Cationic Liposome Development for Efficient Transfection with Reduced Toxicity
Optimize cationic liposome development for plasmid DNA, mRNA, and siRNA delivery by balancing transfection efficiency, physicochemical stability, and cytotoxicity.
Why Cationic Liposome Development Is Challenging
Cationic liposomes remain the cornerstone of non-viral nucleic acid delivery. Defined by their positively charged lipid headgroups, these vesicles efficiently form electrostatic interactions with negatively charged genetic materials. This condensation leads to the formation of tightly packed lipoplexes. The surface charge also plays a critical role in facilitating interactions with negatively charged proteoglycans on cell membranes, driving robust cellular uptake via endocytosis.
However, researchers and formulation scientists face a persistent dilemma. The strong cationic nature required for high transfection efficiency is intrinsically linked to dose-limiting cytotoxicity. An excessive positive charge can increase the risk of cellular membrane damage, trigger intracellular reactive oxygen species (ROS) production, and activate complement systems during in vivo administration.
Reducing the cationic lipid content to lower toxicity often leads to unstable physicochemical properties, weak payload complexation, and poor transfection outcomes. Overcoming this trade-off requires a systematic and rational development workflow rather than empirical trial and error. Navigating these complexities is central to professional cationic liposome formulation development, focusing heavily on architectural modifications, precise optimization, and comprehensive biological validation.
Core Challenges Addressed in Development
- Efficiency vs. Toxicity: Balancing cellular entry requirements against membrane disruption and apoptosis.
- Endosomal Escape: Ensuring nucleic acids successfully exit the endosome before lysosomal degradation.
- Batch Reproducibility: Preventing fluctuations in particle sizing, PDI, and encapsulation efficiency.
- Serum Instability: Avoiding premature payload release and macroscopic aggregation within biological fluids.
Key Design Elements in Cationic Liposome Development
Modern cationic liposome design has evolved far beyond rudimentary DOTMA or DOTAP formulations. Designing a system capable of robust in vitro and in vivo performance requires an intelligent integration of multiple functional modules.
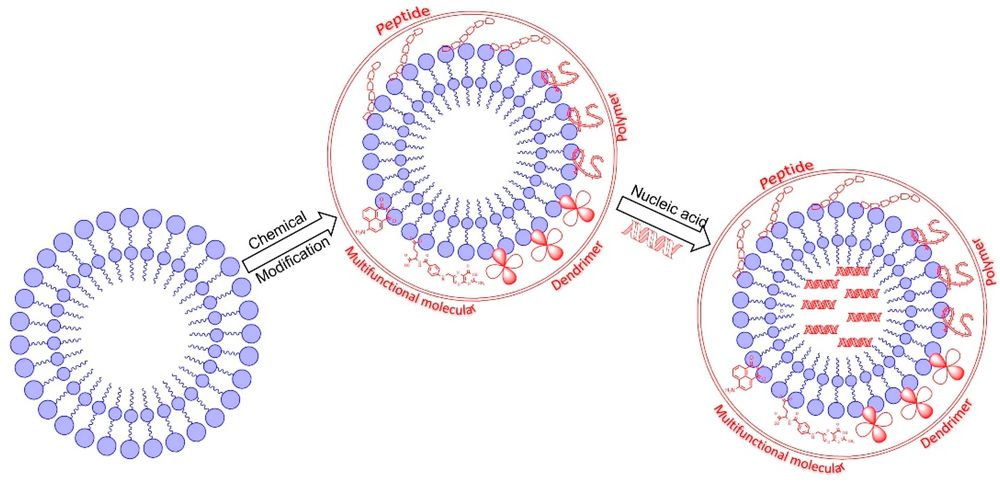
Fig.1 The structure of liposome nanoparticles modified with different chemical molecules. 1,2
This figure illustrates how cationic liposome systems can be rationally engineered through peptide, polymer, dendrimer, or functional-molecule modification. For cationic liposome development, the key is not simply increasing positive charge, but improving intracellular delivery efficiency while mitigating toxicity and nonspecific biological interactions through smarter structural design.
Incorporating complex architectures like dendrimers may promote endosomal escape through buffering effects and membrane destabilization mechanisms (often termed the "proton sponge" effect). This allows the rapid release of genetic materials into the cytosol before severe degradation occurs.
