Mechanisms of Liposomal Adjuvants in
Enhancing Vaccine Immune Response
Unveiling the immunological pathways and physicochemical strategies of liposome-based adjuvants to enhance antigen presentation and shape immune profiles in vaccine development.
Introduction to Liposomal Adjuvants
The development of modern vaccines has shifted from live-attenuated or inactivated pathogens towards highly purified recombinant subunits, synthetic peptides, and nucleic acids. While these platforms offer superior safety profiles, they often suffer from poor immunogenicity due to the lack of "danger signals" required to activate the innate immune system. Consequently, the incorporation of effective adjuvants has become indispensable for generating robust, long-lasting protective immunity.
Among various adjuvant systems, liposomes—spherical vesicles composed of phospholipid bilayers—have emerged as one of the most versatile and potent platforms. Since their discovery in the 1960s and their first licensed application in the 1990s, liposomal adjuvants have been refined to not only serve as delivery vehicles but also as immunostimulators. Their biocompatibility, biodegradability, and the ability to encapsulate both hydrophilic and hydrophobic antigens make them unique tools for preclinical vaccine research.
Understanding the precise mechanisms by which liposomes enhance immune responses is critical for rational vaccine design. This article delves into the immunological pathways activated by liposomal adjuvants, the physicochemical parameters governing their efficacy, and strategies to optimize lipid-based formulations for specific research goals.
Looking for Custom Formulations?
Creative Biolabs offers comprehensive services for the design and synthesis of liposome-based adjuvants tailored to your specific antigen requirements.
Explore Liposome-based Adjuvant Development Service →Mechanistic Insights into Immune Activation
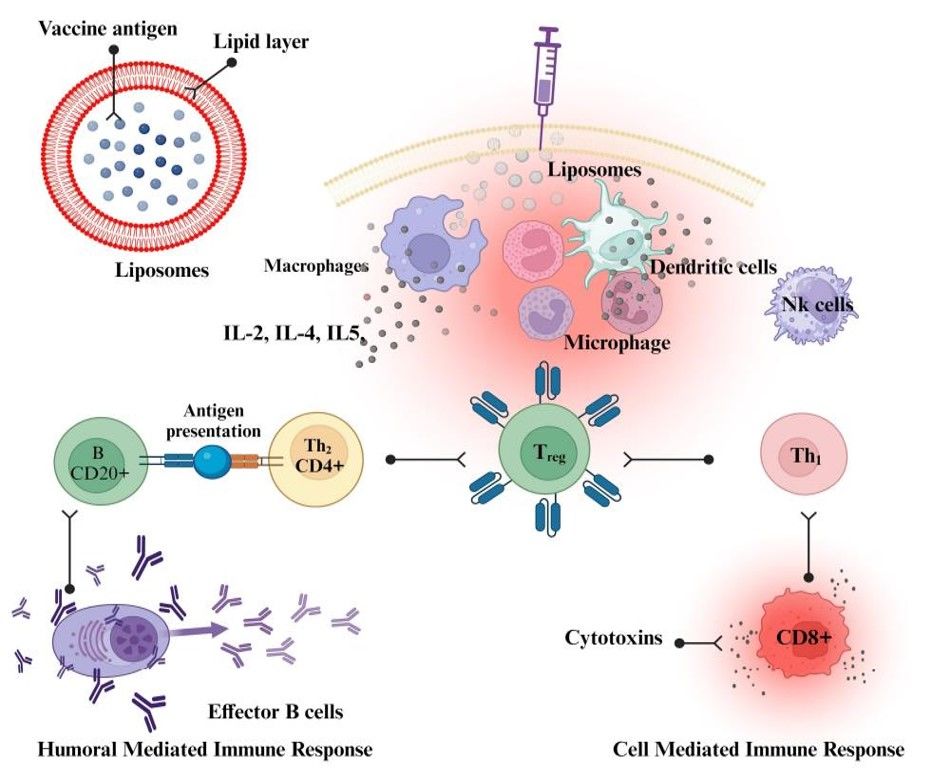
Fig.1 The mechanistic approach of liposomes as vaccine adjuvant.1,2
1. Antigen Persistence and Trafficking
While early theories emphasized the "depot effect"—the retention of antigen at the injection site—contemporary research suggests this is not the sole driver of immunogenicity. More critical is the creation of an immunocompetent environment that recruits innate immune cells (e.g., neutrophils, monocytes) and facilitates the trafficking of antigen-loaded APCs to draining lymph nodes (dLNs). Liposomes protect antigens from rapid enzymatic degradation and clearance, extending the window for uptake by these recruited cells.
2. Enhanced Uptake by Antigen Presenting Cells (APCs)
Liposomes act as particulate carriers that are avidly internalized by APCs, particularly dendritic cells and macrophages, via mechanisms such as phagocytosis, endocytosis, or membrane fusion. Compared to soluble antigens, which are often cleared rapidly or ignored by APCs, liposome-associated antigens are processed more efficiently. This processing leads to the loading of antigenic peptides onto MHC class II molecules, stimulating CD4+ T helper cell responses essential for antibody production.
