How Adjuvant Liposome Composition Shapes the
Immune Activation Window and Safety Profile
Learn how phospholipid type, sterol content, charge, and immunostimulatory components influence the immune activation window, reactogenicity risk, and development readiness of adjuvant liposome systems.
Why Composition Is a Strategic Variable in Adjuvant Liposome Design
For adjuvant liposome developers, composition is not just a formulation detail. The choice of phospholipids, sterols, charge-inducing lipids, and immunostimulatory components can directly influence immune intensity, tolerability, formulation stability, and development feasibility.
The real challenge is not maximizing stimulation, but defining a composition that keeps immune activation within a useful and safe operating window. A formulation must robustly break tolerance for purified subunit antigens without inducing overwhelming systemic inflammation.
This is a recurring challenge for formulation scientists, immunology leads, and CMC teams. A composition that improves antigen-presenting cell (APC) uptake may also increase local reactogenicity. A bilayer designed for stronger retention may improve tolerability but reduce release efficiency or complicate downstream processing. Early development therefore requires a framework for balancing potency, safety, stability, and scalability rather than optimizing any single parameter in isolation.
How Composition Shapes Liposome Physicochemical Properties
The fundamental biophysical behavior of a liposomal adjuvant—such as membrane fluidity, robustness, and release kinetics—is dictated by its lipid backbone. The core selection often begins with evaluating the phase transition temperature (Tm) of the primary phospholipids.
High-Tm lipids (like DSPC or DPPC) generally form more rigid bilayers at physiological temperature, which may improve particle integrity and prolong local retention of formulation components. Depending on the overall formulation and administration context, this can contribute to more sustained immune stimulation. Conversely, low-Tm unsaturated lipids (like DOPC) typically increase bilayer fluidity, which may alter membrane interaction, cargo release behavior, and cellular processing kinetics.
This is why incorporating Structure and Composition Analysis early in the R&D workflow is critical. Furthermore, cholesterol serves as a vital fluidity buffer. It prevents the crystallization of highly saturated lipids below their Tm while restricting excessive permeability in unsaturated systems, ultimately fine-tuning how the liposome behaves in the complex biological milieu.
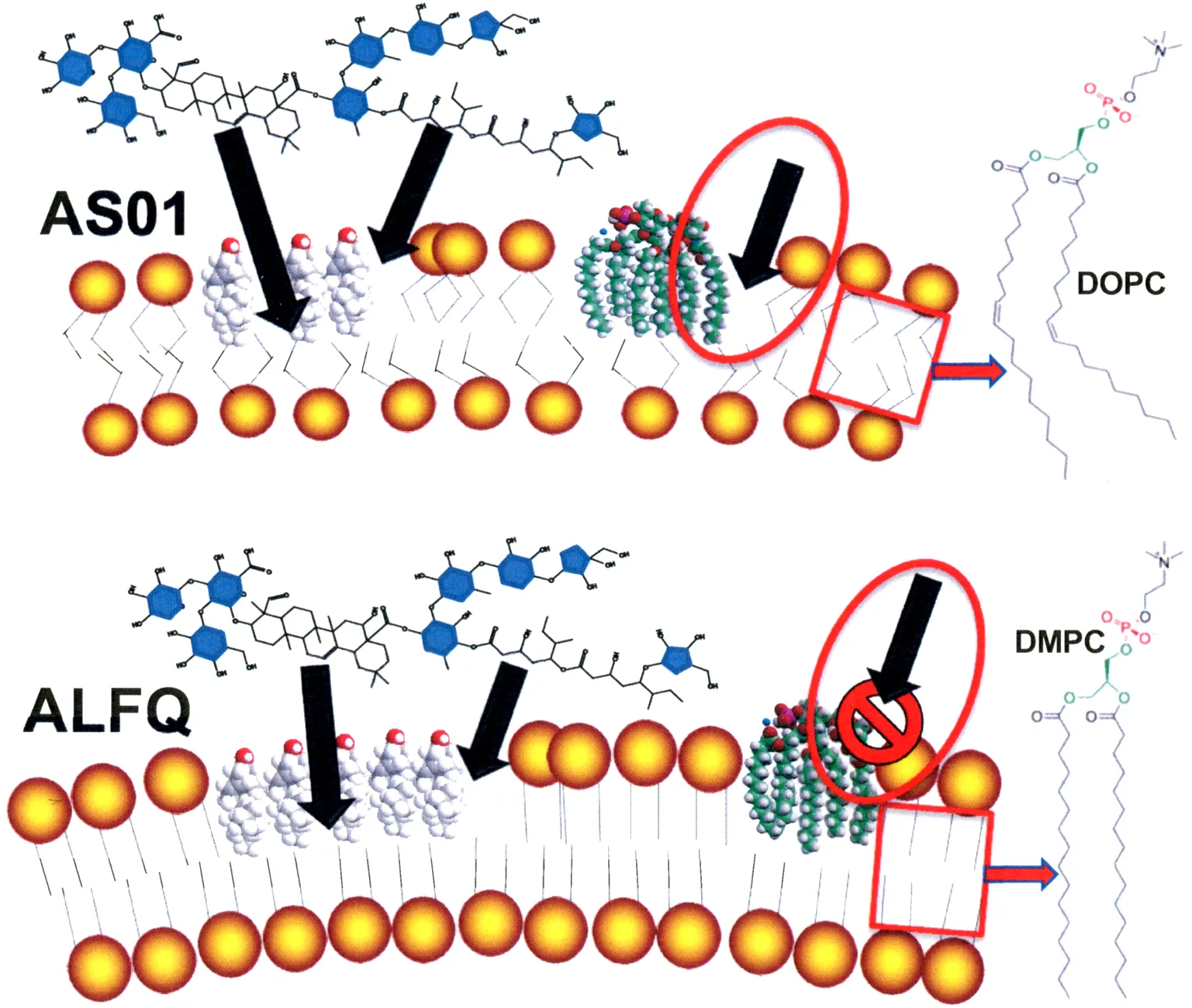
Fig.1 Theoretical comparative vulnerabilities of AS01 and ALFQ to toxic effects of MPLA and to attack by PLA2. 1,2
Published comparisons of representative adjuvant liposome systems suggest that membrane composition may influence both adjuvant exposure and susceptibility to enzymatic degradation, two factors closely linked to the safety window of the formulation. Modifying the lipid ratios changes the structural accessibility of immunostimulants to surrounding cells and endogenous enzymes.
