Anti-α-Gal Antibody Development Service
The α-Gal epitope (Galα1-3Galβ1-4GlcNAc-R) is a carbohydrate structure abundantly expressed on cells of most mammals but absent in humans, apes, and Old World monkeys due to the inactivation of the alpha-1,3-galactosyltransferase gene. Consequently, humans produce high levels of natural anti-α-Gal antibodies. This unique immunological status makes the study of this epitope critical. At Creative Biolabs, we provide tailored antibody generation services to support research in xenotransplantation rejection, alpha-gal syndrome (AGS), and pathogen glycan mimicry, leveraging our advanced capabilities in Non-Human Glycan Antibody Development.
Background: The Clinical Relevance of Alpha-Gal
The interaction between the alpha-gal epitope and the human immune system is a double-edged sword. In the context of xenotransplantation, the binding of human natural anti-gla antibodies to porcine organs triggers hyperacute rejection, leading to rapid graft failure. Conversely, in the field of allergy, the bite of the lone star tick can sensitize individuals to this carbohydrate, causing delayed anaphylaxis upon consumption of red meat (Alpha-gal Syndrome). Furthermore, certain pathogens may express alpha-gal-like structures to exploit the host's immune response or, paradoxically, to be targeted by natural antibodies.
Evolutionary Loss and High Immunogenicity
In non-primate mammals, the enzyme α1,3-galactosyltransferase (α1-3GT) synthesizes the α-Gal epitope. However, in humans, apes, and Old World monkeys, the GGTA1 gene encoding this enzyme was inactivated due to a premature stop codon millions of years ago. As a result, the human immune system recognizes α-Gal as a foreign antigen. This evolutionary loss has led to the continuous production of natural anti-Gal antibodies (IgM, IgG, and IgA), which can constitute up to 1% of total circulating immunoglobulins. This high baseline immunogenicity is why α-Gal triggers such robust reactions, ranging from hyperacute rejection of xenografts to severe allergic responses.
Structural Homology with Blood Group B Antigen
The α-Gal epitope (Galα1-3Gal) shares a striking structural similarity to the Blood Group B antigen (Galα1-3(Fucα1-2)Gal). The only difference is the absence of a fucose residue in α-Gal. Research indicates that individuals with Blood Group B or AB are generally protected from developing high titers of IgE against α-Gal, likely due to immune tolerance mechanisms recognizing the structure as "self." Conversely, individuals with Blood Group A or O lack this tolerance and are at a significantly higher risk of sensitization and developing Alpha-gal Syndrome following tick exposure.
Widespread Distribution of α-Gal Epitopes
The α-Gal epitope is ubiquitous in mammalian tissues and can be found in various therapeutic products derived from animals. Understanding these sources is critical for antigen selection and specificity testing during antibody development.
| Source Category | Examples | Key α-Gal Carrying Molecules Identified |
|---|---|---|
| Mammalian Meat | Beef, Pork, Lamb, Rabbit, Horse | Laminin γ-1, Collagen α-1 (VI), Bovine Gamma Globulin (BGG), Creatine kinase M type, Aspartate aminotransferase |
| Innards (High Risk) | Pork Kidney, Beef Kidney | Aminopeptidase N, Angiotensin-converting enzyme 1 (ACE I) |
| Pharmaceuticals | Monoclonal Antibodies, Antivenoms | Cetuximab (Fab region glycosylation), Equine/Ovine polyclonal Fab fragments |
| Medical Products | Vaccines, Plasma Substitutes, Heart Valves | Gelatin stabilizers (MMR, Zoster vaccines), Gelatin-based plasma expanders, Porcine/Bovine pericardium |
| Ticks | Ixodes ricinus, Amblyomma americanum | Salivary glycoproteins, Vitellogenins, α-2-macroglobulin |
Clinical Implications for Development
Given the wide distribution of this epitope, developing antibodies requires rigorous negative screening. Key considerations include:
- Cross-reactivity: Antibodies must be screened against Blood Group B antigens to ensure specificity for the linear α-Gal structure.
- Format Selection: Since natural IgG antibodies are abundant, developing monoclonal IgE or specific IgG subclasses (e.g., IgG1) is often necessary for mechanistic studies.
- Carrier Variability: As shown in the table, α-Gal appears on diverse carriers (lipids vs. proteins like Collagen or ACE). We tailor the immunogen (e.g., neoglycoproteins) to match the target context.
Challenges in Anti-α-Gal Antibody Research
Generating reliable antibodies against the alpha-gal epitope presents significant hurdles for researchers.
Low Immunogenicity
As a carbohydrate antigen, alpha-gal is T-cell independent and generally weakly immunogenic in standard laboratory animals that express the epitope themselves (tolerance).
Cross-Reactivity
Many antibodies fail to distinguish between the specific Galα1-3 linkage and other terminal galactose structures (e.g., Galα1-4 or Galβ1-4), leading to false positives in tissue staining.
