Optimizing Nucleic Acid Liposomal Delivery:
Endosomal Escape and Expression
Move beyond formulation screening with a mechanism-driven optimization strategy for nucleic acid liposomal delivery. Understand where intracellular delivery fails—from endosomal retention and recycling to lysosomal degradation and incomplete functional release—and redesign formulations for stronger, more reproducible expression.
Beyond Cellular Uptake: The Endosomal Escape Barrier in Liposomal Delivery
A persistent development challenge is bridging the gap between cellular uptake and reproducible functional output in nucleic acid liposomal and LNP delivery systems.
Many nucleic acid delivery programs do not fail at encapsulation or cellular uptake—they stall after internalization. Teams often see acceptable uptake of mRNA, siRNA, saRNA, or plasmid formulations, yet protein expression, knockdown efficiency, batch reproducibility, or cell-type transferability remain highly inconsistent.
In these cases, the problem is usually not whether particles enter cells, but where intracellular delivery breaks down: endosomal retention, recycling to the extracellular space, lysosomal trafficking, incomplete cytosolic release, or poor downstream functional utilization. Because only a small fraction of internalized cargo typically reaches the cytosol—and reported escape efficiencies are strongly assay-dependent—improving this step can have an outsized effect on final functional output.
Accelerate Your Project with Expert Development Services
Creative Biolabs provides comprehensive Liposome-encapsulated Nucleic Acid Development Services to help drug developers diagnose and bypass these intracellular roadblocks. By leveraging rationally designed lipid libraries and precise analytical profiling, we ensure your payloads achieve superior cytosolic delivery and consistent functional expression.
Explore Nucleic Acid ServicesWhy High Cellular Uptake Does Not Predict Functional Expression
Total cellular uptake does not distinguish between productive and non-productive internalization. A formulation that heavily loads into a cell may still yield negligible therapeutic benefit if it fails to navigate the complex intracellular sorting machinery.
The Recycling Trap
Not all endosomes mature inward. A portion of internalized particles may be redirected into recycling pathways (recycling endosomes), which transport the intact liposomes back to the plasma membrane for exocytosis. This effectively "spits out" the payload before it has a chance to escape, severely reducing productive cytosolic delivery.
Lysosomal Degradation
If endosomal escape kinetics are slower than endosomal maturation kinetics, the nucleic acid carriers are shuttled into late endosomes and eventually fuse with lysosomes. The highly acidic, nuclease- and lipase-rich environment inside the lysosome rapidly and irreversibly degrades the therapeutic payload.
Cell-Type Variability
Formulations optimized for robust expression in standard in vitro models (e.g., HEK293) often underperform in primary cells or in vivo. Different cell types utilize distinct endocytic routes, possess varying membrane lipid compositions, and exhibit divergent endosomal maturation and lysosomal trafficking speeds.
Process & Batch Drifts
High uptake but variable expression across batches often points to process inconsistencies. Subtle shifts in N/P ratio, mixing flow rates, PEG-lipid desorption kinetics, or internal morphology can alter how the particle disassembles in the endosome, directly impacting the reproducibility of escape.
The Intracellular Journey: Endosomal Entrapment vs. Cytosolic Release
Understanding the exact trajectory of a liposome or LNP after it binds to the cell membrane is essential for formulation optimization. The process initiates with endocytosis—often clathrin-mediated, caveolae-mediated, or macropinocytosis, depending heavily on the target cell type and the particle's physicochemical properties.
However, the true critical bifurcation point occurs within the early endosome. Once inside, liposomal carriers face divergent fates. They can be recycled out of the cell, trapped indefinitely, or pass into the late endosome where the microenvironment becomes increasingly acidic (pH drops to roughly 5.0 - 5.5).
It is within this narrow biological window that the engineered lipids must orchestrate a rapid endosomal escape. Endosomal escape remains a central intracellular bottleneck in many nucleic acid liposomal and LNP delivery systems. Optimization must systematically target this bottleneck to rescue trapped therapeutic payloads.
