Liposome vs. LNP:
The Key Difference in Nucleic Acid Delivery
A practical decision guide for choosing the right lipid carrier for mRNA, siRNA, ASO, and plasmid DNA programs.
Liposome vs. LNP: Quick Decision Snapshot
- • Choose LNP first if you are developing mRNA, siRNA, or other RNA payloads that require high encapsulation and efficient cytosolic release.
- • Consider liposomes first if you need a more classical vesicular system, flexible surface engineering, or early-stage plasmid complexation and in vitro transfection.
- • Re-evaluate immediately if your current formulation shows low encapsulation, poor serum stability, weak endosomal escape, or excessive in vivo liver clearance.
Common Misconceptions in Liposome vs. LNP Selection
In nucleic acid delivery, liposome and LNP are often used as if they mean the same thing—but they do not. This confusion can lead to poor payload matching, low encapsulation, weak in vivo performance, and unnecessary iteration in early development. For teams evaluating genetic medicine programs, the real question is not which term sounds newer, but which platform is structurally and functionally suited to the translational goal.
"LNP is just a newer name for liposome."
Not exactly. The difference is not branding, but architecture, loading mechanism, and biological behavior. They interact with nucleic acids fundamentally differently during formulation.
"If it works for plasmid DNA in vitro, it works for mRNA in vivo."
Not necessarily. Payload size, intracellular destination, and systemic exposure requirements are vastly different. In vitro lipoplex success rarely scales directly to in vivo mRNA delivery.
"Encapsulation efficiency alone determines success."
Incomplete picture. High EE matters, but endosomal escape, serum stability, biodistribution, and manufacturability are often equally decisive for clinical translation.
