Protein & Peptide Liposomes:
Preventing Denaturation and Achieving Controlled Release
Proteins and peptides are highly vulnerable to solvent exposure, shear stress, freeze-thaw cycles, and pH or temperature shifts during liposome preparation. Learn how formulation and membrane-engineering strategies can reduce denaturation, limit burst release, and improve payload availability at the target site.
Why Proteins and Peptides Are Difficult to Formulate in Liposomes
Proteins and peptides have fundamentally transformed modern medicine, offering high specificity and efficacy. However, translating these macromolecules into viable clinical therapeutics requires scientists to overcome severe biophysical barriers. Unlike small molecules, biologics rely on intricate secondary and tertiary structures to maintain pharmacological activity.
One of the primary formulation bottlenecks for protein liposomes and peptide liposomes is the high risk of conformational instability during processing and storage. The journey from a purified biologic to a fully assembled liposomal formulation is fraught with hazards that can trigger unfolding or aggregation.
Furthermore, formulators face a fundamental paradox: protecting the cargo from the biological environment (enzymes, clearance mechanisms) requires maintaining robust liposome stability. Yet, if the bilayer is over-stabilized, the liposome may reach the target tissue but fail to release the biologic at therapeutic concentrations. Conversely, formulating a highly fluid membrane to encourage release often leads to premature leakage (burst release) in systemic circulation, causing off-target toxicity and sub-therapeutic efficacy. Successfully achieving controlled release liposomes hinges on precisely balancing these competing requirements.
Key Formulation Risks That Affect Biologic Integrity
Interfacial Stress
Traditional liposome preparation methods, such as thin-film hydration or reverse-phase evaporation, frequently expose therapeutic proteins to residual organic solvents (chloroform, ethanol) and high-energy water-lipid interfaces. This exposure disrupts delicate non-covalent internal interactions, leading to irreversible protein denaturation during protein encapsulation in liposomes.
Mechanical Stress
Size reduction processes are essential for creating uniform liposomes, but intense cavitation from probe sonication or high-shear forces generated by high-pressure homogenization can physically tear macromolecular structures, especially fragile, high-molecular-weight proteins.
Thermal and pH Stress
Lipids often require heating above their phase transition temperature (Tm) to hydrate effectively. If this temperature exceeds the unfolding threshold of the co-encapsulated protein, thermal denaturation occurs. Similarly, localized pH shifts during active loading gradients can cause isoelectric precipitation.
Aggregation and Adsorption
Unfolded proteins tend to expose hydrophobic core regions, driving rapid aggregation. Furthermore, strong electrostatic interactions between oppositely charged peptide domains and lipid headgroups can cause the biologic to adsorb onto the membrane surface rather than encapsulate in the aqueous core, altering both liposome stability and release kinetics.
Freeze-Drying (Lyophilization) Damage
To ensure long-term shelf life, liquid formulations are often lyophilized. The freezing step induces ice crystal formation that can puncture lipid bilayers and mechanically stress proteins, while the drying phase removes the essential hydration shell that maintains the protein's native fold.
Strategies to Reduce Denaturation During Liposome Preparation
To reduce formulation-induced instability, developers increasingly favor mild encapsulation workflows, predominantly aqueous and lower-stress processing environments, and formulation screening strategies specifically tailored to protein and peptide sensitivity.
• Advanced Manufacturing Paradigms: Microfluidic mixing has emerged as a gold standard for protein encapsulation in liposomes. By precisely controlling fluid dynamics in laminar flow channels, lipids dissolved in miscible, protein-tolerable solvents (like ethanol) rapidly self-assemble into vesicles as they meet the aqueous protein stream. This occurs in milliseconds, minimizing solvent exposure time and eliminating the need for destructive post-processing (sonication).
