Directional Antibody Conjugation and
Activity Retention in Immunoliposome Development
Evaluating structural architecture, orientation control, and formulation trade-offs to balance biological activity with translational manufacturability.
Navigating the Complexity of Active Targeting
The translation of lipid-based nanoparticles from passive delivery vehicles into actively targeted nanomedicines fundamentally relies on the precise surface functionalization of ligands. Among these, antibody-functionalized liposomes represent a critical approach for site-specific targeted drug delivery platforms.
However, development teams frequently encounter a practical bottleneck. The central challenge is not simply attaching antibodies to the liposome surface, but selecting a conjugation architecture that preserves target engagement without compromising particle quality or downstream manufacturability. Traditional methods utilizing random primary amine crosslinking often result in unpredictable antibody orientation on liposomes. This arbitrary linkage frequently targets residues near the antigen-binding site (paratope), leading to steric hindrance, structural deformation, and variable loss of target recognition.
To effectively translate these promising therapeutics to clinical models, development workflows must prioritize site-specific antibody conjugation. In many full-length IgG-based designs, directional strategies aim to bias attachment away from the antigen-binding region so that Fab accessibility is retained. Addressing these structural variables early on is an essential aspect of comprehensive immunoliposome formulation optimization.
Core Formulation Objectives
- Preserve Antibody Binding Activity: Minimizing chemical interference near critical antigen-binding domains to maintain in vivo target avidity.
- Colloidal Stability Control: Mitigating cross-linking between multiple liposomes which leads to poor polydispersity and rapid systemic clearance.
- Process Compatibility: Avoiding assembly procedures that expose delicate biological ligands to severe organic solvents or uncontrolled shear stress.
Conjugation Strategy Selection
The foundation of functional immunoliposomes requires selecting an orthogonal bioconjugation strategy. The chosen chemistry directly influences the trade-off between orientation control and process simplicity.
| Conjugation Strategy | Target Reactive Group | Orientation Control | Development Consideration |
|---|---|---|---|
| Random Amine Coupling | Surface Lysines | Poor | Useful for early simple screening only; limited translation control. |
| Thiol-Maleimide (Hinge) | Reduced Hinge Cysteines | Excellent | Highly effective when hinge-accessible thiols can be introduced or exposed mildly. |
| Bioorthogonal Click Chemistry | Engineered Azide/Alkyne | Excellent | Provides strong site control, but strictly requires engineered antibody inputs. |
| Enzymatic Conjugation | Specific Glycans (Fc region) | Very Good | Attractive for Fc-directed workflows, though process complexity and cost are typically higher. |
Balancing Thiol Chemistry and Accessibility
By utilizing mild reducing agents (such as TCEP or DTT), the disulfide bonds in the hinge region of an IgG antibody can often be selectively reduced into free thiols. Because these thiols are positioned physically apart from the antigen-binding domains, reacting them with maleimide-functionalized PEG-lipids forces the antibody to attach near its "waist."
This optimization generally allows the Fab arms to remain unhindered, facilitating bivalent interactions with cell surface receptors and increasing overall avidity. However, developers must balance the reduction conditions to ensure the structural stability of the antibody is maintained while sufficient reactive sites are exposed.
Addressing the PEG Dilemma
The steric environment surrounding the conjugated antibody is a crucial development factor. If the targeting ligand is coupled directly to a lipid headgroup or a very short PEG chain, it may become buried within the surrounding protective PEG corona (typically PEG2000), which impedes target interaction.
To mitigate this "PEG dilemma," targeted liposome development often employs a dual-PEG system. Standard PEG2000 provides baseline shielding, while a smaller molar percentage of longer functionalized chains (e.g., PEG3400 or PEG5000) acts as an anchor. This ensures the targeting ligand protrudes sufficiently beyond the stealth hydration layer, facilitating efficient receptor docking.
