Liposome Lamellarity Analysis Service
Background Our Services Workflow Applications Why Choose Us Related Services FAQs
In the intricate design of lipid-based drug delivery systems, the lipid bilayer is more than a boundary—it is the functional core of the therapeutic carrier. Beyond particle size, the specific lamellarity (number of lipid layers) critically influences encapsulation efficiency, drug release kinetics, and in vivo stability. Accurate elucidation of this internal structure is pivotal for validating formulation consistency and ensuring therapeutic efficacy. Creative Biolabs draws upon deep expertise in lipid nanotechnology to deliver precision lamellarity analysis, empowering researchers to achieve the exact structural attributes required for clinical success.
Scientific Principles of Lipid Vesicle Architecture
Defining Liposome Structural Classes
Liposome lamellarity refers to the architectural arrangement of lipid bilayers that enclose the aqueous core of a vesicle. This internal structure is generally categorized into three distinct classes:
-
Small Unilamellar Vesicles (SUVs): Nanoscopic vesicles (<100 nm) consisting of a single lipid bilayer, ideal for passive targeting and long circulation.
-
Large Unilamellar Vesicles (LUVs): Larger structures (>100 nm) with a single bilayer, offering an optimized volume-to-lipid ratio for hydrophilic drug encapsulation.
-
Multilamellar Vesicles (MLVs): "Onion-like" structures containing multiple concentric bilayers separated by aqueous compartments, often used for depot release formulations.
-
Multivesicular Vesicles (MVVs): Also known as Multivesicular Liposomes (MVLs), these are large (>1 µm) structures containing multiple non-concentric aqueous chambers within a single outer membrane, resembling a "honeycomb." They are engineered for high-capacity loading and sustained depot release of biologics.
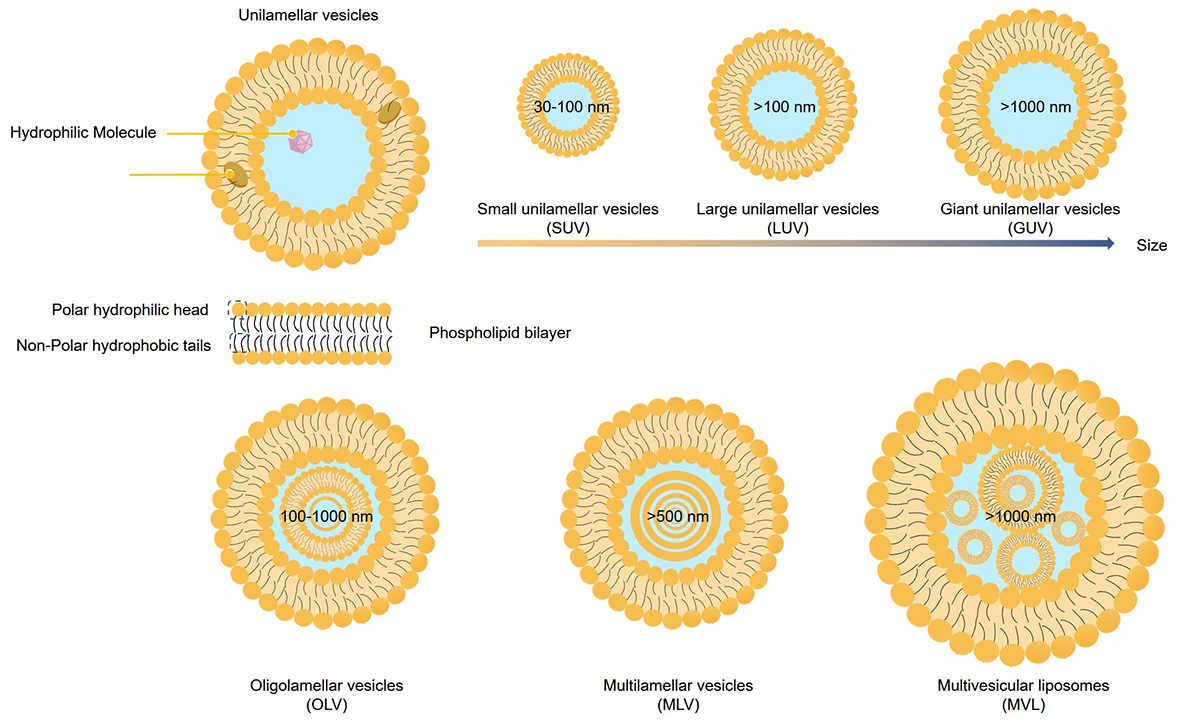 Fig.1 Schematic representation of different sizes and lamellar structures of liposomes.
