Liposome Payload Troubleshooting:
Root Causes of Low Encapsulation, Precipitation & Uncontrolled Release
A troubleshooting resource for scientists diagnosing low encapsulation, payload precipitation, and uncontrolled release in liposomal systems—linking observable failure modes to formulation variables, loading strategy, and analytical readouts.
Liposome Formulation Challenges: Encapsulation, Stability, and Release
In liposomal payload development, formulations often appear successful by size and macroscopic appearance but fail on the metrics that actually matter for in vivo translation: encapsulation efficiency, colloidal stability under physiological conditions, and controlled release kinetics.
This resource is designed for preformulation scientists, CMC managers, and analytical development teams to break these failures into root-cause categories. It demonstrates how lipid composition, cholesterol level, surface chemistry, loading method, buffer conditions, and analytical design can each contribute to observed failures.
Because low encapsulation, precipitation, and uncontrolled release are inherently coupled phenomena, troubleshooting should focus on the underlying thermodynamic and biophysical mechanisms rather than treating isolated symptoms.
Diagnosing Low Encapsulation Efficiency
A. What You Observe
- Encapsulation Efficiency (EE) < 15% despite high initial drug inputs.
- Initial loading appears acceptable, but payload drops drastically after purification (dialysis, SEC, or TFF).
- Acceptable particle size and PDI, but unacceptably low retained drug concentration.
B. Most Likely Root-Cause Buckets
The failure usually falls into one of these distinct categories:
- Payload–Lipid Compatibility Mismatch: The drug is neither highly hydrophilic (to stay in the core) nor highly lipophilic (to partition into the bilayer).
- Internal Volume Limitation: Passive loading is often constrained by the trapped aqueous volume (typically 2-5% of total volume).
- Gradient Establishment Failure: In active (remote) loading, the transmembrane gradient (pH or ion) dissipates prematurely.
- Thermal Processing Errors: Loading temperature falls below the required phase transition temperature ($T_m$) of the main lipid.
C. What to Check First
- Measure post-loading leakage before the final assay step.
- Verify intraliposomal pH and gradient persistence over the duration of the loading process.
- Check the theoretical lipid:drug ratio against the absolute trapped volume ceiling.
- Confirm the completeness of external buffer replacement.
D. What to Change Next
- Switch from passive to active remote loading if the payload is an amphipathic weak base/acid.
- Change internal salt/gradient chemistry (e.g., from ammonium sulfate to calcium acetate for better retention).
- Adjust loading temperature relative to the bilayer state.
- Perform robust EE analysis to validate the separation method.
🚨 Typical Misinterpretations: Formulation or Assay Artifact?
Often, the formulation is fine, but the analytical method fails. Common artifacts include: The free-drug separation step itself (e.g., spin columns) exerting excessive shear and causing leakage; incomplete liposome lysis underestimating the encapsulated fraction; or assay signal quenching leading to severe under-recovery. Always validate mass balance before changing the formulation.
Physical Instability: Precipitation and Aggregation
Payload precipitation is not a single phenomenon. It can occur within the core, within the membrane, or in the bulk media, each driven by different biophysical triggers.
1. Intraliposomal Precipitation
During active remote loading, forming a precipitate inside the aqueous core is often beneficial (it "traps" the drug). However, if crystallization is uncontrolled, large crystal morphologies can mechanically puncture the bilayer from the inside out, causing massive leakage. Next Step: Alter the internal counterion or adjust the drug-to-lipid ratio to control crystal size.
2. Membrane-Associated Phase Separation
Attempting to over-load highly lipophilic drugs into the bilayer frequently causes hydrophobic mismatch. When the drug concentration exceeds its solubility limit within the acyl chain region, phase separation occurs. The drug is subsequently expelled from the membrane during storage. Next Step: Re-evaluate cholesterol ratios and acyl chain packing; consider complementary co-solvents.
3. External-Medium Colloidal Instability
Highly charged payloads (e.g., nucleic acids) can neutralize surface charges. If the overall Zeta potential approaches the isoelectric point (between -10 mV and +10 mV), repulsive electrostatic forces collapse, leading to van der Waals-driven aggregation. Furthermore, abrupt pH shifts during buffer exchange or high ionic strength environments (like serum) can instantly induce flocculation. Next Step: Optimize PEG lipid density for steric stabilization.
🚨 Typical Misinterpretations: Are you measuring aggregation or precipitation?
A sudden increase in size via Dynamic Light Scattering (DLS) does not strictly mean liposome aggregation. Turbidity or high PDI might actually reflect the crystallization of unencapsulated free drug in the bulk medium. Similarly, excessive centrifugation during purification can induce apparent instability that doesn't exist in the unperturbed formulation.