3. Cross-Presentation and CD8+ T Cell Activation
Cross-presentation is a key mechanism for generating cytotoxic T lymphocyte (CTL) responses against intracellular pathogens and tumors. This process is not solely dependent on cationic surface charge. It involves complex interactions including membrane fusion, endosomal destabilization via protonatable lipids (inducing the "proton sponge" effect), and the specific positioning of the antigen. Encapsulating antigen or anchoring it to the membrane can significantly impact the efficiency of cytosolic delivery and subsequent MHC class I presentation.
4. Innate Immune Activation
Certain cationic liposomes have been reported to activate the NLRP3 inflammasome, serving as "danger signals" or Damage-Associated Molecular Patterns (DAMPs). This activation, which leads to the release of pro-inflammatory cytokines like IL-1β and IL-18, is often context-dependent. Factors such as lipid structure (e.g., DOTAP), dose, and the presence of impurities (like endotoxins) play a role in this pathway, amplifying the innate immune response and bridging it to adaptive immunity.
Physicochemical Modulation of Immune Response
The adjuvant potency of liposomes is strictly dictated by their physicochemical properties. Modifying these parameters allows researchers to shape the type of immune response.
Note: Immune polarization (Th1 vs. Th2) is also heavily influenced by antigen type, co-delivery of PRR agonists (e.g., TLR4, TLR9 ligands), and the route of administration.
Liposome Size
The size of the vesicle dictates its trafficking in vivo. Larger liposomes (>500 nm) tend to be retained at the injection site and are taken up by tissue-resident APCs (monocytes/macrophages). In contrast, smaller nanoparticles (<200 nm) can drain freely into the lymphatic vessels, reaching the lymph nodes directly where they interact with lymph node-resident dendritic cells and B cells.
Surface Charge
Surface charge is a critical determinant. Cationic liposomes generally exhibit stronger electrostatic interactions with the negatively charged cell membranes of APCs, leading to enhanced uptake. Cationic lipids like DOTAP are well-known for facilitating Th1-biased responses. However, charge density must be balanced against potential toxicity.
Membrane Fluidity and Rigidity
The choice of phospholipids (e.g., saturated DSPC vs. unsaturated DOPC) and cholesterol content determines bilayer rigidity. Rigid liposomes act as stable depots and resist rapid breakdown, potentially favoring sustained antigen presentation. Fluid liposomes may fuse more readily with cell membranes but release payload faster.
Antigen Loading Strategy
How the antigen is associated with the liposome—encapsulated in the aqueous core, inserted in the bilayer, or adsorbed on the surface—dramatically affects B cell recognition and APC processing. Surface-exposed antigens can directly cross-link B cell receptors (BCRs) to stimulate antibody production, while encapsulated antigens are protected and primarily processed after uptake.
PEGylation and Surface Modification
While PEGylation improves stability and circulation time, it can hinder cellular uptake ("PEG dilemma"). In vaccine design, sheddable PEG lipids or careful optimization of PEG density is required to balance lymph node trafficking with efficient APC internalization.
Need high-quality liposomal products for your vaccine research?
View Liposomal Vaccine or Adjuvant ProductsFrom Mechanism to Design: A Selection Guide
Choosing the right liposomal parameters is essential for achieving specific immunological outcomes. The table below outlines recommended features and verification assays for common research goals.
| Research Goal | Recommended Liposome Features | Suggested Assays & Readouts |
|---|---|---|
| Strong CTL Response (Th1) |
|
|
| Robust Humoral Immunity (Th2) |
|
|
| Mucosal Immunity (IgA) |
|
|
Frequently Asked Questions
Unlike aluminum salts (alum), which primarily induce a humoral (Th2) response and have limited ability to stimulate cellular immunity, liposomes are versatile platforms. They can encapsulate various antigens, codeliver immunostimulators (like TLR agonists), and be engineered to induce both Th1 (cellular) and Th2 (humoral) responses. Furthermore, liposomes can facilitate cross-presentation to activate CD8+ cytotoxic T cells, which alum cannot do effectively.
Liposomal formulations can be susceptible to hydrolysis and oxidation of phospholipids, aggregation, or leakage of the encapsulated payload during storage. To mitigate these issues, formulation scientists often use high-transition temperature lipids, add cholesterol for rigidity, include antioxidants, or employ lyophilization (freeze-drying) with cryoprotectants to ensure long-term stability ex vivo.
Yes. Liposomes are excellent candidates for mucosal delivery (e.g., intranasal, oral). They can protect antigens from the harsh environment of the gastrointestinal tract or nasal enzymes and enhance uptake by M cells in the mucosal-associated lymphoid tissue (MALT), triggering both systemic (IgG) and mucosal (IgA) immunity.
Encapsulation efficiency is typically measured by separating free antigen from liposome-associated antigen using techniques like size exclusion chromatography (SEC), ultracentrifugation, or dialysis. The quantity of protein in the separated fractions is then analyzed via BCA assay, HPLC, or other quantitative methods suitable for your specific antigen.
References
- Moni, Sivakumar S., et al. "Advancements in vaccine adjuvants: the journey from alum to nano formulations." Vaccines 11.11 (2023): 1704. https://doi.org/10.3390/vaccines11111704
- Under Open Access license CC BY 4.0, without modification.
Online Inquiry
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.