Fig.1 Schematic representation of different sizes and lamellar structures of liposomes.
Critical Role of Lamellarity in Formulation Performance
The number of bilayers is a Critical Quality Attribute (CQA) that directly impacts the physicochemical performance of the carrier:
-
Release Kinetics: MLVs creates a complex diffusion barrier, slowing drug release (depot effect), whereas unilamellar vesicles typically facilitate rapid release.
-
Loading Capacity: Lamellarity defines the available internal volume (Encapsulation Efficiency). A shift from unilamellar to multilamellar structure can significantly reduce the payload capacity for hydrophilic drugs.
-
Membrane Mechanics: The number of lamellae influences membrane rigidity and deformability, affecting how the liposome interacts with cell membranes and endosomal pathways.
Key Analytical Techniques for Lamellarity Determination
To accurately determine the number of bilayers, various biophysical methods are employed. The choice of technique often depends on the required resolution and the nature of the sample.
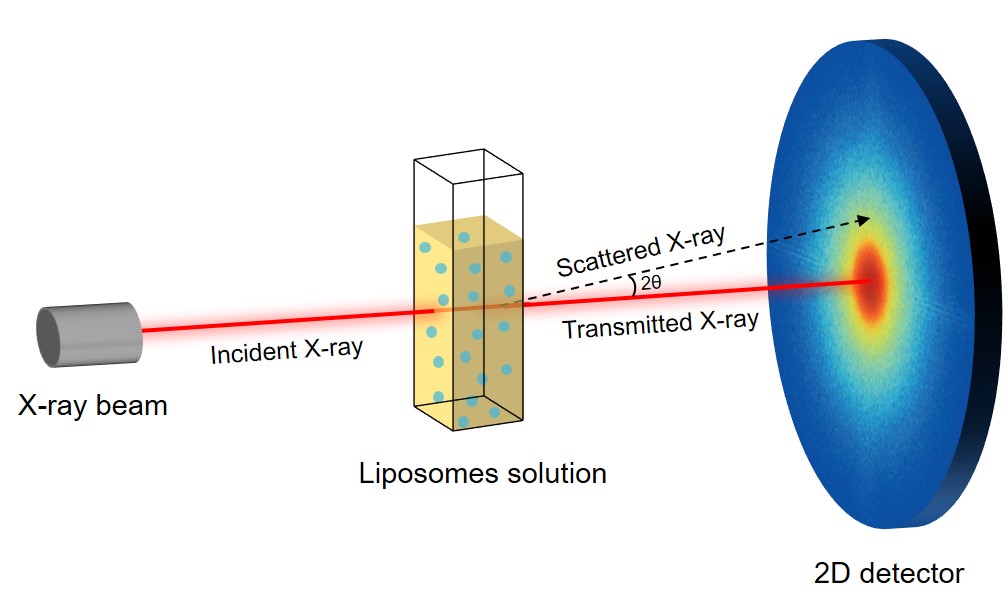 Fig.2 Principle of the SAXS method.
Fig.2 Principle of the SAXS method.
|
Technique
|
Underlying Principle
|
Primary Application
|
|
31P-NMR Spectroscopy
|
Uses paramagnetic shift reagents (e.g., Mn2+, Pr3+) to separate signals from outer and inner leaflet phospholipids.
|
Quantitative determination of average lamellarity in bulk liposome populations.
|
|
Small-Angle X-ray Scattering (SAXS)
|
Measures elastic scattering of X-rays to determine electron density profiles and repeat distances.
|
Analyzing bilayer spacing (d-spacing) and identifying multilamellar or liquid crystalline structures.
|
|
Cryo-Electron Microscopy (Cryo-EM)
|
Direct visualization of samples vitrified in liquid ethane to preserve native hydration.
|
Visual assessment of heterogeneity, invaginations, and distinct structural populations.
|
|
Fluorescence Quenching
|
Uses membrane-impermeable quenchers to reduce fluorescence from outer leaflet dyes.
|
Estimating the fraction of exposed lipids (lamellarity index) in dilute suspensions.
|
Comprehensive Lamellarity Characterization Solutions
Creative Biolabs provides a comprehensive suite of analytical services designed to resolve the "black box" of liposomal structure. Our multi-modal platform ensures we can characterize a wide range of lipid-based systems, from standard liposomes to complex Lipid Nanoparticles (LNPs).
