Multivesicular Liposomes (MVLs):
Engineering High Payload Capacity for Sustained Drug Release
A robust engineering framework designed for formulation scientists and R&D leaders to overcome the limitations of conventional liposomes, enabling exceptional hydrophilic payload capacity, smoothed burst release, and extended therapeutic duration.
Why Conventional Liposomes Often Fall Short for High-Payload Sustained Delivery
In the landscape of long-acting injectables, local sustained analgesia, and peptide/protein delivery, formulation scientists continually encounter significant bottlenecks. Traditional liposomal architectures—such as small unilamellar vesicles (SUVs) and large unilamellar vesicles (LUVs)—possess inherently limited aqueous cores. When attempting to formulate highly hydrophilic payloads, such as complex peptides or large proteins, this restricted volume inevitably leads to suboptimal encapsulation efficiency and low payload capacity.
Furthermore, conventional multilamellar vesicles (MLVs), characterized by their concentric, onion-like lipid bilayers, fail to provide a definitive solution. Although they offer greater lipid mass, the aqueous volume fraction remains severely constrained by the tight inter-lamellar spacing. For R&D teams, this translates into a persistent formulation dilemma: attempting to force high doses of water-soluble therapeutics into these structures frequently results in uncontrolled burst release upon administration.
This challenge is compounded when trying to balance extended release cycles with strict requirements for in vivo membrane stability, structural integrity, and batch-to-batch consistency during manufacturing scale-up. To solve these critical pain points, it is necessary to transition from basic liposomal encapsulation to advanced architectural engineering utilizing Multivesicular Liposomes (MVLs).
Core Challenges in Long-Acting Formulations
- 1 Insufficient Aqueous Volume: Conventional liposomes restrict the high-efficiency loading of highly hydrophilic drugs and large biologics.
- 2 Uncontrollable Burst Release: High concentration gradients combined with thin bilayer barriers often result in a massive initial drug dump, increasing toxicity risks and shortening the therapeutic window.
- 3 Structural Instability & Scalability: Ensuring the complex internal architecture remains stable throughout shelf-life and biological residence while maintaining batch-to-batch consistency during clinical scale-up.
- 4 Lack of an Engineering Framework: Difficulty in establishing a clear predictive model that links structural formulation inputs to in vitro and in vivo release behaviors.
The Architectural Superiority of the Multivesicular Framework
The defining characteristic that distinguishes MVLs from conventional systems is their unique internal topology. Establishing this structural baseline is critical for understanding their functional capabilities in clinical applications.
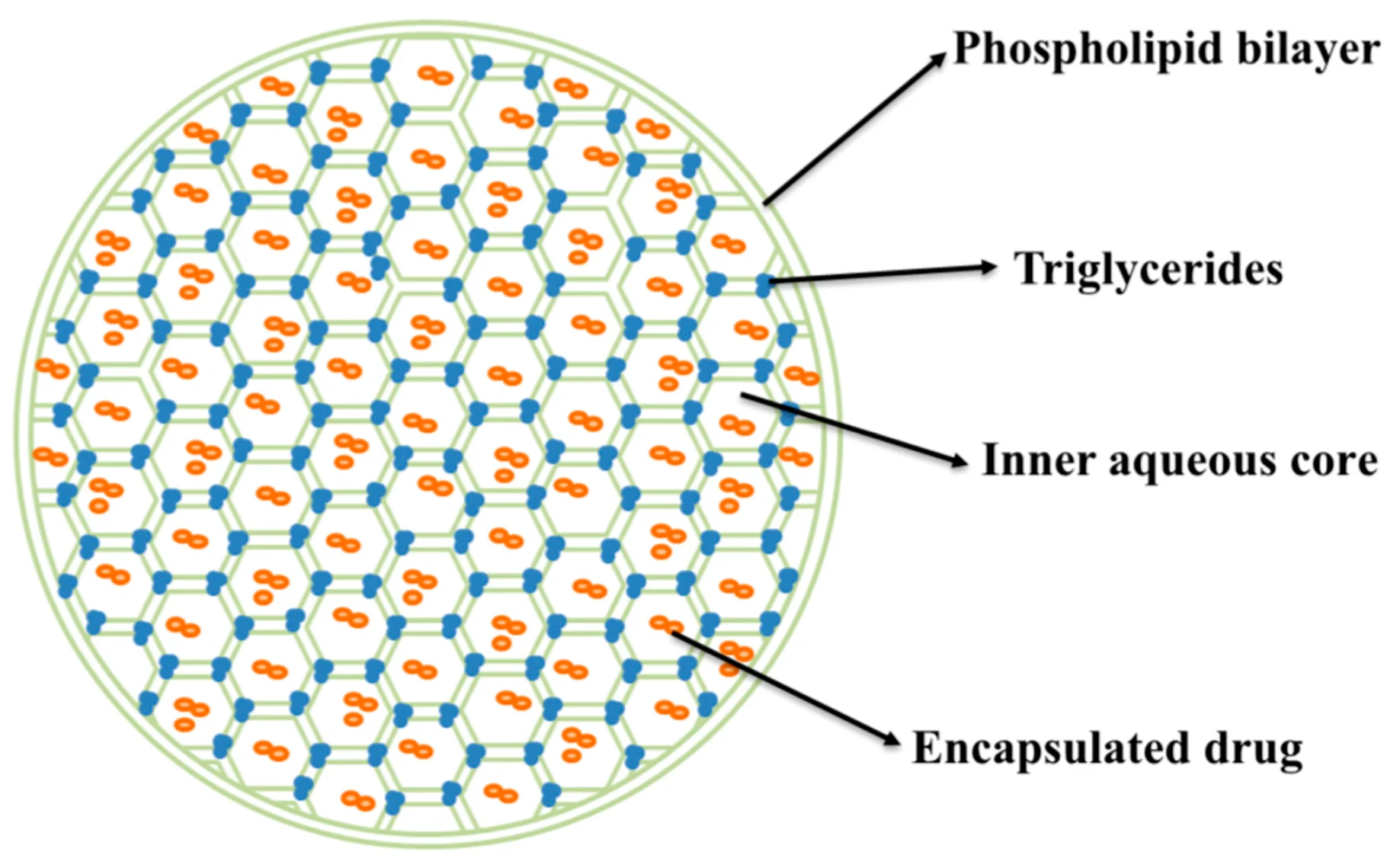
MVLs contain multiple non-concentric aqueous chambers, creating a substantially larger internal aqueous space than conventional unilamellar or multilamellar liposomes. Unlike MLVs where aqueous layers represent a minute fraction of the total particle volume, this honeycomb-like internal architecture provides a strong structural basis for high encapsulation of hydrophilic payloads and improved drug-to-lipid loading strategies.