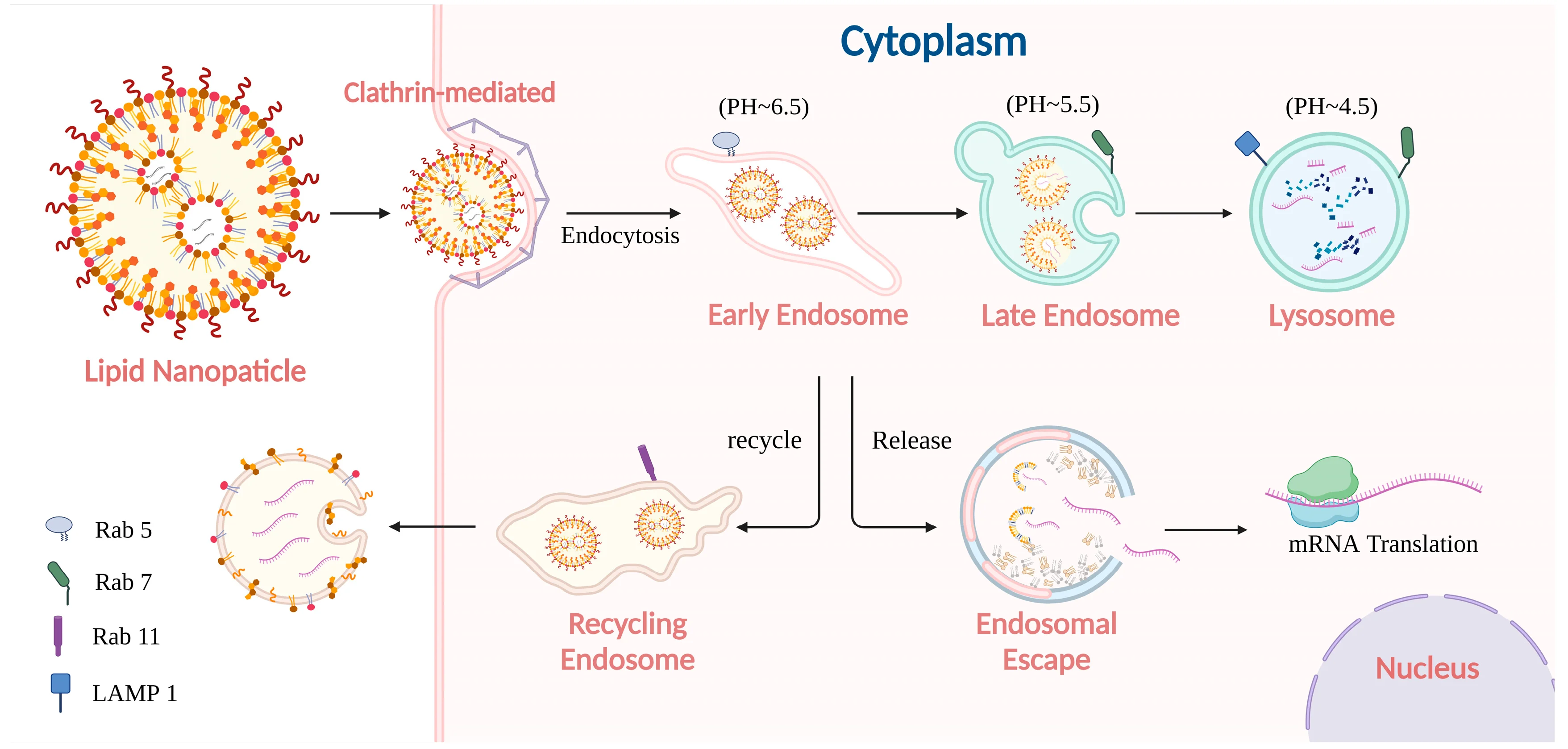
Mechanism-Driven Optimization Workflow for Nucleic Acid Delivery
To resolve low or inconsistent expression, our teams shift from standard matrix screening to a precise, five-step diagnostic and optimization workflow that pinpoints exactly where your delivery fails.
Step 1
Confirm Productive Uptake
Quantify cellular internalization across relevant target cell models using fluorescently labeled carriers, ensuring the baseline problem is not merely a lack of cell-membrane interaction or poor receptor binding.
Step 2
Map Intracellular Trafficking
Utilize confocal microscopy and organelle-specific markers (e.g., Rab5, Rab7, LAMP1) to differentiate early endosomal retention, recycling pathways, late endosome progression, and lysosomal routing.
Step 3
Measure Escape vs. Output
Compare the distinct rates of endosomal escape (cytosolic availability), downstream reporter expression, and knockdown/transfection outcomes to identify if the bottleneck is release, stability, or translation.
Step 4
Re-engineer Formulation Levers
Based on the trafficking data, rationally optimize ionizable lipid pKa, helper lipid ratios, cholesterol analogs, PEG-lipid shedding rates, N/P ratios, and microfluidic mixing parameters.
Step 5
Validate Safety & Reproducibility
Reassess cellular viability, cytotoxicity, and inflammatory responses in parallel with functional efficacy, ensuring the optimized formulation maintains batch-to-batch consistency before scale-up.
