Charged Liposome Development Decision Tree:
Cationic vs Neutral vs Anionic
A practical selection guide for formulation scientists and translational teams to balance payload encapsulation, biological barriers, and scale-up feasibility in liposome design.
How Surface Charge Shapes Charged Liposome Development
Surface charge is one of the most important variables in charged liposome development. Whether a team is designing liposomes for nucleic acids, small molecules, vaccines, or diagnostic payloads, selecting a cationic, neutral, or anionic formulation can significantly influence encapsulation strategy, biological interactions, and translational feasibility.
In the past, liposome charge selection was often driven mainly by in vitro uptake or transfection performance. However, modern charged liposome development requires a more practical and translational perspective. In addition to delivery efficiency, formulation teams must evaluate colloidal stability, tolerability, in vivo circulation behavior, and manufacturing constraints.
After entering biological fluids, liposomes rapidly interact with serum proteins and form a protein corona, which can alter their biological identity and downstream performance. Studies have also shown clear differences in stability across charge profiles. As illustrated in Figure 1, cationic formulations are more likely to undergo size expansion or aggregation, while neutral and anionic liposomes often provide better long-term colloidal stability.
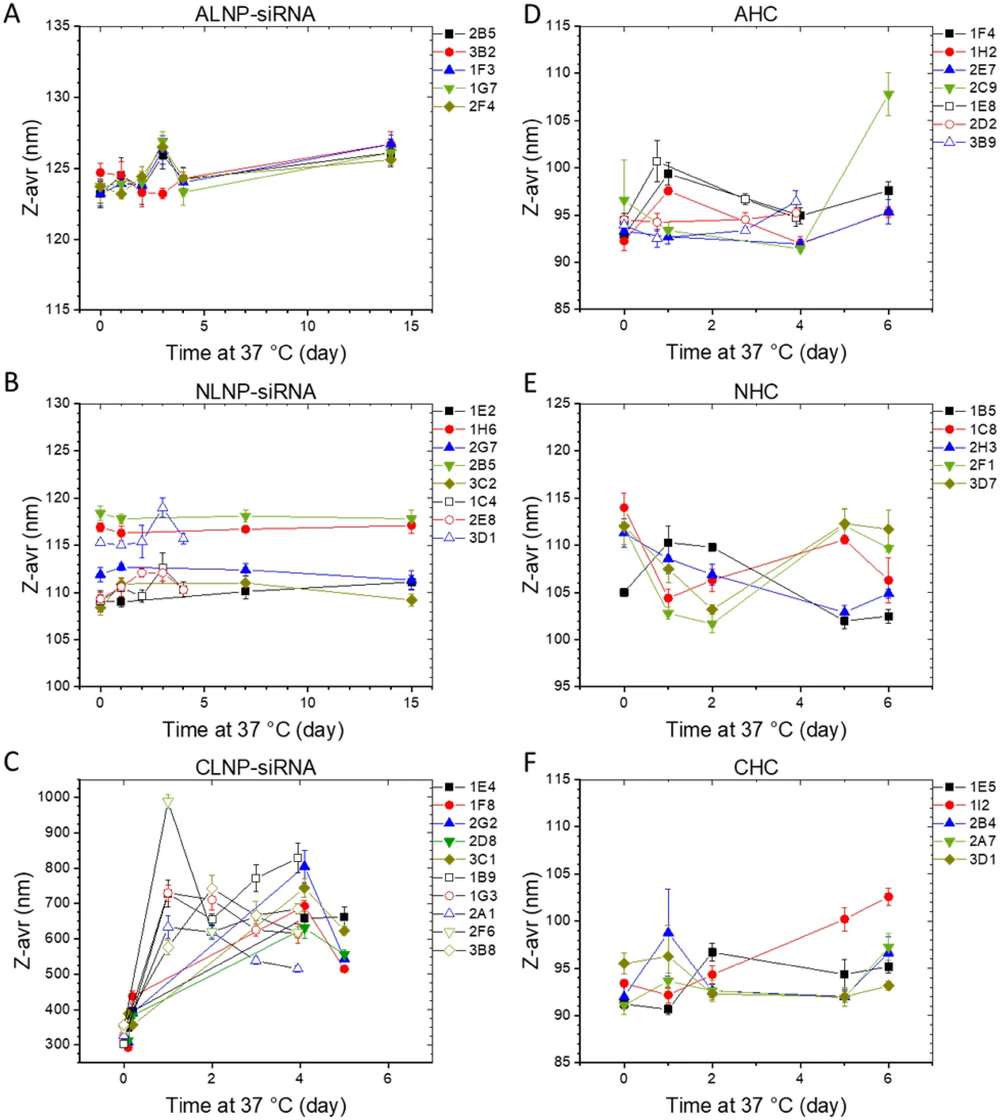
Key Decision Factors in Charged Liposome Formulation
Before choosing a cationic vs neutral vs anionic liposome selection, professional formulation scientists must weigh multiple translational variables. Charge dictates far more than just cellular uptake; it influences everything from payload loading mechanics to scale-up feasibility.