Key Takeaway
Lipid composition does not simply define particle architecture. It also governs how long immune stimuli are retained, how rapidly components are exposed or degraded in vivo, and how safely the formulation can operate within a useful immune activation window.
How These Changes Translate into the Immune Activation Window
A successful adjuvant liposome formulation aims for a precise therapeutic range. It must provoke sufficient local signaling and APC recruitment without triggering widespread systemic cytokine release. Here is how composition translates to immunological reality:
Targeted APC Uptake
Surface charge directly impacts how macrophages and dendritic cells recognize the liposome. Cationic charge density enhances electrostatic binding to cell membranes, significantly increasing cellular uptake. However, overly intense cationic profiles may reduce tolerability ceilings.
Th1 vs. Th2 Skewing
The selection of specific immunostimulatory lipids (like TLR4 or TLR7/8 agonists) alongside the lipid backbone directs the immune bias. A rational composition can facilitate robust cross-presentation, driving CD8+ T-cell and Th1-biased responses critical for oncology and specific infectious targets.
Kinetics & Exposure
Membrane rigidity influences retention versus release. A composition that retains its cargo efficiently dictates whether the host experiences a prolonged, localized immune education at the draining lymph node, or a rapid pulse that could carry a systemic spillover risk.
How Composition Also Shapes the Safety Profile
Beyond immune potency, the lipid matrix governs the formulation's reactogenicity. Formulations with excessive cationic charge might disrupt host cell membranes excessively, potentially shifting the local environment towards necrosis rather than controlled apoptosis.
One often underappreciated factor is the susceptibility of the lipid membrane to endogenous degradation pathways, including phospholipase-mediated hydrolysis (e.g., PLA2 attack). If the bilayer is destabilized too rapidly in vivo, highly potent adjuvant components may be exposed or released faster than intended, potentially narrowing the tolerability margin and leading to systemic reactogenicity.
Composition strategies such as sterol incorporation, tighter lipid packing, or use of structurally robust lipid species may help moderate this risk, although the final effect remains formulation-dependent. Evaluating these parameters via rigorous Formulation Safety Evaluation (such as ex vivo human PBMC assays) is essential before advancing to preclinical safety models.
Reactogenicity Risk Factors
- ● Burst Release: Rapid membrane lysis leading to uncontrolled systemic TLR agonist exposure.
- ● Enzymatic Degradation: Unchecked PLA2 hydrolysis exposing internal payloads prematurely.