Isotype Limitations
Natural anti-α-Gal antibodies in humans are a mixture of IgM, IgG, and IgA. Research often requires specific monoclonal isotypes to dissect the mechanism of complement activation versus allergic sensitization (IgE).
Our Solutions: Comprehensive Anti-α-Gal Antibody Services
Creative Biolabs overcomes these obstacles by utilizing alpha-1,3-galactosyltransferase knockout (KO) mice and specialized immunization protocols. We offer a full spectrum of services tailored to your specific research needs.
Monoclonal Antibody Discovery
We utilize GalT-KO mice immunization followed by hybridoma or phage display screening to generate high-affinity monoclonal antibodies (mAbs) that are strictly specific for the Galα1-3Gal terminus. We can select for specific isotypes, including IgG for cytotoxicity studies or IgE for allergy models.
Antibody Humanization & Engineering
For projects focused on potential therapeutic applications or minimizing anti-drug antibodies in preclinical models, we offer antibody humanization services. We also engineer Fc regions to modulate effector functions, such as enhancing or silencing ADCC/CDC activity.
Detection Assay Development
We develop and validate matched antibody pairs for Sandwich ELISA to quantify alpha-gal content in biological samples (e.g., porcine heart valves, vaccines, or food products). We also optimize protocols for Western Blot and Flow Cytometry.
Alpha-Gal Conjugates & Controls
To support your assays, we supply high-purity Alpha-Gal-BSA/HSA conjugates as coating antigens or positive controls. We also provide negative control proteins with related glycan structures (e.g., Galβ1-4) to confirm specificity.
Project Workflow
Core Advantages
High Specificity
Validated against comprehensive glycan arrays to ensure no cross-reactivity with beta-gal.
KO Mouse Models
Access to unique GalT-KO hosts allows for robust immune responses against the epitope.
Versatile Formats
Development of IgM, IgG, IgE, or recombinant fragments (scFv) tailored to your study.
Fast Turnaround
Optimized workflows ensure you receive your research materials within competitive timelines.
Inquire about Anti-α-Gal Antibodies
Published Data
Recent advancements in glyco-immunology have validated a robust methodology for isolating and characterizing antibodies specific to the galactose-alpha-1,3-galactose (α-Gal) epitope. Utilizing a precise affinity chromatography approach involving a synthetic α-Gal matrix, researchers successfully purified a polyclonal antibody population from human plasma sources. Detailed isotype profiling revealed a predominance of the IgG2 subclass (approximately 83%), consistent with an immune response driven by gastrointestinal bacterial flora, rather than the IgG1 profile typically associated with tick-induced sensitization.
Crucially for assay development, these purified antibodies demonstrated remarkable specificity in Enzyme-Linked Immunosorbent Assays (ELISA). As illustrated in the accompanying data, the antibodies exhibited strong, dose-dependent binding to α-Gal trisaccharides and disaccharides while maintaining negligible reactivity towards structurally homologous glycans, including N-acetyllactosamine (LacNAc) and Blood Group A or O antigens. Only minor interaction was observed with Blood Group B, confirming the high selectivity required for distinguishing α-Gal in complex biological matrices. Further kinetic characterization via Surface Plasmon Resonance (SPR) established high-affinity binding with dissociation constants (KD) in the nanomolar range (144 nM), far exceeding the typical affinity of anti-carbohydrate antibodies. This combination of high specificity and strong affinity confirms the suitability of such antibodies for sensitive detection applications, including Western Blotting and flow cytometry, in both xenotransplantation safety and allergy diagnostics.