Formulation Optimization Levers: Enhancing Endosomal Escape Efficiency
| Optimization Lever | Observed Bottleneck / Issue | Mechanism-Driven Strategy |
|---|---|---|
| Ionizable Lipids | Strong internalization with high lysosomal colocalization; failure to protonate in time. | Fine-tune apparent pKa (typically 6.0-6.5). Design lipids with branched tails or optimized linker chemistry to maximize membrane-destabilizing cone shape upon protonation. |
| Helper Lipids (e.g., DOPE) | Cargo gets trapped in highly stable endosomes without membrane fusion. | Incorporate DOPE to promote transition to an inverted hexagonal (HII) phase in acidic environments, mechanically disrupting the endosomal bilayer. |
| PEG-Lipid Dynamics | The "PEG Dilemma": good circulation but heavily sterically hindered membrane fusion. | Optimize acyl-chain length (e.g., C14 PEG-DMG) to control desorption kinetics, shedding PEG to reveal the active fusion surface post-cellular entry. |
| Cholesterol Analogs | Instability during endocytosis or lack of morphological flexibility for release. | Substitute standard cholesterol with specific phytosterols (e.g., $\beta$-sitosterol) to alter membrane fluidity and boost cytosolic release kinetics. |
|
Process Parameters (Mixing, Flow, N/P Ratio) |
Potent in vitro but unstable or variable in repeat batches; structural heterogeneity. | Standardize microfluidic mixing rates to control particle size/PDI. Optimize N/P ratio to balance core packing density with the ability to unpackage in the cytosol. |
Finding the perfect stoichiometric ratio and process parameters is complex. Through our rigorous Liposome Formulation Optimization platform, Creative Biolabs utilizes Design of Experiments (DoE) methodologies to rapidly screen, identify, and validate high-performing formulations tailored to your specific application.
Cargo-Specific LNP Design: mRNA, siRNA, saRNA, and Plasmid Delivery
The journey does not end identically for every payload. Nucleic acid liposomal systems must be tailored because different cargo types face distinctly different post-entry biological barriers and functional utilization pathways.
mRNA
Requires rapid endosomal escape to preserve single-stranded integrity. Optimization must prioritize complete cytosolic release and translation readiness, while minimizing innate immune sensor activation (e.g., TLRs) within the endosome.
siRNA
While structurally more stable, siRNA must be unpackaged and made conformationally available for RISC loading in the cytosol. Optimization emphasizes productive cytosolic availability rather than just total bulk cellular uptake.
saRNA
Self-amplifying RNA is significantly larger and more sensitive to shear stress and degradation. Formulations must balance robust endosomal escape with gentle intracellular unpackaging to preserve its replication and translation competence.
Plasmid DNA
Faces a dual-barrier challenge. Plasmids must not only successfully escape the endosomal compartment but also require subsequent nuclear access barriers to be overcome for functional transcription.
Engineering Ionizable Lipids for Enhanced Membrane Destabilization
The architectural core of any successful nucleic acid delivery vehicle is its cationic or ionizable lipid. Endosomal escape in ionizable lipid systems is generally associated with pH-triggered protonation, ion-pair formation with endosomal membrane lipids, and membrane destabilization that promotes non-bilayer transitions.
Ionizable lipids are meticulously engineered to remain neutral at physiological pH (pH 7.4) while circulating in the bloodstream, preventing non-specific protein adsorption and toxicity. However, as the particle enters early and late endosomes, the environmental pH plummets. The amine headgroups rapidly become protonated, acquiring a strong positive charge.
These protonated lipids form ion pairs with the endogenous anionic phospholipids lining the endosomal membrane. This electrostatic interaction forces a transition from a stable bilayer structure into an inverted hexagonal phase, physically destabilizing the endosomal membrane and spilling the therapeutic cargo into the cytoplasm before the destructive lysosomal stage.
Precision Lipid Design
For this reason, the exact structure of the lipid—including headgroup chemistry, apparent pKa, linker design, tail branching, and unsaturation—strongly influences whether intracellular trafficking ends in productive cytosolic release or lysosomal loss.
Creative Biolabs offers specialized Cationic Liposome Development Services, formulating customized lipid combinations to maximize the critical membrane-destabilizing effects tailored to your specific cell targets and cargo type.
Explore Cationic Liposome ServicesBalancing Endosomal Escape Efficacy with Liposome Formulation Safety
A paramount challenge in translational development is managing the therapeutic index. Formulations engineered to dramatically improve endosomal escape by increasing membrane destabilization may inherently raise cytotoxicity, inflammatory signaling, or off-target cellular membrane effects.
Because the mechanisms that disrupt endosomes can also stress the plasma membrane or mitochondrial pathways, optimization cannot focus on escape efficiency alone. For true translational success, developers should evaluate productive delivery, tolerability, and process reproducibility in parallel rather than sequentially. Our comprehensive Formulation Safety Evaluation allows you to assess parameters like complement activation and cytokine release early, confidently advancing your most robust and safe candidates toward in vivo milestones.
Frequently Asked Questions
High cellular uptake only confirms that particles have crossed the cell membrane into early endosomes. It does not guarantee that the cargo will be utilized. Many highly internalized formulations are subsequently expelled from the cell via recycling endosomes or permanently trapped and degraded in lysosomes. True functional efficiency relies heavily on post-uptake intracellular trafficking and successful cytosolic release, not just initial membrane penetration.
Reference
- Wang, Jiapeng, et al. "Endo/lysosomal-escapable lipid nanoparticle platforms for enhancing mRNA delivery in cancer therapy." Pharmaceutics 17.7 (2025): 803. https://doi.org/10.3390/pharmaceutics17070803
- Under Open Access license CC BY 4.0, without modification.
Online Inquiry
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.