1. Payload Class & Loading Mechanism
Is your payload localized in the aqueous core (hydrophilic), inserted in the lipid bilayer (lipophilic), or electrostatically complexed on the surface (nucleic acids)? Nucleic acids typically necessitate cationic or ionizable lipids for efficient complexation and encapsulation, whereas small molecules can be actively loaded into neutral or anionic systems via transmembrane gradients.
2. Route-Specific Biological Barriers
Intravenous (IV) systemic delivery demands stealth properties (neutral/PEGylated) to evade the reticuloendothelial system (RES) and serum opsonins. Conversely, inhalation routes require stability against nebulization shear, where strong electrostatic repulsion (anionic) can be beneficial. Intramuscular (IM) or topical routes face completely different tissue matrices.
3. Desired Uptake & Release Profile
Does the therapy require rapid intracellular delivery (favoring cationic charge for membrane interaction and endosomal escape) or a slow, sustained extracellular release profile in the systemic circulation (favoring stable neutral formulations)?
4. Safety and Tolerability
Highly positive surface charges can cause cellular membrane thinning, generate reactive oxygen species (ROS), and induce systemic toxicity or rapid pulmonary clearance. Balancing the required efficacy with the acceptable in vivo safety threshold is a paramount consideration.
5. CMC & Manufacturability
The CMC strategy is heavily impacted by liposome charge optimization. Considerations include the availability of cGMP-grade functional lipids, controlling the Polydispersity Index (PDI) during microfluidic mixing, sterile filtration feasibility (0.22 µm membranes may bind charged particles), and adaptability for lyophilization.
How to Use This Charged Liposome Development Decision Tree
Navigating the complexities of liposome design requires a structured approach. Follow these consecutive steps to narrow down the optimal liposome surface charge selection for your project.
Start with Your Payload
• If the payload is nucleic acids (mRNA, siRNA, pDNA) and strong complexation is required → evaluate cationic or ionizable systems first.
• If the payload is small molecules and long systemic circulation is the priority → start from neutral liposomes.
• If the goal is macrophage engagement, topical tolerance, or selected pulmonary use cases → assess anionic liposomes.
Check the Route of Administration
• IV Systemic Delivery: Demands stealth. Strongly cationic particles will aggregate and clear rapidly; neutral or heavily PEGylated systems are preferred.
• Local Injection (IM/SC): Tolerates a wider range of charges, depending on whether depot effect or rapid draining to lymph nodes is desired.
• Ex Vivo / In Vitro: Cationic systems excel here due to lack of serum interference.
Evaluate Primary Trade-offs
No formulation is perfect. You must balance Uptake vs. Toxicity (cationic charge increases both), Circulation vs. Release (highly stable neutral systems may fail to release payload intracellularly), and Manufacturability vs. Performance (complex multi-lipid systems are harder to scale up).