The Key Dividing Line: Liposome vs. Lipid Nanoparticle Architecture
The key difference is not simply vesicle versus nanoparticle, but whether the system is built around passive/vesicular encapsulation or ionizable-lipid-driven nucleic acid complexation.
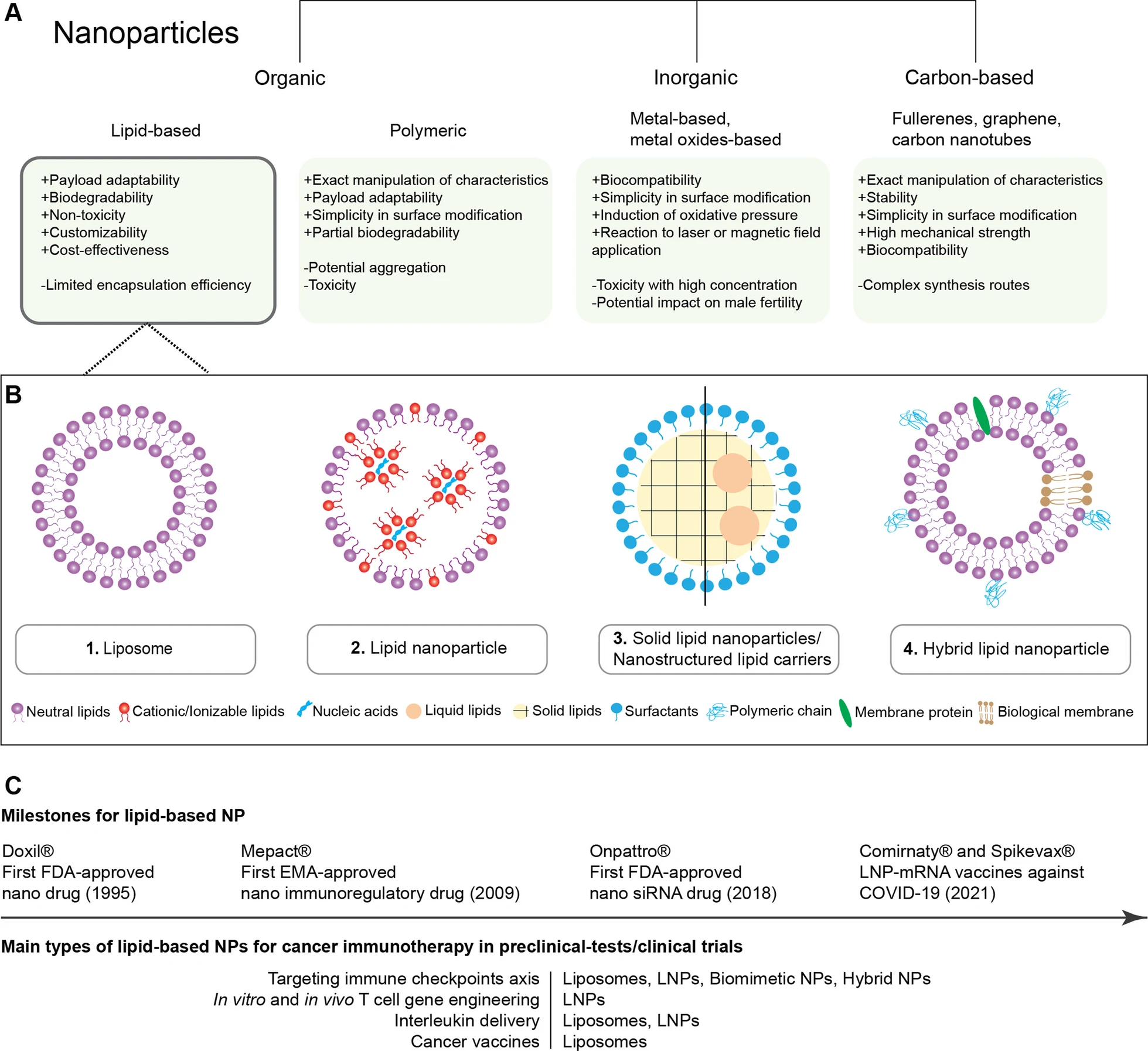
Classical Liposomes
Classical liposomes are typically defined as continuous lipid bilayer vesicles enclosing an aqueous core. When encapsulating hydrophilic payloads, the cargo is localized within this internal fluid space or bound electrostatically to the membrane.
Because the internal volume is defined by the bilayer structure, classical liposome formulations often show lower and more variable nucleic acid encapsulation than LNP systems, unless active complexation strategies are meticulously introduced.
Lipid Nanoparticles (LNPs)
LNPs, by contrast, are generally described as non-bilayer, heterogeneous lipid assemblies. In these systems, ionizable lipids complex nucleic acids into an electron-dense interior rather than a classic hollow aqueous-core vesicle.
The ionizable lipid is the critical engine: it condenses nucleic acids during acidic formulation and supports endosomal escape after cellular uptake. This structural advantage is why LNPs have become the dominant platform for modern RNA therapeutics.
Payload-to-Platform Matching: Optimizing mRNA, siRNA, and pDNA Delivery
Liposome and LNP are not interchangeable terms in nucleic acid delivery. Selecting the correct carrier depends heavily on your payload's molecular weight, structural fragility, and intended mechanism of action. To reduce trial-and-error, our Liposome Encapsulated Nucleic Acid Development Service provides customized screening to match your specific cargo.
