Anionic Liposomes in Drug Delivery: How Surface Charge Affects Interactions and Cellular Uptake
Negative surface charge changes protein adsorption, influences macrophage recognition, shifts the uptake route, and affects retention vs. clearance. Therefore, it must be carefully optimized during anionic liposome development.
Surface Charge as a Critical Quality Attribute
In the evolving field of nanomedicine, lipid composition dictates not only the physical boundaries of a liposomal carrier but also its immunological and biological fate. While neutral liposomes offer prolonged circulation and cationic liposomes excel at nucleic acid complexation, an effective anionic liposome formulation occupies a unique functional niche. Through precise anionic lipid selection and surface charge optimization, these carriers can achieve targeted cellular interactions while exhibiting reduced cytotoxicity compared to highly cationic systems.
However, navigating the negative charge spectrum presents distinct challenges. For formulation scientists and preclinical drug delivery teams, it is rarely as simple as adding a negatively charged lipid. In practice, successful anionic liposome development depends on balancing negative surface charge against serum stability, macrophage recognition, and the intended uptake route in target cells.
Tuning the charge density correctly is essential to avoid premature drug leakage in plasma, evade unwanted complement activation, and ensure that the payload successfully reaches the intended cytosolic or endosomal target.
Core Challenges in Anionic Liposome Engineering
- ✓ Serum-Mediated Instability: Uncontrolled electrostatic interactions with plasma proteins can cause structural disintegration and premature payload leakage.
- ✓ Macrophage Recognition: Certain negative charges act as robust "eat me" signals, resulting in rapid clearance by the mononuclear phagocyte system (MPS).
- ✓ Cellular Repulsion: Overcoming the electrostatic repulsion between the negatively charged liposome and the similarly charged cellular glycocalyx requires precise zeta potential tuning.
The Protein Corona and Liposomal Integrity
Upon introduction into a biological fluid, liposomes are immediately coated by a dynamic layer of biomolecules known as the protein corona. The serum stability of anionic liposomes is profoundly influenced by the density and presentation of the negative charge.
It is crucial to note that not all anionic liposomes behave the same. Low-to-moderate anionic density may support colloidal stability, while excessive negative charge may intensify unfavorable plasma interactions. Charge density must be screened together with lipid packing and cholesterol content during anionic liposome development.
This figure below shows that increasing anionic lipid content does not simply reduce biointeraction. Instead, a more negative surface can profoundly reshape protein adsorption in plasma and markedly affect liposome integrity. In a pivotal study by Tretiakova et al. (2023), highly anionic POPG-rich liposomes exhibited substantially greater leakage of hydrophilic payloads when exposed to human plasma compared to their neutral or low-charge counterparts.
For development teams, this highlights the need to evaluate leakage not only in buffer, but also in serum-containing media that better reflect biological exposure. If the formulation destabilizes in the serum due to intense protein corona-induced membrane tension, the downstream cellular uptake behavior becomes largely irrelevant.
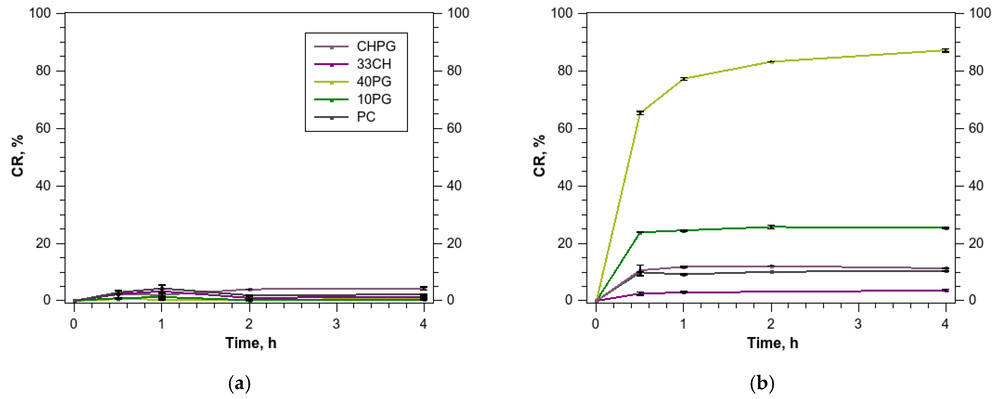
Modulating Cellular Uptake Pathways
Cellular uptake of nanocarriers is a complex interplay of size, shape, elasticity, and predominantly, surface charge. Mammalian cell membranes possess a net negative charge due to the presence of proteoglycans and sialic acid residues in the glycocalyx. Consequently, highly anionic liposomes face inherent electrostatic repulsion when approaching the cell surface.
Despite this repulsion, anionic liposomes can be internalized efficiently in certain cell systems, but uptake is highly dependent on composition, corona formation, and cell phenotype. Anionic liposomes may show reduced nonspecific adsorption to cell membranes compared with cationic systems. Their uptake can depend on lipid composition, protein corona, cell type, and serum conditions; in phagocytic or scavenger receptor-rich cells, receptor-associated uptake may become more prominent.
Understanding the macrophage uptake of anionic liposomes makes them incredibly valuable for interventions targeting the mononuclear phagocyte system, such as macrophage reprogramming in tumor microenvironments or specific immune cell delivery.