• Process Optimization: Maintaining temperatures within a protein-tolerable range while also considering lipid phase transition behavior is critical. Furthermore, buffer optimization (selecting appropriate pH and ionic strength) and utilizing stabilizing excipients (such as trehalose or sucrose as cryoprotectants during lyophilization) are mandatory steps to protect the structural integrity of the active pharmaceutical ingredient.
Core Principles for Biologic Protection
- ✓ Utilize low-shear, continuous-flow manufacturing techniques.
- ✓ Implement solvent-free or rapid-dilution aqueous loading strategies.
- ✓ Incorporate specific lyoprotectants to prevent vesicle fusion and protein denaturation during dehydration.
- ✓ Strictly monitor buffer compatibility to avoid isoelectric point precipitation.
Practical Levers for Controlled Release Design
How do you achieve controlled release without over-stabilizing the membrane? Meticulous liposome formulation optimization allows scientists to dictate exactly how and when the peptide is released. The relationship between bilayer composition and release behavior is illustrated by published studies using model protein-loaded liposomes.
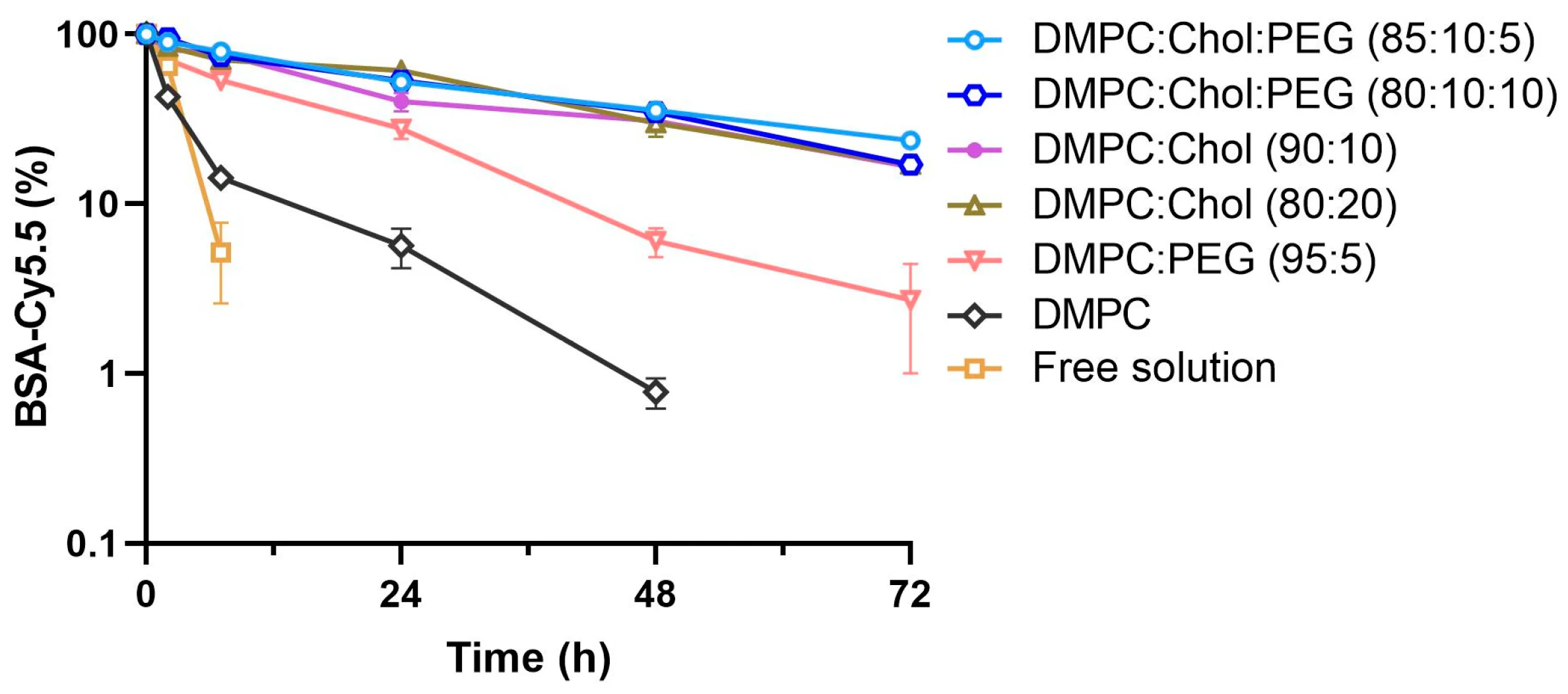
As shown in the evaluation of model protein BSA-Cy5.5 (Fig. 1), liposomal encapsulation significantly retards protein diffusion compared to the free solution. Importantly, the incorporation of cholesterol markedly enhances membrane stability and suppresses unwanted burst release, demonstrating that optimizing protein liposomes and peptide liposomes requires meticulous tuning of the membrane to balance cargo protection with precise release kinetics.