- ● Off-Target Exposure: Instability leading to inappropriate organ accumulation and potential systemic cytokine storm.
A Practical Framework for Balancing the Window
In practice, the goal is not to identify a universally "best" adjuvant liposome composition, but to define a formulation space that is appropriate for the antigen, immune objective, administration route, and development path.
Teams often need to weigh at least four variables simultaneously:
- Potency: to ensure adequate antigen priming and immune memory generation.
- Tolerability: to avoid excessive local site reactions or systemic inflammation.
- Stability: to preserve particle integrity, size distribution, and component activity over time.
- Manufacturability: to support reproducible scale-up, sizing processes, and downstream storage strategies.
A rational screening strategy should therefore compare lipid compositions not only by immunogenicity readouts, but also by degradation behavior, release tendency, colloidal stability, and safety-related in vitro signals.
Practical Considerations for Early Screening and CMC Feasibility
A composition that performs well in an exploratory in vivo study is not automatically development-ready. The same lipid choices that affect immune behavior can also influence storage stability, oxidation sensitivity, process robustness, lyophilization compatibility, and batch reproducibility.
| Lipid Component Type | Typical Examples | Primary Function | Impact on Immune Window & Development |
|---|---|---|---|
| Neutral Phospholipids | DOPC, DSPC, DPPC | Forms primary structural matrix; dictates baseline phase transition (Tm). | High Tm improves physical retention of payload, lowering burst release risk, but may require higher processing temperatures during extrusion. |
| Sterols | Cholesterol | Acts as a fluidity buffer and packs lipid tails. | Enhances degradation resistance (e.g., against PLA2), potentially shifting the safety profile favorably, and improves shelf-life stability. |
| Cationic Lipids | DOTAP, DDA, DC-Chol | Facilitates electrostatic interaction with target cells. | Strongly enhances APC uptake and Th1 priming, but high ratios can elevate local reactogenicity and require careful sizing control. |
| Immunostimulatory Lipids | MPLA, α-GalCer | Directly agonizes pattern recognition receptors. | Sets the amplitude of the immune response. Must be stably anchored within the bilayer to prevent systemic circulation side effects. |
For this reason, composition screening should be linked to downstream CMC expectations from an early stage. Teams must leverage continuous Formulation Stability Monitoring Service to track particle size drift, zeta potential shifts, and lipid oxidation. While off-the-shelf Liposomal Vaccine or Adjuvant products can accelerate early screening, bespoke adjustments are often required for final translational success.
In adjuvant liposome development, composition is not a downstream optimization detail.
It is the upstream design variable that defines how strongly the system activates immunity, how safely it is tolerated, and how realistically it can advance toward scalable development. A composition strategy that integrates immunology, biophysics, and CMC thinking from the start is often the most effective way to shorten iteration cycles and reduce translational risk.
Frequently Asked Questions
Screening should begin during the lead formulation selection phase. Evaluating in vitro cytotoxicity and ex vivo cytokine release early prevents costly failures in preclinical animal models caused by excessively reactogenic lipid ratios.
Phospholipid saturation and cholesterol content are critical. Highly saturated lipids may boost in vivo retention and potency by forming rigid bilayers, but they can complicate extrusion processes and require significantly higher operating temperatures during manufacturing.
Rigid bilayers (high-Tm lipids) are typically prioritized when a sustained "depot" effect and prolonged local antigen presentation are desired. Fluid bilayers (low-Tm lipids) might be considered when rapid cellular uptake or specific endosomal processing is required, though they demand stricter stability monitoring to prevent burst release.
Researchers can utilize specialized in vitro enzymatic degradation assays, specifically testing the formulation against relevant endogenous phospholipases (like PLA2) found in interstitial fluid, to predict the release kinetics and potential systemic exposure risks accurately.
Beyond standard immunogenicity endpoints (e.g., antibody titers, T-cell ELISPOT), development teams should actively monitor local injection site reactogenicity, systemic inflammatory cytokine profiles, and antigen-presenting cell (APC) activation markers in the draining lymph nodes to ensure the formulation remains within the optimal safety/efficacy window.
Reference
- Alving, Carl R., Mangala Rao, and Gary R. Matyas. "Similarities and differences of chemical compositions and physical and functional properties of adjuvant system 01 and army liposome formulation with QS21." Frontiers in immunology 14 (2023): 1102524. https://doi.org/10.3389/fimmu.2023.1102524
- Under Open Access license CC BY 4.0, without modification.
Online Inquiry
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.