Furthermore, modifying the surface with specific peptides (such as cell-penetrating sequences) encourages targeted receptor-mediated endocytosis, which can reduce the absolute dependency on excessive cationic charge. Integrating polymers like PEG-lipids can reduce nonspecific interactions and improve colloidal behavior in serum-containing environments. The integration of all these components forms the backbone of advanced cationic liposome for nucleic acid delivery.
How to Optimize Formulation Parameters for pDNA, mRNA, and siRNA Delivery
The biophysical behavior of a cationic liposome formulation is deeply influenced by the nature of its payload. A "one-size-fits-all" approach rarely succeeds in clinical applications. Utilizing precise liposome formulation optimization is critical to adapt the vector for different nucleic acid geometries.
pDNA Delivery
Plasmid DNA requires nuclear entry. Stronger condensation may be required to package the large structural footprint, but oversized lipoplex complexes can drastically reduce ultimate gene expression levels.
mRNA Delivery
Since mRNA functions in the cytosol, developers must prioritize rapid endosomal release pathways and strictly preserve payload integrity against ubiquitous environmental RNases.
siRNA Delivery
While siRNA is a much smaller cargo, it exhibits high sensitivity to serum protein interactions. The formulation must offer robust protection during circulation alongside efficient intracellular release efficiency.
Critical Development Variables in Cationic Liposome Formulation
| Parameter | Function & Impact in Development |
|---|---|
| Cationic Lipid Type | Determines primary complexation strength and baseline cytotoxicity. Selecting biodegradable or specific headgroups strongly influences intracellular clearance. |
| Helper Lipid Type | Lipids like DOPE adopt inverted hexagonal structures under acidic conditions, dictating the fusogenic capacity and endosomal escape efficiency. |
| Cholesterol Content | Modulates membrane fluidity and rigidity. Vital for in vivo stability to prevent premature nucleic acid leakage in circulation. |
| PEG-lipid Density | Regulates stealth properties and colloidal stability. Over-PEGylation can hinder cellular uptake, requiring fine-tuning of molar percentages. |
| N/P Ratio | The ratio of lipid nitrogen (N) to nucleic acid phosphate (P). Crucial for balancing encapsulation efficiency against potential membrane damage. |
| Particle Size / PDI | Directly affects biodistribution and cellular internalization pathways. Must be tightly controlled for batch acceptance. |
| Mixing Method & Conditions | Flow rate ratios and mixing speeds dictate the self-assembly process, which heavily impacts structural homogeneity and reproducibility. |
Strategies to Improve Transfection While Reducing Toxicity
Mitigating the safety profile is a paramount objective during cationic liposome optimization. Formulations that excel in transfection but trigger widespread apoptosis are fundamentally non-viable for downstream applications. To lower toxicity, formulation scientists deploy advanced strategies tailored for biological environments.
Introducing biodegradable linkers (such as ester or disulfide bonds) within the hydrophobic tails of the cationic lipids is highly recommended. These structures are rapidly metabolized by intracellular esterases or the reducing environment of the cytosol, effectively neutralizing the residual charge after the payload is delivered.
Additionally, asymmetrical helper lipid ratios and stimulus-responsive charge-masking polymers can shield the particles until they reach the intended microenvironment. Verifying these improvements requires rigorous formulation safety evaluation, including detailed cell viability assays, hemolysis profiling, and cytokine release measurements to assure a benign profile.
Core Toxicity Mitigation Strategies
- 1. Biodegradable Lipid Chemistries Implementation of cleavable bonds to ensure rapid post-transfection intracellular clearance, reducing sustained cellular stress.
- 2. Temporary Charge Shielding Utilizing pH-sensitive coatings that conceal the cationic charge during systemic circulation but expose it in acidic endosomal environments.
- 3. Strict N/P Ratio Control Empirically identifying the absolute minimum ratio required for total complexation without introducing toxic free lipids.
How to Improve Stability and Batch-to-Batch Reproducibility
A major pain point in cationic liposome development is moving a formulation from lab-scale benchwork to larger batches. Poor batch-to-batch reproducibility can halt research efforts entirely. Ensuring consistency necessitates establishing rigid controls over raw materials and the precise biophysical forces used during self-assembly.
1. Raw Material Consistency
Fluctuations in lipid purity or degradation during storage directly alter the phase behavior of the liposomes. Standardizing lipid sourcing and validating raw material integrity are the first steps to reproducibility.