In recent engineering studies, such as the formulation development for sustained delivery of liraglutide utilizing mixed phospholipids (Zhang et al.), the structural distinctness of MVLs was central to resolving peptide encapsulation limits. The large internal aqueous space may support higher drug-to-lipid ratios, although the final loading and structural integrity remain formulation-dependent.
By optimizing the specific lipid composition—balancing amphipathic phospholipids with critical neutral lipids—the size and number of these internal non-concentric chambers can be precisely controlled, providing an engineering parameter directly linked to payload retention.
Engineering Tunable Release Kinetics and Smoothing Burst Release
For BD and technical evaluation teams comparing platform technologies, the release profile is the primary performance metric. Traditional liposomes typically exhibit release governed by simple diffusion or immediate membrane rupture, resulting in steep initial concentration peaks (Cmax) and rapid clearance.
Because the active pharmaceutical ingredient (API) is distributed across hundreds of individual internal chambers, the release is not a singular event. Sustained release from MVLs is generally attributed to a combination of diffusion across lipid septa, gradual membrane erosion, and staged exposure of deeper internal compartments. As the outermost lipid membranes undergo gradual erosion and biological degradation, the interior compartments remain intact. The drug from the outermost chambers diffuses first, followed sequentially by payload release from the deeper, newly exposed chambers. This creates a staggered, controlled release profile that can be maintained for days or even weeks.
Based on recent engineering studies, tuning membrane composition can further modulate MVL stability and release kinetics. Adjusting the ratio of high-transition-temperature lipids (e.g., DSPC, DPPC), modulating cholesterol content, or selecting specific acidic lipids (like DPPG) directly alters the fluidity and degradation rate of the bilayers. A rigorous understanding of these dynamics requires precise in vitro release kinetics analysis to mathematically model the relationship between formulation variables and the resulting release curve, ensuring accurate predictability before advancing to in vivo models.
| Delivery Platform | Internal Architecture | Hydrophilic Encapsulation | Primary Release Mechanism | Burst Release Risk |
|---|---|---|---|---|
| Conventional SUVs/LUVs | Single aqueous core | Low | Simple Diffusion / Rupture | High |
| PLGA Microspheres | Polymer matrix | Moderate | Bulk Erosion / Diffusion | Moderate (dependent on porosity) |
| Multivesicular Liposomes (MVLs) | Non-concentric aqueous chambers | Very High | Sequential Disruption & Diffusion | Moderate to Low (Formulation-Dependent) |
Ensuring Structural Stability and Manufacturing Consistency
The structural integrity of MVLs relies heavily on a specialized lipid formulation. Neutral lipids such as triolein are commonly used in MVL formulations to help stabilize the junctions between internal compartments, although the optimal composition remains formulation-specific. These neutral lipids partition into the hydrophobic bilayer intersections (the nodes of the honeycomb matrix), physically stabilizing the non-concentric architecture and preventing the fusion of internal compartments into a single unstable core.