Post-Insertion Method for Immunoliposomes
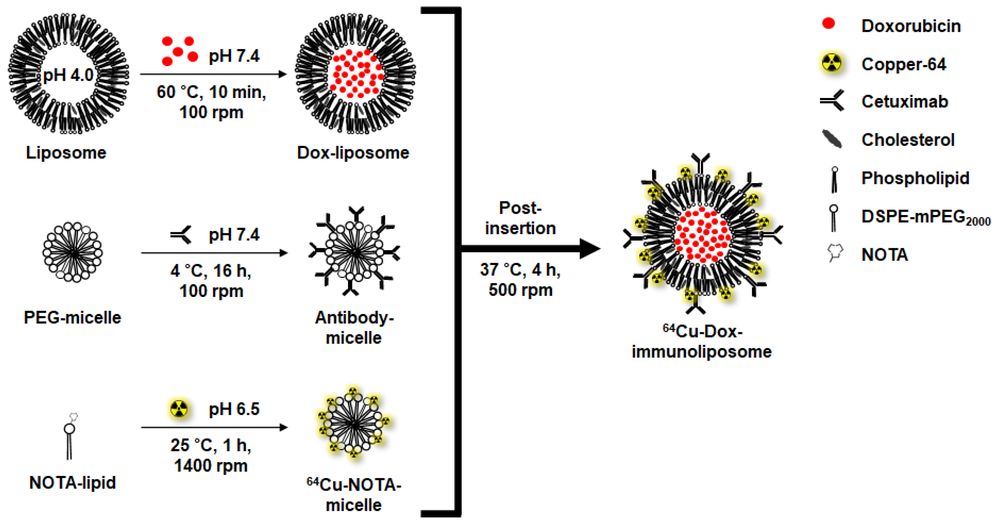
Traditional liposome manufacturing often requires energy-intensive mechanical processes—such as high-pressure extrusion or sonication—to form uniform vesicles. When antibodies are conjugated to lipids prior to vesicle formation (pre-insertion), these energetic processes can lead to protein denaturation and diminished binding efficacy.
One representative application of mild workflows designed to mitigate this risk is theranostic immunoliposome assembly via the post-insertion method. This methodology uncouples the bulk nanoparticle formation steps from the sensitive bioconjugation process.
In this approach, antibody-conjugated PEG-lipid micelles are prepared independently in an aqueous buffer. These functionalized micelles are subsequently co-incubated with preformed, drug-loaded liposomes at physiologically relevant temperatures (e.g., 37°C to 60°C, depending on the lipid phase transition characteristics). The thermodynamic driving force naturally transfers the functional PEG-lipid anchors into the outer leaflet of the preformed vesicles.
This strategy biases ligand presentation toward the external surface, improving the likelihood of outward-facing ligand display. By limiting the biological component's exposure to potentially damaging forces, the resulting immunoliposomes generally retain higher functionality and stability suitable for complex in vivo applications.
Development Considerations Beyond Conjugation Efficiency
Achieving a successful covalent linkage is merely the prerequisite step in formulating targeted delivery vehicles. For translational teams, evaluating the manufacturability of immunoliposomes is equally critical to ensure progression toward clinical models.
Antibody Density and Steric Crowding
Increasing ligand densities does not guarantee enhanced cellular uptake in a linear fashion. Excessive surface functionalization can induce steric crowding, mask active paratopes, and escalate the risk of nanoparticle aggregation. Establishing a controlled, optimized ligand-to-lipid ratio is necessary to balance avidity with colloidal stability.
Impurity Clearance and Purity
Post-conjugation purification strategies must effectively remove unbound free antibodies, residual micelle structures, and unconjugated PEG-lipids. Incomplete clearance of these reaction byproducts can confound in vitro cell binding assays and may act as competitive inhibitors in vivo, complicating pharmacokinetic analyses.
Formulation Integrity Verification
Following conjugation or post-insertion thermal incubation, the nanoparticle's structural integrity must be re-evaluated. Development parameters dictate that the mean size, polydispersity index (PDI), zeta potential, and active payload encapsulation metrics must remain within strict specifications compared to baseline unmodified controls.