| Payload | Best Starting Platform | Why | Key Trade-off |
|---|---|---|---|
| mRNA | Ionizable LNP | High encapsulation capability, superior cytosolic delivery, and clinically validated for RNA vaccines and protein replacement. | Strong default liver tropism; strict cold-chain/stability requirements; formulation sensitivity. |
| siRNA / miRNA | LNP or Specialized Liposomes | Short, rigid double strands pack efficiently into the LNP core. Flexible for liver targeting or, with surface engineering, extrahepatic sites. | Competition with GalNAc conjugates for liver targets; requires robust endosomal escape design. |
| Plasmid DNA (pDNA) | Cationic Liposomes (Lipoplexes) | Large plasmid size makes passive encapsulation difficult. Electrostatic complexation is highly effective for early-stage in vitro screens. | In vivo translation is much harder; what works in vitro may not scale biologically due to systemic toxicity. |
| ASO (Antisense Oligos) | Both Options / Naked Delivery | ASOs may not always require lipid encapsulation due to chemical modifications, but lipid carriers enhance biodistribution and tissue-specific uptake when needed. | Added manufacturing complexity compared to delivering chemically stabilized bare oligos. |
Translation Path: LNP Manufacturing & In Vivo Delivery Performance
Process Dependencies
The transition from academic research to GMP clinical manufacturing often reveals stark differences. Well-optimized ionizable LNP formulations frequently achieve very high RNA encapsulation efficiency (often >90%) because they are predominantly manufactured using rapid microfluidic mixing. This kinetically controlled precipitation induces instantaneous self-assembly, creating highly uniform nanoparticles scalable from micro-liters to industrial liters.
Traditional liposome synthesis often relies on bulk methods like thin-film hydration or extrusion. While effective for smaller batches or specific hydrophilic drugs, these processes can struggle with batch-to-batch consistency when handling sensitive, large nucleic acids. Robust validation through Lipid-based Basic Characterization Services is imperative for both platforms to confirm DLS, Zeta potential, and encapsulation integrity.
Explore LNP Development ServicesThe Biological Reality
Permanently charged cationic liposomes, while excellent for in vitro transfection, are often associated with increased toxicity and rapid clearance in systemic in vivo settings. To bridge this gap, targeted Cationic Liposome Development is required to optimize surface parameters for localized applications or immunomodulation.
Modern LNPs bypass many systemic toxicity issues because their ionizable lipids remain near-neutral at physiological pH (7.4), reducing non-specific protein binding. Once endocytosed, the acidic endosomal environment triggers protonation, causing membrane disruption and safe cytosolic payload release.
Crucial Caveat: That said, LNPs are not universally superior. Their default liver tropism, formulation sensitivity, and strict IP landscapes can become liabilities when extrahepatic targeting, local delivery, or non-RNA payloads are the true priority.
Frequently Asked Questions
While technically possible, classical liposomes generally yield poor systemic results for mRNA. mRNA is massive and negatively charged, making passive encapsulation into a neutral aqueous core highly inefficient. Utilizing LNPs with ionizable lipids is the industry standard to achieve high encapsulation efficiency and the endosomal escape necessary for effective mRNA translation in vivo.
Standard LNPs naturally accumulate in the liver largely due to their interaction with Apolipoprotein E (ApoE) in the bloodstream. ApoE adsorbs onto the surface of the LNP, effectively disguising it as an endogenous lipoprotein. This facilitates rapid uptake by hepatocytes via Low-Density Lipoprotein Receptors (LDLR). Researchers utilize strategies like Selective Organ Targeting (SORT) to overcome this natural tropism.
Cationic liposomes (e.g., those containing DOTAP) maintain a permanent positive charge regardless of environmental pH. This makes them excellent for complexing plasmids in vitro, but they are often associated with increased toxicity and rapid systemic clearance in vivo. LNPs use "ionizable" lipids that are only positively charged at an acidic pH (during formulation and inside endosomes) but remain near-neutral at physiological pH, significantly improving their safety profile.
Yes, our formulation scientists provide comprehensive platform selection consultation. We evaluate your payload (mRNA, siRNA, pDNA, CRISPR/Cas), target disease model, and translation goals to design, synthesize, and rigorously characterize the optimal lipid-based delivery system—whether it requires a specialized liposome or an advanced LNP.
Reference
- Cheng, Ziyun, et al. "Lipid-based nanosystems: the next generation of cancer immune therapy." Journal of Hematology & Oncology 17.1 (2024): 53. https://doi.org/10.1186/s13045-024-01574-1
- Under Open Access license CC BY 4.0, without modification.
Online Inquiry
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.