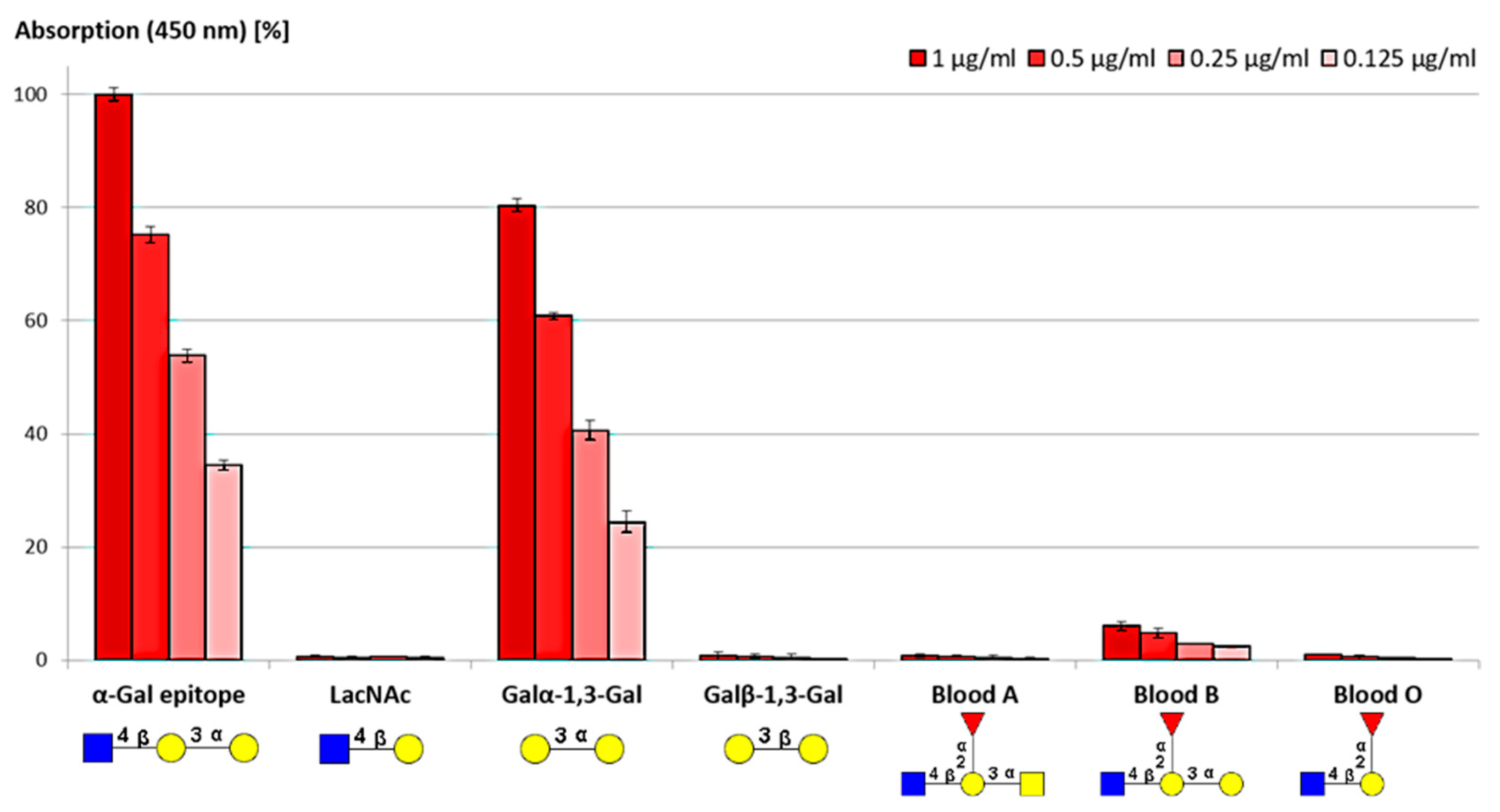 Fig.1 Specificity profile of affinity-purified anti-α-Gal IgG antibodies against a panel of
structurally related glycan epitopes in ELISA.1
Fig.1 Specificity profile of affinity-purified anti-α-Gal IgG antibodies against a panel of
structurally related glycan epitopes in ELISA.1
FAQs
Do your antibodies cross-react with Blood Group B antigens?
The Blood Group B antigen structure (Galα1-3(Fucα1-2)Gal) is very similar to the alpha-gal epitope. However, our screening process specifically includes Blood Group B antigens as negative controls. We select clones that bind exclusively to the linear alpha-gal structure without fucose, minimizing cross-reactivity.
Can I use these antibodies for staining porcine tissues?
Yes. Our anti-α-Gal antibodies are validated for Immunohistochemistry (IHC) and Immunofluorescence (IF) on porcine tissues (e.g., kidney, heart) to detect the expression of the epitope. They are excellent tools for characterizing GalT-KO pigs.
Are these antibodies suitable for ELISA detection of alpha-gal in food?
Absolutely. We can develop a sandwich ELISA kit using a matched pair of our antibodies to detect alpha-gal presence in red meat extracts, gelatin, or other mammalian-derived food products relevant to Alpha-gal Syndrome research.
What is the host species for your standard anti-α-Gal antibody production?
We primarily use Alpha-1,3-galactosyltransferase Knockout (GalT-KO) mice. Since these mice lack the epitope, they can produce a robust immune response against it, unlike wild-type mice which are tolerant to alpha-gal.
Do you offer IgE isotype anti-α-Gal antibodies?
Yes, we can generate IgE class-switched antibodies or engineer recombinant IgE formats. These are particularly useful for establishing in vitro models of basophil activation or mast cell degranulation in allergy studies.
What Our Customers Say
"We needed a control antibody for our xenotransplantation model. The clone Creative Biolabs developed showed zero background on human cells but stained porcine endothelium beautifully. The specificity is exactly what we needed."
"Investigating alpha-gal syndrome requires precise reagents. Their team suggested using a specific neoglycoprotein for screening, which solved our cross-reactivity issues with other dietary glycans."
Reference:
- Zappe, A., et al. "Purification and Characterization of Antibodies Directed against the α-Gal Epitope." BioChem 1.2 (2021): 81-97. Distributed under Open Access license CC BY 4.0. https://doi.org/10.3390/biochem1020008