Scale-Up Compatibility
Early design iterations should prospectively account for process robustness. A conjugation chemistry or purification method that performs efficiently at bench-scale centrifugation may introduce variance during larger-scale tangential flow filtration (TFF). Selecting scalable routes is a core requirement in translating targeted formulations.
Rigorous Analytical Validation of Conjugates
To advance a directional conjugation strategy into a viable translational asset, empirical validation of both physical properties and biological activity is required. Comprehensive analytical methods confirm that the targeting moiety remains functional and properly oriented.
Physicochemical Characterization
Modifying the lipid bilayer with large biological ligands can alter foundational metrics. Researchers monitor changes in particle size (Dynamic Light Scattering), polydispersity index, and surface charge to ensure the system remains monodisperse and free of antibody-induced aggregation.
Review Structure Analysis MethodsBiological Binding Activity
Proving that the paratopes are functional requires comprehensive in vitro assessment. Techniques such as Surface Plasmon Resonance (SPR) quantify the binding kinetics of the conjugated liposome to immobilized antigens. Cellular uptake assays verify ex vivo receptor-mediated interactions.
View Basic Characterization ServicesFor comprehensive support in translating your targeting strategy, Creative Biolabs offers specialized development capabilities.
Antibody-Coupled Liposome ServicesFrequently Asked Questions
Pre-insertion involves conjugating antibodies directly to lipid mixtures before vesicle formation, which may expose the antibodies to high shear forces, organic solvents, or extreme pH during extrusion and sizing. The post-insertion method separates these harsh processing steps from the biological components. It is generally preferable when working with fragile ligands that are susceptible to denaturation, as it relies on milder thermal incubation to insert functionalized PEG-lipid micelles into preformed, sized vesicles.
The choice primarily depends on the desired circulation profile and target accessibility. Full-length IgG molecules offer bivalent binding but contain an Fc region that can trigger rapid clearance by the reticuloendothelial system via Fc receptor-mediated phagocytosis. Antibody fragments like Fab' or scFv mitigate this Fc-mediated clearance and reduce the overall hydrodynamic diameter of the nanoparticle, potentially enhancing tissue penetration. However, fragments may exhibit monovalent binding depending on the selected engineering strategy.
Orientation dictates the effective binding avidity of the formulation. Random conjugation often results in a variable fraction of antibodies binding to the lipid surface in ways that sterically hinder their antigen-binding sites. Directional strategies, such as hinge-region thiol conjugation, aim to consistently orient the Fab domains outward. This structural control helps maintain biological activity and ensures batch-to-batch reproducibility, which is a prerequisite for clinical translation.
Polyethylene glycol (PEG) is routinely used to provide steric stabilization and prolong circulation half-life. However, a dense PEG corona can physically shield targeting ligands from interacting with cellular receptors—a functional trade-off known as the PEG dilemma. Formulation optimization typically addresses this by employing a dual-PEG architecture, conjugating the targeting antibody to a PEG chain slightly longer than the surrounding protective PEG layer to preserve binding accessibility.
While higher ligand densities might intuitively seem advantageous for targeting, overloading the liposome surface routinely compromises manufacturability. High antibody densities can provoke inter-particle crosslinking, leading to aggregation, unacceptable polydispersity indices (PDI), and complicated downstream purification. Establishing an optimal ligand-to-lipid ratio involves balancing targeting efficiency with essential colloidal stability.
References
- Jeong, Hwa Yeon, et al. "Development of PET radioisotope copper-64-labeled theranostic immunoliposomes for EGFR overexpressing cancer-targeted therapy and imaging." International journal of molecular sciences 25.3 (2024): 1813. https://doi.org/10.3390/ijms25031813
- Under Open Access license CC BY 4.0, without modification.
Online Inquiry
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.