2. Process Engineering
Replacing traditional thin-film hydration with scalable microfluidic mixing allows for precise control over the Total Flow Rate (TFR) and Flow Rate Ratio (FRR). This governs the self-assembly kinetics, ensuring uniform nanoparticle sizing across varying scales.
3. Defined Release Criteria
Setting uncompromising QC thresholds for nucleic acid loading inconsistency, post-insertion variability, and residual solvent levels ensures that no defective batch progresses to in vivo testing stages.
Critical Characterization Methods for Cationic Liposome Formulations
Comprehensive characterization is essential to confirm that the developed cationic liposome transfection system meets the rigorous demands of modern nucleic acid delivery. A successful formulation development protocol must incorporate a multi-tiered analytical approach to assess physical properties, encapsulation metrics, and biological behavior.
Initially, Dynamic Light Scattering (DLS) and Electrophoretic Light Scattering (ELS) are utilized to establish the particle size, Polydispersity Index (PDI), and exact zeta potential. Next, dye-exclusion assays (like RiboGreen or PicoGreen) determine the Encapsulation Efficiency (EE%), verifying the strength of the lipoplex condensation. Furthermore, dynamic assessments including serum stability and storage stability tests are conducted to observe potential macroscopic aggregation or payload leakage over time. Finally, functional validation is achieved through parallel in vitro transfection assays and cell viability screens (MTT/CCK-8) to document the crucial balance between efficacy and toxicity. Meeting strict release criteria across all these metrics confirms high reproducibility across batches.
Explore Lipid-based Basic Characterization ServiceWhen Custom Cationic Liposome Development Is Needed
Off-the-shelf reagents are often insufficient for specialized cell lines, sensitive in vivo models, or unique therapeutic payloads. When you require engineered specificity, optimized safety margins, and scalable manufacturing methodologies, custom formulation is imperative.
Access Cationic Liposome Development ServiceFrequently Asked Questions
The primary factors include the specific selection of cationic lipid chemistry (which dictates binding affinity and baseline toxicity), the inclusion of helper lipids like DOPE or cholesterol to govern membrane phase transitions, the integration of PEG-lipids for colloidal stability, and the precise optimization of the N/P ratio. Process controls such as mixing speed and temperature are also critical factors influencing the final lipoplex architecture.
Cationic liposomes improve transfection efficiency by spontaneously complexing with negatively charged nucleic acids, protecting them from nuclease degradation. Their net positive surface charge strongly interacts with anionic cellular membranes, facilitating robust endocytosis. Furthermore, specially formulated helper lipids promote endosomal membrane fusion, ensuring the genetic cargo successfully escapes into the cytoplasm for translation or transcription.
Toxicity can be reduced by substituting traditional permanent cationic lipids with biodegradable alternatives that are cleaved post-transfection. Other strategies include fine-tuning the N/P ratio to eliminate excess free lipid, adjusting helper lipid stoichiometry to lower total cationic dependency, and utilizing protective PEGylation to shield the strong surface charge from nonspecific interactions in serum.
There is no universal optimal N/P ratio, as it heavily depends on the specific lipid combination and the physicochemical properties of the payload. Generally, effective ratios range between 2:1 and 5:1. Larger payloads like pDNA may require slightly varied condensation parameters compared to smaller, highly structured siRNA molecules. Extensive screening matrices are required during early development to establish the optimal ratio for each application.
Helper lipids like DOPE adopt distinct geometric shapes that facilitate the transition of the liposome from a stable lamellar phase to an inverted hexagonal structure, which actively destabilizes the endosomal membrane to promote payload release. Cholesterol acts as a membrane modulator, filling gaps between lipid tails to reduce membrane permeability, effectively preventing premature leakage of the complexed nucleic acids while circulating in the blood.
Improving batch-to-batch reproducibility requires standardizing raw lipid purity, switching from variable bulk mixing methods (like thin-film hydration) to scalable, automated microfluidic systems, and strictly controlling process parameters like flow rate ratios (FRR) and temperature. It is also vital to implement robust Quality Control release criteria based on DLS and ELS measurements before downstream processing.
References
- Gao, Yongguang, et al. "Recent advance of liposome nanoparticles for nucleic acid therapy." Pharmaceutics 15.1 (2023): 178. https://doi.org/10.3390/pharmaceutics15010178
- Under Open Access license CC BY 4.0, without modification.
Online Inquiry
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.


