation. Higher ethanol concentrations improve drug solubility in the aqueous phase and enhance partitioning into the bilayer. Yet beyond a threshold concentration, excess ethanol destabilizes the vesicular structure entirely, leading to increased particle size, polydispersity, or even disassembly of the bilayer architecture.
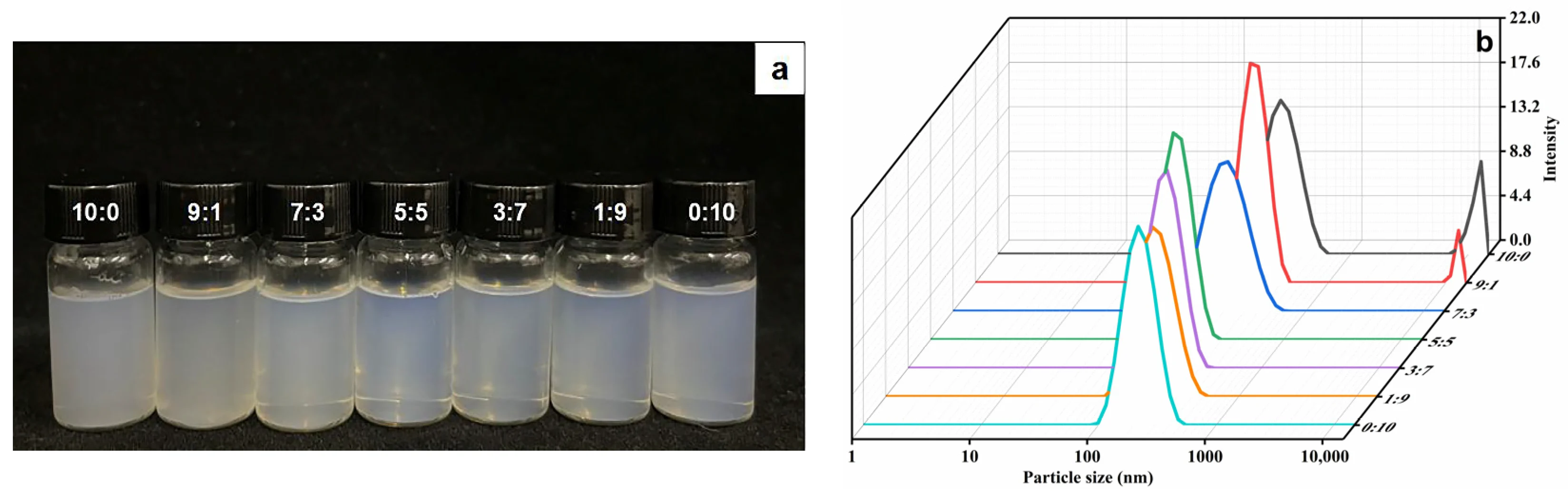
Fig.1 (a) Appearance pictures and (b) variation trend of particle size with EtOH/PG mass ratios of 10:0, 9:1, 7:3, 5:5, 3:7, 1:9 and 0:10. 1,2
Evidence from Binary Ethosome Research
A pivotal insight from recent literature on binary ethosomes — systems incorporating both ethanol (EtOH) and propylene glycol (PG) as co-solvents — is that simply increasing the ethanol fraction does not guarantee superior formulation performance. A study evaluating phloretin-loaded propylene glycol binary ethosomes demonstrated that as the EtOH/PG mass ratio was systematically varied (from 10:0 through intermediate ratios to 0:10), both vesicle appearance and particle size distribution changed substantially.1
Formulations at the ethanol-only extreme (10:0) more frequently exhibited bimodal particle size distributions and signs of aggregation — hallmarks of physical instability. Introducing PG as a co-solvent progressively reduced mean particle size and narrowed the distribution, while also improving encapsulation efficiency (EE) and overall system stability. These findings reinforce a key principle for ethosome developers: the optimization target should not be "maximum ethanol" but rather an ethanol–co-solvent–phospholipid balance that simultaneously satisfies drug loading, particle integrity, and stability criteria.
Optimizing Ethosome Formulation: The Three-Way Trade-Off
Achieving a viable transdermal ethosome product requires simultaneous optimization across drug loading, physical stability, and skin tolerability — three parameters that are mechanistically coupled.
Drug Loading Efficiency in High-Ethanol Vesicles
Encapsulation efficiency (EE) in ethosomes is governed by the interplay between drug lipophilicity, the phospholipid bilayer thickness, and the ethanol-induced changes in membrane permeability. For lipophilic drugs, higher ethanol concentrations initially increase EE by expanding the hydrophobic domain and improving drug–lipid affinity. However, past an optimal threshold, excess ethanol increases membrane permeability to the point where drug leakage during or after preparation offsets any gains.