Release Kinetics: Troubleshooting "Too Fast" vs. "Too Slow"
When Release is Too Fast (Burst Release)
- Membrane too fluid: The $T_m$ of the formulation is too low relative to the assay or physiological temperature.
- Incomplete intraliposomal trapping: The drug gradient is weak or easily reversible.
- Purification damage: High shear stress during downstream processing physically compromised membrane integrity.
- PEG dilemma: Certain PEG-lipid architectures can increase membrane permeability if not anchored correctly.
When Release is Too Slow (Over-Retention)
- Overly rigid bilayer: Excessive cholesterol dampens fluidity, essentially "locking" the drug inside.
- High-Tm systems without a trigger: Using lipids like DSPC without an active release mechanism (e.g., pH sensitivity, enzyme cleavage).
- Precipitate too stable: The intraliposomal drug crystal is so insoluble it cannot dissolve even when the membrane permeabilizes.
- Assay artifacts: The in vitro release setup lacks biorelevant components (like sink conditions or serum proteins).
The Crucial Role of Membrane Rigidity
"Published data show that liposomal membrane composition strongly reshapes payload retention and release behavior. In DMPC-based protein liposomes, adding an appropriate level of cholesterol improved encapsulation-associated retention and markedly slowed premature release, whereas overly fluid membranes released cargo much faster. This makes membrane rigidity one of the first variables to check when troubleshooting low retention or burst release."
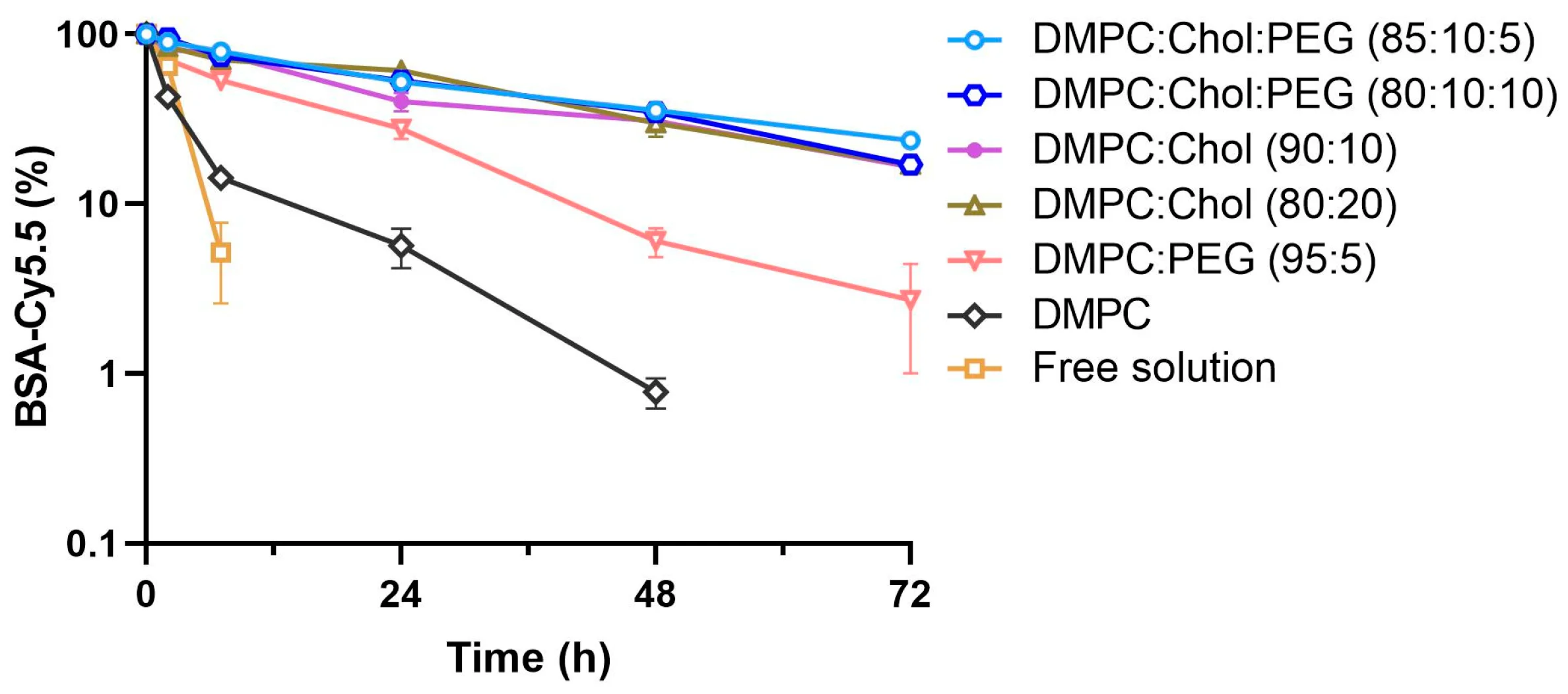
Cholesterol acts as a bidirectional fluidity buffer. Therefore, fine-tuning the sterol content is frequently one of the most influential variables when modulating in vitro release kinetics analysis. Be highly cautious of Assay Artifacts: ensure that your release method (e.g., dialysis bags) does not suffer from membrane binding or violate sink conditions, and remember that serum-free release tests are often not predictive of actual in vivo behavior.