Formulation Variables That Influence Release Behavior
| Formulation Lever | Mechanism of Action | Impact on Release & Stability |
|---|---|---|
| Lipid Phase Behavior (Tm) | Selecting high-Tm saturated lipids (e.g., DSPC) creates rigid, gel-phase bilayers; low-Tm unsaturated lipids (e.g., DOPC) create fluid membranes. | High-Tm lipids restrict diffusion, preventing leakage but potentially hindering payload availability. Low-Tm lipids encourage rapid release but risk premature burst release. |
| Cholesterol Percentage | Acts as a membrane buffer, filling inter-lipid gaps and dampening sharp phase transitions. | Crucial for liposome stability in serum. It tightens fluid bilayers to suppress burst release and limits the membrane's permeability to water and small peptides. |
| Surface Modification | Grafting PEGylated lipids provides a steric hydration shell. | Improves circulation time (stealth effect), but the "PEG dilemma" can severely reduce cellular uptake and subsequent intracellular cargo release. |
| Cargo Localization | Aqueous core encapsulation versus lipid bilayer embedding or surface adsorption. | Core-encapsulated proteins show sustained, diffusion-limited release. Surface-associated peptides generally exhibit rapid burst release upon administration. |
| Stimuli-Responsive Design | Use of pH-sensitive, enzyme-responsive, or thermosensitive lipid variations. | Enables site-triggered release. The liposome remains highly stable systemically but rapidly destabilizes and releases the biologic within specific microenvironments (e.g., tumor acidity or endosomes). |
Route-Specific Design Considerations
The desired route of administration heavily dictates how the liposome must be formulated to balance stability and release:
- • Injectable Formulations (IV/SC): Require strict particle size control (usually < 200nm for systemic circulation), high serum stability to prevent opsonization, and rigorous sterility. Sustained release is often the primary goal.
- • Inhalable Formulations: Designed to withstand the intense shear stress generated during nebulization. They must efficiently traverse or adhere to the pulmonary mucus layer and avoid rapid clearance by alveolar macrophages to ensure local action.
- • Oral Delivery: The most challenging route for peptide delivery liposomes. The formulation must survive severe acidic degradation in the stomach, resist destabilization by bile salts and lipases in the intestine, and facilitate epithelial permeation.
Analytical Tools for Integrity and Release
Continuous analytical evaluation is mandatory to verify that the biologic has not aggregated, liposome size remains uniform, and the release profile aligns with therapeutic targets:
- • Physicochemical Properties: Dynamic Light Scattering (DLS) for tracking Size and PDI; Zeta potential for evaluating surface charge and stability against flocculation. Encapsulation Efficiency (EE%) assessment.
- • Protein Structural Integrity: Circular Dichroism (CD) to monitor secondary structure shifts; Size Exclusion Chromatography (SEC) or SDS-PAGE to detect aggregation/fragmentation; rigorous bioactivity assays (e.g., ELISA) to confirm functional potency.