For moderately hydrophilic drugs, ethanol can facilitate entrapment in the aqueous core by enhancing drug solubility in the vesicle interior. Co-solvents such as PG further stabilize the aqueous compartment, reduce osmotic stress across the membrane, and in some reported systems, produce synergistic increases in both EE and permeation flux compared to ethanol-only formulations.
Our Lipid-Based Basic Characterization Service provides comprehensive EE determination, particle size analysis (DLS), zeta potential measurement, and morphological assessment to benchmark your ethosome formulation before proceeding to permeation studies.
Key Formulation Variables and Their Effects
| Variable | Effect on EE | Effect on Stability | Effect on Skin Tolerability |
|---|---|---|---|
| EtOH % increase | Increases then decreases | Decreases at high % | Worsens above threshold |
| PG addition | Improves in binary systems | Improves (reduces aggregation) | Generally improves |
| Phospholipid type/concentration | Higher SPC concentration increases EE | Increases bilayer rigidity | Generally neutral/positive |
| Cholesterol addition | Variable (drug-dependent) | Increases membrane rigidity | Neutral |
| pH adjustment | Affects ionizable drugs | Impacts zeta potential | Relevant for skin pH match |
Service Spotlight: End-to-End Ethosome Development
Creative Biolabs offers a systematic ethosome development workflow covering formulation screening, optimization, and analytical verification:
Physical and Chemical Stability Challenges in Ethosome Systems
Long-term stability remains one of the most frequently underestimated challenges in ethosome formulation development. Ethanol, while essential for vesicle formation and skin penetration, also accelerates several common degradation pathways. High ethanol content increases the risk of phospholipid hydrolysis (especially under acidic or alkaline conditions), promotes vesicle aggregation or fusion upon storage, and can facilitate drug leakage over time by maintaining bilayer fluidity even at refrigerated temperatures.
Physical stability is typically monitored through changes in particle size (hydrodynamic diameter measured by DLS), polydispersity index (PDI), and zeta potential. A shift toward larger particle sizes or broadening of the size distribution over time indicates the onset of aggregation or fusion. Zeta potential values beyond ±30 mV are generally considered indicative of adequate electrostatic repulsion for colloidal stability.
Binary ethosome systems incorporating PG alongside ethanol have demonstrated superior stability profiles in several in vitro stability studies. The proposed mechanism involves PG occupying space within the hydration layer surrounding the vesicle, reducing the rate of intermolecular collisions and providing a steric barrier against aggregation. The result, as documented in phloretin binary ethosome research1, is a simultaneous improvement in both EE and colloidal stability — a genuinely synergistic outcome.
To support IND submissions and internal quality documentation, our Formulation Stability Monitoring Service conducts ICH-aligned real-time and accelerated stability studies on ethosome and other lipid-based formulations.
Critical Stability Parameters to Monitor
| Parameter | Analytical Method | Acceptable Range |
|---|---|---|
| Particle size (Z-avg) | DLS | <200 nm |
| PDI | DLS | <0.3 |
| Zeta potential | Laser Doppler | |ZP| > 30 mV |
| Encapsulation efficiency | HPLC / UV-Vis | >70% (target-dependent) |
| Phospholipid degradation | HPLC-MS or TLC | <5% degradation |
Formulation Tip: Storage Conditions
Ethosome dispersions are typically most stable at 4°C in sealed, nitrogen-blanketed amber glass vials. Refrigeration reduces phospholipid oxidation and minimizes Ostwald ripening, while light exclusion prevents photodegradation of both the carrier lipids and any light-sensitive APIs. Accelerated stability at 40°C/75% RH allows rapid prediction of shelf-life under ICH Q1A(R2) conditions.
Managing Skin Irritation: Ethanol Concentration and Barrier Integrity
Skin tolerability is a non-negotiable requirement for any topical or transdermal formulation intended for repeated use. In ethosomes, this requirement directly conflicts with the formulation's need for sufficient ethanol to achieve its penetration-enhancing function.
Mechanisms of Ethanol-Induced Irritation
At concentrations exceeding approximately 40–50% v/v under prolonged contact, ethanol can cause protein denaturation in keratinocytes, extract intercellular lipids from the stratum corneum, and impair barrier recovery. These effects manifest clinically as erythema, dryness, and sensory discomfort — unacceptable for products requiring daily or twice-daily application.
In ethosome gel formulations, the vehicle (gel base) dilutes the overall ethanol concentration at the skin surface, but the vesicle interior still carries a high ethanol fraction. The kinetics of ethanol release from the vesicle into the stratum corneum therefore determine the effective irritation potential more than the bulk formulation concentration alone.
Ex vivo skin irritation models — using human cadaveric skin or reconstructed human epidermis (RHE) — provide the most predictive safety data before moving to in vivo patch testing. Endpoints include trans-epidermal water loss (TEWL) recovery, interleukin-1α (IL-1α) release, and visual assessment of skin surface morphology.