Formulators face rigorous challenges when scaling up the double-emulsion (water-in-oil-in-water, W/O/W) manufacturing process required to form MVLs. Precisely controlling shear forces, temperature, and solvent removal rates is imperative to dictate the final particle size and internal compartment density. Variations in these parameters lead to inconsistent release behaviors.
Implementing comprehensive formulation stability monitoring service protocols is vital to evaluate the preservation of the architectural framework across varying storage conditions and biological media. Maintaining structural fidelity ensures the theoretical release kinetics are translated faithfully into physical reality.
Critical Scale-Up Parameters
- Primary Emulsion (W/O): Optimization of lipid-to-aqueous phase ratios to maximize hydrophilic entrapment.
- Secondary Emulsion (W/O/W): Precise shear control to define outer particle diameter (typically 10-100 μm) ensuring consistent injectability.
- Solvent Extraction Kinetics: Controlled gas sparging or cross-flow filtration to dictate the solidification rate of the lipid framework.
Clinical Translation and Biocompatibility
A major advantage of MVLs over traditional polymeric matrices (such as standard PLGA microspheres) lies in their superior biocompatibility profile, particularly concerning the microenvironment generated during degradation. PLGA degradation generates acidic monomers (lactic and glycolic acid) which can drastically lower local pH, potentially causing denaturation, aggregation, or loss of bioactivity for sensitive encapsulated proteins and peptides.
Conversely, MVLs are composed entirely of natural or synthetic physiological lipids. Their biological degradation yields benign lipid metabolites, helping to maintain a more neutral pH microenvironment. Compared with polymeric depot systems, MVLs may offer a more favorable microenvironment for sensitive peptides and proteins, especially where acidity-induced instability is a concern. This makes MVLs an attractive option for liposome encapsulated protein peptide development.
Long-Acting Local Analgesia
By encapsulating local anesthetics (e.g., bupivacaine) within the multi-compartment structure, MVL-based local anesthetic depots have been explored to extend analgesic duration beyond that of conventional solutions, while reducing the sharp exposure peaks associated with immediate release. The controlled sequential breakdown of the chambers helps prevent systemic toxicity caused by sudden Cmax spikes, offering an effective approach to post-operative pain management.
Systemic Peptide Delivery
Peptides, such as GLP-1 receptor agonists (e.g., liraglutide), suffer from short in vivo half-lives due to rapid enzymatic degradation and renal clearance. MVL encapsulation shields these peptides from protease activity while ensuring a sustained release depot at the subcutaneous injection site, drastically reducing dosing frequency and improving patient compliance.
Frequently Asked Questions
Unlike conventional unilamellar liposomes (which have a single aqueous core) or multilamellar vesicles (which consist of concentric, onion-like lipid layers), MVLs possess a unique honeycomb-like architecture. They consist of numerous non-concentric aqueous chambers separated by a network of intersecting lipid bilayers. This multi-compartment design drastically increases the internal aqueous volume, making them highly efficient for encapsulating large quantities of hydrophilic drugs, peptides, and proteins.
MVLs achieve sustained release through a process of "sequential disruption." Rather than releasing all contents simultaneously via simple diffusion or a single membrane rupture, the outermost lipid chambers of the MVL degrade and release their payload first. As the outer layers are cleared or erode in the biological environment, deeper internal chambers are exposed and begin to release their contents. This staggered, compartmentalized release mechanism effectively smooths the initial burst release and extends the therapeutic window.
Neutral lipids, specifically triglycerides like triolein, play a critical structural role in MVLs. Due to their extreme hydrophobicity, these lipids partition into the junction points—or nodes—where multiple lipid bilayers intersect within the honeycomb matrix. By occupying these junctions, neutral lipids stabilize the complex non-concentric architecture, preventing the internal chambers from fusing together and collapsing into a conventional, less efficient vesicle structure.
The primary advantage lies in biocompatibility and the preservation of biological activity. When PLGA (poly lactic-co-glycolic acid) microspheres degrade, they produce acidic monomers that can significantly lower the local pH within the polymer matrix. This acidic microenvironment can denature or aggregate sensitive peptides and proteins. MVLs, composed of physiological lipids, degrade into neutral, naturally occurring lipid metabolites, thereby maintaining a benign environment that protects the structural integrity and bioactivity of encapsulated macromolecules.
Yes. Although MVLs are typically larger than conventional liposomes (often ranging from 10 to 100 μm in diameter to act as a localized depot), their lipid bilayer structure remains highly deformable. Practical injectability depends on particle size distribution, suspension properties, formulation robustness, and needle selection.
Reference
- Zhang, Runpeng, et al. "Sustained Delivery of Liraglutide Using Multivesicular Liposome Based on Mixed Phospholipids." Pharmaceutics 17.2 (2025): 203. https://doi.org/10.3390/pharmaceutics17020203
- Under Open Access license CC BY 4.0, without modification.
Online Inquiry
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.