- • Kinetics & Stability: In vitro release assays simulating physiological conditions; serum stability testing to assess early leakage; and long-term reconstitution stability for lyophilized powders.
How Creative Biolabs Supports Protein & Peptide Liposome Development
We leverage years of specialized lipid nanoparticle and liposomal engineering expertise to help biotech and pharmaceutical developers overcome the complexities of biologic delivery. Whether you need early formulation feasibility or comprehensive release profiling, our tailored workflows minimize risk and accelerate translation.
Encapsulation Strategy Screening
Leverage our Liposome-Encapsulated Protein & Peptide Development Service to identify mild, low-shear loading methods that protect your specific cargo from denaturation.
Lipid Optimization
Utilize our comprehensive Liposome Formulation Optimization to fine-tune the lipid-to-cholesterol ratio, Tm, and surface chemistry for targeted systemic or local delivery.
Release Kinetics Tuning
We conduct rigorous In Vitro Release Kinetics Analysis to eliminate burst release and ensure your therapeutic window is maintained effectively.
Long-term Evaluation
Ensure robust shelf-life and structural integrity over time with our Formulation Stability Monitoring Service, covering both liquid and lyophilized states.
Frequently Asked Questions
Passive encapsulation (e.g., mixing proteins during aqueous hydration or via microfluidics) is generally preferred for fragile, high-molecular-weight proteins because it avoids severe pH or ion gradients. Active loading (e.g., pH gradient method) can achieve much higher encapsulation efficiencies but is typically reserved for smaller peptides or amphipathic molecules that can tolerate transient pH extremes without isoelectric precipitation or unfolding.
Balancing this paradox involves precise lipid engineering. We typically incorporate rigidifying lipids (like high-Tm phospholipids and cholesterol) to prevent premature leakage during systemic circulation. To ensure subsequent payload release at the target site, we can implement stimuli-responsive components—such as pH-sensitive lipids that destabilize in acidic tumor microenvironments/endosomes, or utilize sheddable PEG-coatings that expose fusogenic lipid surfaces only upon reaching the target tissue.
Yes, but they require highly specialized formulations. Inhalable liposomes must be engineered to withstand the shear forces of nebulizers without leaking their cargo, and surface-modified (often PEGylated) to penetrate pulmonary mucus. Oral delivery of peptides is extremely challenging due to GI enzymes and bile salts; however, utilizing highly robust lipid mixtures (e.g., archaeosomes or polymer-coated liposomes) can help shield encapsulated peptides and proteins from enzymatic degradation, provided that vesicle integrity is maintained in vivo.
Validating biologic integrity requires specialized assays after carefully disrupting the liposomal membrane to extract the protein. We utilize Circular Dichroism (CD) to verify the conservation of secondary structure (alpha-helices/beta-sheets), Size Exclusion Chromatography (SEC) to confirm the absence of aggregates, and target-specific in vitro bioassays (like ELISA, enzymatic activity assays, or cell-based receptor binding) to definitively prove that the pharmacological potency remains intact.
Freeze-drying (lyophilization) is vital for shelf-life, but the freezing step can generate ice crystals that mechanically rupture lipid bilayers, leading to significant cargo leakage upon reconstitution. Additionally, removing water can cause protein denaturation and vesicle fusion. These risks are mitigated by adding specific lyoprotectants (such as trehalose or sucrose) to both the internal aqueous core and external buffer, which replace the water hydration shell and stabilize both the lipid membrane and the protein structure during dehydration.
References
- Ponkshe, Pranav, Yingzhe Wang, and Chalet Tan. "Systemic protein delivery via inhalable liposomes: Formulation and pharmacokinetics." Pharmaceutics 15.7 (2023): 1951. https://doi.org/10.3390/pharmaceutics15071951
- Under Open Access license CC BY 4.0, without modification.
Online Inquiry
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.