Our Formulation Safety Evaluation Service includes skin irritation screening using standardized in vitro and ex vivo assays to support regulatory submissions and guide iterative formulation refinement.
Strategies to Reduce Irritation Without Sacrificing Penetration
Replace Partial Ethanol with PG or Transcutol
Propylene glycol and diethylene glycol monoethyl ether (Transcutol P) are GRAS-listed skin penetration enhancers with superior tolerability profiles. Binary and ternary ethosome systems substituting a fraction of ethanol with these agents maintain comparable permeation flux while reducing primary skin irritation scores in standardized assays.
Optimize Phospholipid Concentration
Higher phospholipid content (particularly HSPC or soy phosphatidylcholine) can help buffer the irritant potential of ethanol by increasing the vesicle bilayer's ability to sequester the solvent within the membrane, reducing free ethanol available to interact with keratinocytes.
Incorporate Anti-Inflammatory Excipients
Skin-calming additives such as allantoin, panthenol, or ceramide-mimicking lipids can be co-incorporated into ethosome formulations to counteract ethanol-driven barrier disruption, improving the overall tolerability profile without altering vesicle architecture.
Frequently Asked Questions
The generally accepted ethanol concentration range for ethosome systems is 20–45% v/v. Below 20%, the bilayer does not achieve sufficient fluidization to confer the deformability advantage over conventional liposomes. Above 45%, the risk of vesicle disruption, excessive membrane permeability, and skin irritation increases sharply. However, the optimal concentration for any specific API depends on the drug's physicochemical properties, target skin depth, and the choice of co-solvents. In binary ethosome systems combining ethanol with propylene glycol, effective formulations have been achieved at even lower absolute ethanol levels — demonstrating that the co-solvent ratio matters as much as the total alcohol content.
Both ethosomes and transfersomes are deformable vesicular carriers for transdermal delivery, but their mechanisms differ meaningfully. Transfersomes penetrate primarily through an osmotic gradient driven by an occlusion-free application: the water activity difference between the skin surface and viable dermis propels the vesicle through skin pores and intercellular channels. Ethosomes, by contrast, rely on ethanol-mediated dual action — simultaneous membrane fluidization and direct disruption of stratum corneum lipid domains — making them less dependent on the osmotic gradient. This distinction means that ethosomes can be formulated in semi-occlusive gels without sacrificing performance, whereas transfersomes require open application conditions. For researchers with specific delivery targets, the choice between the two platforms should be guided by the drug's properties and the intended application format.
Ethosomes have been explored as carriers for small peptides and certain protein antigens in vaccine applications, where transcutaneous immunization is the goal. For small peptides (typically below 1.5 kDa), in vitro and ex vivo studies have demonstrated measurable permeation enhancement compared to aqueous solution. For larger macromolecules including intact proteins or oligonucleotides, the stratum corneum remains a significant barrier even with enhanced ethosome formulations. In such cases, combining ethosomes with physical enhancement methods (microneedles, iontophoresis) or considering alternative lipid nanoparticle systems may be necessary. Creative Biolabs' formulation team can conduct feasibility assessments to guide the most appropriate delivery platform selection for macromolecular cargo.
Prior to clinical use, ethosome formulations should undergo both real-time and accelerated stability testing in accordance with ICH Q1A(R2) guidelines. Core analytical endpoints include: particle size and PDI by DLS (monthly for 3–6 months at 4°C and 25°C; bi-weekly at 40°C/75% RH), zeta potential, encapsulation efficiency by validated HPLC or UV-Vis method, visual appearance (color, clarity, phase separation), and pH. Chemical stability of the drug substance within the vesicle should also be assessed, particularly for drugs prone to hydrolysis or oxidation. Phospholipid degradation products (lysophospholipids) should be quantified using HPLC-MS at defined intervals. Freeze-thaw cycling studies (3–5 cycles) are additionally recommended if the product is intended to be stored frozen.
Yes. Creative Biolabs provides fully customized ethosome development services tailored to your specific API's physicochemical profile, target indication, and regulatory pathway. Our approach begins with a feasibility assessment covering drug-lipid compatibility and solubility in ethanol/co-solvent systems, followed by systematic Design of Experiment (DoE)-driven optimization of ethanol content, phospholipid type and concentration, and co-solvent ratios. We then provide full analytical characterization, in vitro permeation testing (ex vivo Franz cell assays using excised skin), and initial safety screening. All work is conducted under ISO 9001:2015-certified quality systems.
References
- Zhang, Meng, et al. "Designed fabrication of phloretin-loaded propylene glycol binary ethosomes: Stability, skin permeability and antioxidant activity." Molecules 29.1 (2023): 66. https://doi.org/10.3390/molecules29010066
- Under Open Access license CC BY 4.0, without modification.
Online Inquiry
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.


