Enzyme-Loaded Liposomes:
How to Verify Activity Retention, Protection, and Batch Consistency
Practical analytical strategies to assess post-encapsulation enzyme activity, confirm protection during processing and storage, and generate reproducible batch comparison data for research and scale-up programs.
Why Verification Is Critical for Enzyme-Loaded Liposomes
Enzyme-loaded liposomes present a unique analytical challenge: a low activity readout after encapsulation does not always mean the enzyme has been damaged. In many cases, scientists need to distinguish true activity loss from limited substrate access, incomplete release, surface adsorption, or batch variability introduced during manufacturing.
To build a reliable development package, formulation and analytical teams typically need evidence in three areas: whether enzymatic activity is retained after encapsulation, whether the lipid bilayer protects the payload during processing and storage, and whether critical quality attributes remain consistent across batches. This resource outlines practical strategies to generate that evidence using orthogonal analytical methods.
How to Verify Activity Retention
The processes involved in liposome generation—ranging from extrusion to microfluidic mixing—expose delicate protein payloads to shear stress, solvent interfaces, and thermal fluctuations. It is critical to differentiate between enzymes securely encapsulated within the aqueous core and those merely adsorbed to the outer lipid membrane or inactivated by the process.
What Should Be Measured?
- Free enzyme activity before formulation to establish a baseline.
- Apparent activity of intact liposomes to assess background or surface-associated activity.
- Total recoverable activity after liposome lysis.
- Protein recovery and encapsulation efficiency.
Useful Methods & Considerations
For many enzyme assays, intact liposomes can limit or prevent substrate access to the encapsulated cargo, making a controlled lysis step necessary for total activity measurement. Researchers typically employ target-specific fluorometric or colorimetric substrate assays.
Validating a non-denaturing detergent (such as 0.1% to 1% Triton X-100) is necessary to release the cargo for quantitation without denaturing the active site. Comparing the activity of intact liposomes versus lysed liposomes allows teams to separate true inactivation from restricted substrate access.
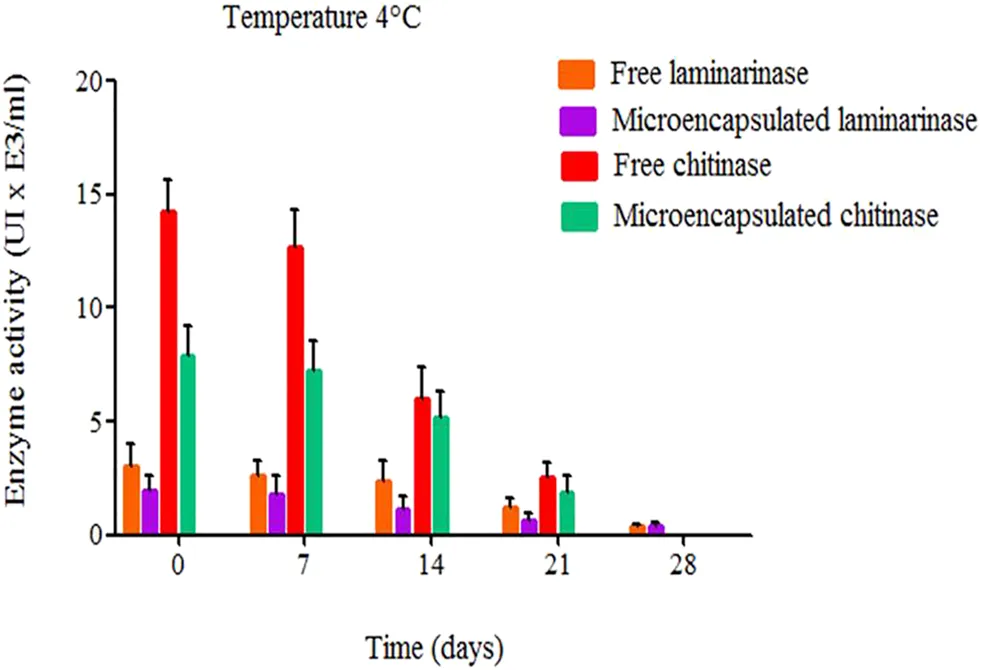
Insights from Literature
A study by Alonso-Estrada et al. investigated the thermodynamic aspects of immobilizing chitinase and laminarinase in liposomes. The data demonstrated that liposomal encapsulation preserved the structural integrity of these enzymes, significantly extending their functional half-life and specific activity over extended storage periods compared to free enzymes (as shown in Fig.1). This underscores the capacity of engineered lipid bilayers to maintain complex kinetic parameters.
How to Verify Protection During Processing and Storage
Achieving initial encapsulation is only the first step; analytical teams must confirm that the lipid bilayer acts as a physical shield. Formulations are typically subjected to controlled stress tests that simulate both manufacturing extremes and the intended physiological environment.
1. Processing Stress Assessments
To evaluate stability against manufacturing stresses, liposomes should be subjected to agitation/shear challenges, freeze-thaw cycles, or temperature excursion studies. If relevant to the administration route, nebulization or homogenization stress tests help determine if physical forces induce premature enzyme leakage or vesicle rupture.