-
Quantitative Average Lamellarity Assessment: Precise determination of outer-to-inner lipid ratios using 31P-NMR with paramagnetic shift reagents (e.g., Mn2+). Delivers calculated indices for validating batch consistency and unilamellarity.
-
Internal Nanostructure & Bilayer Spacing Profiling: Non-destructive SAXS analysis to resolve internal architecture. Determines bilayer repeat distances (d-spacing) and identifies complex phases in multilamellar depot formulations.
-
Single-Particle Visualization & Heterogeneity Analysis: Direct visualization via Cryo-EM to assess population uniformity. Enables counting of lamellae per particle and identification of structural defects in the native hydrated state.
Workflow
Applications of Lamellarity Analysis in Modern Research
Our lamellarity analysis services enable breakthroughs across a diverse range of pharmaceutical and biomedical applications:
-
Gene Delivery Optimization: Verifying the unilamellar structure of liposome carrying mRNA or siRNA to ensure efficient cytosolic delivery and endosomal escape.
-
Vaccine Adjuvant Development: Characterizing the multi-layered structure of antigen-carrying vesicles to predict immune response and stability.
-
Topical & Transdermal Formulations: Analyzing the layer structure and deformability of transferosomes or ethosomes designed to penetrate the stratum corneum.
-
Manufacturing Scale-Up: Monitoring critical process parameters, such as microfluidic flow rates, to ensure bilayer integrity is maintained during the transition from lab to pilot scale.
Why Choose Creative Biolabs for Liposome Lamellarity Analysis?
-
Multi-Modal Technology: We do not rely on a single approach; we integrate NMR, SAXS, and Cryo-EM to provide a complete structural picture.
-
Regulatory Compliance: Our analysis protocols are designed to support IND and NDA filings, providing data that meets rigorous regulatory standards.
-
Customized Protocols: We tailor our methods to your specific lipid composition, accommodating PEGylated, cationic, or anionic formulations.
-
Fast & Reliable: Optimized workflows deliver high-quality data within competitive timelines, accelerating your decision-making process.
-
Expert Consultation: You receive direct support from formulation scientists who help interpret data and suggest process improvements.
Creative Biolabs stands at the forefront of lipid-based drug delivery innovation. Our Liposome Lamellarity Analysis Service combines cutting-edge biophysical techniques with deep industry expertise to provide the structural certainty your research demands. Whether you are optimizing a novel gene therapy vector or validating a generic formulation, our precise data empowers you to move forward with confidence. Our dedicated specialists are available to answer your technical questions and provide a customized quote tailored to your specific research goals.
Related Services
FAQs
How much sample volume is required for analysis?
Typically, we require 200–500 µL of liposome suspension. However, requirements vary by method; please consult our team for specific volume requirements based on your concentration.
Can you analyze samples containing high concentrations of cholesterol?
Yes. While high cholesterol can affect membrane fluidity and NMR signals, our experts can adjust acquisition parameters or recommend complementary techniques like SAXS to ensure accurate characterization.
Do you accept lyophilized liposome samples?
Yes, we accept lyophilized samples. We can perform reconstitution according to your specific protocol before analysis to ensure the structure reflects the intended use state.
What is the typical turnaround time for results?
Standard turnaround is approximately 10–15 business days from sample receipt. Expedited services are available upon request for time-sensitive projects.

For Research Use Only. Not For Clinical Use
 Fig.1 Schematic representation of different sizes and lamellar structures of liposomes.
Fig.1 Schematic representation of different sizes and lamellar structures of liposomes.
 Fig.2 Principle of the SAXS method.
Fig.2 Principle of the SAXS method.
 For Research Use Only. Not For Clinical Use
For Research Use Only. Not For Clinical Use