Comparative Profile of Liposomal Surface Charges
| Liposome Type | Primary Uptake Mechanism | Serum Stability | Cytotoxicity |
|---|---|---|---|
| Neutral | Slower, fluid-phase endocytosis | High (Long circulation) | Very Low |
| Cationic | Rapid, adsorptive endocytosis | Low (Rapid opsonization) | High |
| Anionic | Composition- and cell type-dependent; may involve scavenger receptor-associated uptake | Variable; strongly dependent on lipid composition, charge density, membrane rigidity, and serum environment | Low to Moderate |
A Practical Development Framework for Anionic Liposome Optimization
Systematic anionic liposome development requires moving beyond simple charge assignment. To achieve predictable delivery, developers should follow a structured evaluation pathway.
Define Therapeutic Objective
Determine whether the goal is long circulation, targeted macrophage interaction, or direct intracellular delivery. The objective dictates the acceptable negative charge threshold.
Select Anionic Lipid Class
Evaluate PG versus PS or mixed lipid systems based on required bio-signaling. Determine the baseline structural lipids needed for support.
Tune Charge Density & Membrane
Adjust the molar percentage of the anionic lipid, incorporate cholesterol, and optimize the ratio of saturated vs. unsaturated hydrocarbon tails to govern membrane rigidity.
Validate in Biological Media
Move beyond PBS. Test serum leakage, monitor zeta shifts post-incubation, profile protein corona formation, and run specific macrophage interaction assays.
Lipid Selection and Formulation Optimization
The biological outcome is rarely determined by charge sign alone; it is shaped by headgroup chemistry, charge density, membrane rigidity, and the biomolecular corona formed under relevant serum conditions. Two of the most utilized lipids in this space are Phosphatidylglycerol (PG) and Phosphatidylserine (PS).
Phosphatidylglycerol (PG)
PG lipids (e.g., DSPG, DMPG, POPG) are vital for maintaining colloidal stability through electrostatic repulsion, preventing particle aggregation during storage. PG-containing formulations may reduce aggregation and, under some compositions, show controlled biointeractions, but they should not be considered functionally equivalent to stealth formulations. As in vitro data suggests, hyper-anionic PG formulations still risk membrane destabilization in plasma.
Explore PG Liposome DevelopmentPhosphatidylserine (PS)
Conversely, PS is heavily implicated in biological signaling. In mammalian cellular biology, surface-exposed PS is widely associated with apoptotic-cell recognition and can promote phagocytic uptake in relevant biological contexts. By integrating PS into liposomal bilayers through specialized PS anionic liposome development services, researchers can intentionally trigger rapid macrophage engulfment for delivering immunomodulators or antigens.
Balancing the hydrocarbon tail saturation (e.g., DOPG vs. DSPG) alongside the anionic headgroup is critical. Saturated tails enhance bilayer packing and reduce plasma leakage, counteracting the destabilizing effects of a highly negative surface corona.
Development Considerations for Anionic Liposomes
When optimizing anionic liposome development, teams should avoid treating negative charge as a binary formulation label. Instead, the key variable is charge density in the context of membrane composition and intended biological use. Increasing the molar fraction of anionic lipids may improve colloidal dispersion in some systems, but it can also intensify serum-driven destabilization, alter protein corona composition, and accelerate macrophage-associated clearance. A practical development strategy is to screen multiple lipid compositions across a controlled zeta potential window, then compare serum leakage, uptake behavior, and target-cell compatibility under biologically relevant conditions.
Advanced Characterization: What to Measure
Theoretical formulation must be validated with robust empirical data. Characterizing the surface charge is not a static measurement; it is an evaluation of dynamic interactions. For successful anionic liposome development, measuring the following checklist of parameters in biologically relevant media is essential.
Frequently Asked Questions
There is no universal "ideal" zeta potential; it depends entirely on the therapeutic objective. Low-to-moderate anionic density (e.g., -10 mV to -30 mV) can often provide sufficient electrostatic repulsion for colloidal stability while minimizing severe plasma protein adsorption. However, highly negative potentials (e.g., beyond -40 mV) may trigger intense protein corona formation, disrupting the lipid bilayer and causing rapid leakage in vivo. Charge density must be systematically optimized during the formulation phase.
Not necessarily. While cationic liposomes typically bind rapidly and non-specifically to negatively charged cell membranes via electrostatic attraction, anionic liposomes may exhibit reduced non-specific adsorption. However, their cellular uptake can still be highly efficient depending on lipid composition, protein corona presentation, and cell type. In cells rich in phagocytic or scavenger receptors (like macrophages), receptor-associated uptake of anionic liposomes can be quite prominent.
Phosphatidylserine (PS) acts as a powerful biological signaling molecule. In normal mammalian biology, surface-exposed PS is a hallmark of apoptotic cells, serving as a widely recognized "eat me" signal. When integrated into liposomes, PS is rapidly recognized by specific receptors on macrophages and other mononuclear phagocytes, accelerating phagocytic clearance. This makes PS highly valuable in targeted immunotherapies but detrimental if long systemic circulation is desired.
Serum-induced leakage, driven by unfavorable protein corona integration, can be mitigated by fine-tuning the membrane's structural rigidity. Formulation scientists can reduce the molar fraction of the anionic lipid to lower the overall charge density. Concurrently, utilizing lipids with saturated hydrocarbon tails (such as DSPC or DSPG) increases the transition temperature, while incorporating optimal amounts of cholesterol helps tighten membrane packing, thereby dramatically enhancing serum stability.
References
- Tretiakova, Daria, et al. "Protein corona of anionic fluid-phase liposomes compromises their integrity rather than uptake by cells." Membranes 13.7 (2023): 681. https://doi.org/10.3390/membranes13070681
- Under Open Access license CC BY 4.0, without modification.
Online Inquiry
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.