2. Protease Challenge
To assess protection against physiological degradation, formulations are incubated with broad-spectrum proteases (e.g., Proteinase K or Trypsin) in vitro. If the enzyme is effectively protected within intact liposomes, the protected fraction should remain detectable (e.g., via SDS-PAGE) after the protease challenge, whereas free or surface-associated enzyme is expected to be degraded.
3. Stability in Biological Matrices
The behavior of the formulation in biological fluids is a useful predictor of in vivo performance. Formulations are often incubated in physiological matrices like fetal bovine serum (FBS) or human plasma at 37°C. Monitoring the samples over time helps characterize premature leakage and activity decay.
Recommended Verification Package for Enzyme-Loaded Liposomes
To generate a robust development package, scientists should not rely on a single readout. A typical orthogonal evaluation strategy includes:
- Encapsulation efficiency and recovered enzyme content.
- Activity assay before and after encapsulation.
- Activity assay after lysis to measure total recoverable activity.
- Protease challenge to assess protection of the encapsulated fraction.
- Serum/plasma incubation for leakage and activity stability.
- Particle size, PDI, and zeta potential across batches.
- Morphology confirmation by Cryo-TEM.
- Orthogonal purity/integrity analysis for lipid and enzyme components.
Implementing an orthogonal Formulation Analysis and Characterization strategy is key to distinguishing formulation artifacts from actual payload degradation.
How to Demonstrate Batch Consistency
For translational programs and later-stage development, establishing a framework to define Critical Quality Attributes (CQAs) is often important for supporting process consistency. To support internal decision-making, tech transfer, and later regulatory documentation, analytical methods must trend these parameters across multiple lots.
* Acceptance criteria should be product-specific and justified based on formulation design, intended use, and process capability.
| Critical Quality Attribute (CQA) | Recommended Analytical Method | Illustrative Acceptance Considerations |
|---|---|---|
| Particle Size & Distribution (PDI) | Dynamic Light Scattering (DLS) / AF4-MALS | Target size range established; reproducible PDI limits (e.g., < 0.2) |
| Encapsulation Efficiency (EE%) | SEC-HPLC / Ultrafiltration & Centrifugation | Consistent EE baseline with defined variance limits |
| Enzyme Catalytic Activity | Specific Substrate Kinetics (Post-lysis) | Maintained specific activity profile relative to unformulated control |
| Morphology | Cryo-Transmission Electron Microscopy (Cryo-TEM) | Confirmed unilamellar/multilamellar structure; minimal free drug/aggregates |
| Lipid & Cargo Integrity | LC-MS / RP-HPLC-CAD | Tracking lipid degradation products and peptide modifications |
How to Maintain Analytical Comparability During Scale-Up
Transitioning an optimized formulation from bench-top R&D to pilot or clinical scale involves significant changes to mixing dynamics, residence times, shear forces, and temperature histories. Because bench-scale data may not translate directly to continuous processes like high-pressure homogenization or tangential flow microfluidic mixing, analytical monitoring must adapt.
During scale-up runs, teams should trend key parameters—activity, EE%, size/PDI, leakage profiles, and morphology—across multiple stages to confirm that larger volumes and increased shear do not induce enzyme denaturation or alter the lipid phase transition behavior.
Building a Robust Verification Strategy
For enzyme-loaded liposomes, activity retention, protection, and batch consistency should be demonstrated through orthogonal analytical methods rather than a single readout. A strong verification strategy combines enzyme activity assays, encapsulation analysis, protection studies, physicochemical characterization, and batch trending data to support formulation optimization and scale-up decisions.
Ready to establish a robust verification strategy?
Explore our Liposome-Encapsulated Enzyme Development services or talk to our scientists today.
Frequently Asked Questions
Separating free enzyme from intact liposomes typically involves Size Exclusion Chromatography (SEC) or ultrafiltration (using centrifugal filter units). Once the unencapsulated fraction is removed or quantified, you can lyse the liposomes to measure the internal protein content. Because orthogonal methods are often needed, teams commonly verify SEC results with dynamic light scattering (DLS) to ensure the separation process didn't compromise liposome integrity.
A lower readout doesn't always mean the enzyme denatured. Intact lipid bilayers can restrict substrate diffusion to the active site. To confirm true activity retention, compare the specific activity of the free enzyme against the formulated enzyme *after* a controlled lysis step (e.g., using Triton X-100). If activity remains low post-lysis, investigate processing shear, temperature stress, or unfavorable lipid-protein interactions.
A protease protection assay is the standard approach. Incubate the liposomes with a protease like Proteinase K. Surface-associated enzymes will be degraded, while internal enzymes remain protected. Analyzing the pre- and post-incubation samples via SDS-PAGE or activity assays will clearly distinguish the deeply encapsulated fraction from surface-bound cargo.
Because multiple CQAs should be trended across batches rather than relying on particle size alone, typical parameters include size/PDI, zeta potential, encapsulation efficiency, retained specific activity, and lipid degradation profiles. Establishing acceptable variances for these parameters early in development supports smoother technology transfer and scale-up.
Reference
- Alonso-Estrada, Dania, et al. "No solid colloidal carriers: Aspects thermodynamic the immobilization chitinase and laminarinase in liposome." Frontiers in Bioengineering and Biotechnology 9 (2022): 793340. https://doi.org/10.3389/fbioe.2021.793340
- Under Open Access license CC BY 4.0, without modification.
Online Inquiry
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.