Diagnostic Matrix: Orthogonal Troubleshooting
Isolating the root cause requires separating formulation failures from analytical artifacts. Use this framework to plan your next adjustment.
| Failure Symptom | Likely Formulation Cause | Likely Analytical Cause | First Check (Measurement) | Likely Next Step |
|---|---|---|---|---|
| Post-purification EE < 15% | Weak gradient; poor lipid-drug partitioning. | Incomplete lysis prior to total drug assay; purification-induced shear. | Compare pre-purification vs. post-purification mass balance. | Change gradient salt; validate total lipid lysis protocol. |
| Sudden Turbidity / High PDI | Zeta potential neutralization; hydrophobic mismatch and expulsion. | Free drug crystallizing in buffer; DLS multi-scattering artifacts. | Examine morphology via Cryo-TEM instead of relying only on DLS. | Adjust pH away from isoelectric point; optimize PEG density. |
| Burst Release > 40% at 1h | Membrane $T_m$ too low; residual solvent destabilization. | Dialysis membrane binding; failure to maintain true sink conditions. | Differential Scanning Calorimetry (DSC) to confirm actual $T_m$. | Increase cholesterol mol%; switch to longer saturated acyl chains. |
The Bench-to-Scale-up Risk Shift
For CMC managers pushing liposomal payloads from the bench to scalable GMP production, new failure modes emerge that rarely appear in 5 mL R&D batches. Even if benchtop loading looked perfect, scale-up introduces physical stressors that alter the biophysical state of the liposome.
Key stressors include: Extended residence times at elevated temperatures during large-volume mixing, shear forces during Tangential Flow Filtration (TFF) used for buffer exchange, and high pressure during sterile filtration. Furthermore, subjecting the formulation to freeze-thaw cycles or lyophilization often disrupts the hydration shell of the lipid headgroups, triggering massive payload leakage unless properly cryoprotected.
To truly bridge this translational gap, understanding how lipid degradation and payload leakage occur under physiological sinks is critical. Rigorous evaluation via advanced PK/PD analysis ultimately validates whether the targeted retention criteria are successfully met after the formulation scales up and enters complex in vivo matrices.
Frequently Asked Questions
This usually indicates that the drug is only loosely associated with the outer membrane rather than stably trapped in the core, or that the purification process itself (e.g., spin columns or harsh TFF shear) is physically disrupting the lipid bilayer. It can also point to a weak transmembrane gradient that dissipates rapidly once the external free drug is removed, shifting the equilibrium and causing the payload to leak out.
Relying solely on DLS size increases is misleading. The fastest way to distinguish them is through visual inspection using Cryo-TEM, which clearly shows whether you have fused lipid vesicles (aggregation) or distinct electron-dense crystalline structures outside the liposomes (precipitation). Additionally, if adding a small amount of a specific drug solubilizer clears the turbidity without lysing the liposomes, free drug precipitation is the likely culprit.
Perform a mass balance check. If using dialysis, measure the drug remaining inside the dialysis bag *and* the drug in the bulk sink media. If the total drug recovered is significantly less than what you started with, your drug is likely binding to the dialysis membrane, creating an artificial release profile. Also, ensure your sink volume is large enough; saturation of the release media causes apparent (but false) plateauing of release.
Mixing dynamics and residence times are the biggest culprits. At the bench, heating and cooling are almost instantaneous. At scale, extended time at elevated temperatures can degrade sensitive payloads or cause gradient collapse. Tangential Flow Filtration (TFF) introduces continuous mechanical shear and pumping pressure that can literally squeeze the drug out of previously stable vesicles, which doesn't happen during gentle benchtop dialysis.
Absolutely. Creative Biolabs provides comprehensive CMC support that evaluates both the formulation matrix and the analytical methods used to characterize it. We help you establish robust separation methods, validate complete liposome lysis for accurate EE determination, and design biorelevant release assays that actually predict in vivo performance, moving your project safely through pre-clinical bottlenecks.
References
- Ponkshe, Pranav, Yingzhe Wang, and Chalet Tan. "Systemic protein delivery via inhalable liposomes: Formulation and pharmacokinetics." Pharmaceutics 15.7 (2023): 1951. https://doi.org/10.3390/pharmaceutics15071951
- Under Open Access license CC BY 4.0, without modification.
Online Inquiry
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.