Strategic Decision Tree Matrix for Liposome Charge Optimization
| Formulation Parameter | Cationic (Positive) | Neutral (Zwitterionic) | Anionic (Negative) |
|---|---|---|---|
| Optimal Payload | mRNA, siRNA, pDNA, Proteins | Small Molecules (Chemo), Peptides | Small Molecules, Specific Antigens |
| Cellular Uptake Rate | Very High (Electrostatic) | Moderate to Low | Moderate (Receptor-mediated) |
| Systemic Circulation | Short (Rapid Clearance) | Long (Stealth) | Short to Moderate |
| Cytotoxicity Risk | High | Very Low | Low |
| Colloidal Stability | Prone to Aggregation | High (Especially with PEG) | Very High (Electrostatic Repulsion) |
| Primary Indication | Gene Editing, Vaccines | Solid Tumors (IV administration) | Macrophage Targeting, Pulmonary |
When to Choose Cationic Liposomes in Charged Liposome Development
Formulations utilizing positively charged lipids like DOTAP, DOTMA, or DC-Cholesterol, alongside ionizable lipid-based systems, remain widely used for nucleic acid delivery because they support electrostatic complexation and intracellular delivery. Their primary advantage is two-fold: first, the positive charge condenses negatively charged nucleic acids into stable lipoplexes. Second, the cationic surface strongly interacts with negatively charged proteoglycans on the cell membrane, driving robust cellular uptake.
Furthermore, these systems may promote endosomal escape through pH-responsive membrane destabilization, fusion-related effects, or other endosomal escape mechanisms, ensuring the payload reaches the cytosol intact. However, this high efficacy must be weighed against biocompatibility. Permanent cationic charges can disrupt cellular membranes, generate reactive oxygen species (ROS), and induce cytotoxicity.
In vivo, highly cationic liposomes rapidly bind to serum proteins, leading to particle aggregation, rapid clearance by the reticuloendothelial system (RES), and potential accumulation in the lungs (pulmonary first-pass effect). Therefore, precise cationic liposome development often requires transitioning to ionizable lipids or applying careful PEGylation to shield the charge during systemic circulation.
Explore Cationic Liposome ProductsKey Use Cases for Cationic Formulations
- ✓ Nucleic Acid Therapeutics: Strong foundation for mRNA vaccines, siRNA gene silencing, and plasmid DNA transfection.
- ✓ In Vitro / Ex Vivo Research: Exceptional transfection efficiency in cell culture environments lacking complex serum interference.
- ✓ Adjuvanted Vaccines: The inherent immunostimulatory nature of specific cationic lipids can serve as an adjuvant for protein antigens.
Strengths of Neutral Formulations
- ✓ Extended Half-Life: Avoids rapid opsonization, permitting prolonged systemic exposure.
- ✓ High Biocompatibility: Minimal disruption to cell membranes, resulting in exceptionally low in vivo toxicity.
- ✓ Passive Targeting: Can leverage passive accumulation strategies in inflamed tissues or specific solid tumors.
When Neutral Liposomes Are Preferred for Systemic Delivery
Neutral or near-neutral liposomes are often preferred for applications where circulation stability and tolerability are prioritized. Composed predominantly of zwitterionic lipids (such as DSPC, DPPC, or DOPC) and stabilizing agents like cholesterol, they serve as the established foundation for the systemic delivery of small-molecule chemotherapeutics (e.g., liposomal doxorubicin). Because their net surface charge is close to zero, they bypass the intense electrostatic interactions with serum proteins that destabilize cationic variants.
This minimal charge significantly mitigates protein corona formation and reduces recognition by the mononuclear phagocyte system (MPS). As a result, neutral liposomes exhibit vastly superior circulation half-lives in vivo. This "stealth" characteristic may support prolonged circulation and passive accumulation in some tumor models, although the translational relevance of the EPR (Enhanced Permeability and Retention) effect can vary substantially by indication and patient context.
While their cellular uptake is generally slower—relying primarily on non-specific endocytosis rather than active electrostatic binding—their exceptional biocompatibility and formulation stability make them indispensable when limiting systemic toxicity is the primary goal. Combining these cores with PEGylated lipids further enhances steric stabilization against aggregation.
When Anionic Liposomes Support Stability or Macrophage Targeting
While historically overshadowed by other charge profiles, anionic liposomes—incorporating negatively charged lipids such as DSPG, DMPG, or phosphatidylserine (PS)—offer distinct advantages for specialized translational applications. Because the biological environment is generally net-negative, anionic liposomes rely on different biological mechanisms for cellular interaction rather than basic electrostatic adhesion.
Targeted Engagement
Certain anionic formulations may show enhanced interactions with macrophages or scavenger-receptor-associated uptake pathways, depending on composition and biological context. This makes them useful for anti-leishmanial drugs or therapies treating intracellular RES infections.
Superior Stability
The negative surface charge generates strong electrostatic repulsion between particles, preventing aggregation and fusion. As noted in stability studies, this grants them exceptional colloidal stability over long storage periods, simplifying shelf-life strategies.
Topical & Pulmonary Delivery
In aerosolized applications, strong repulsive forces help anionic liposomes maintain integrity during the high shear stress of nebulization. They are also favored in dermal applications due to an established safety profile and low irritancy on the stratum corneum.
To tailor a formulation for specialized uptake or stable aerosol delivery, explore our anionic liposome development services.
Choosing the Right Charge Profile for Translational Success
Successful liposome engineering is not simply about maximizing transfection; it is about establishing a functional balance. Choose cationic when payload complexation and intracellular uptake dominate the criteria. Choose neutral when systemic tolerability and prolonged circulation are the ultimate goals. Choose anionic when colloidal stability or specialized macrophage targeting is the priority. Most importantly, validate your selection with route-specific and CMC-specific screening rather than relying on surface charge alone.
Frequently Asked Questions
Surface charge dictates the interaction between liposomes and serum proteins. Highly charged particles (especially cationic) rapidly adsorb opsonins, marking them for quick clearance by the mononuclear phagocyte system (MPS) in the liver and spleen. Neutral liposomes bind fewer proteins, allowing them to circulate longer, which is essential for systemic tolerability.
Cationic lipids interact strongly with negatively charged cell membranes, which can lead to membrane thinning, pore formation, and disruption of cellular integrity. Additionally, once internalized, high concentrations of cationic lipids can interfere with mitochondrial function, induce reactive oxygen species (ROS), and trigger apoptotic pathways.
Anionic liposomes are ideal when exploring macrophage targeting (e.g., for intracellular infections or immunomodulation). They are also excellent choices for topical/dermal delivery due to low irritancy, and for aerosol delivery because their strong electrostatic repulsion provides superior colloidal stability during mechanical nebulization.
Direct transition is extremely difficult and rarely successful. A formulation optimized for in vitro cell culture (which typically lacks complex serum proteins) will rapidly aggregate or be cleared upon intravenous injection. For systemic in vivo delivery, scientists usually switch to ionizable lipids or employ PEGylation to shield the cationic charge during circulation.
PEGylation forms a steric hydration layer around the liposome that effectively "masks" the underlying surface charge. This reduces Zeta potential closer to neutral, inhibits protein corona formation, and prevents particle aggregation, thereby increasing circulation time regardless of the core lipid composition.
References
- Syama, Krishnapriya, et al. "Development of lipid nanoparticles and liposomes reference materials (II): cytotoxic profiles." Scientific Reports 12.1 (2022): 18071. https://doi.org/10.1038/s41598-022-23013-2
- Under Open Access license CC BY 4.0 https://creativecommons.org/licenses/by/4.0/, without modification.
Online Inquiry
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.


