Anti-Endoglycosidase Antibody Development Service
At Creative Biolabs, we provide comprehensive solutions for antibody generation against carbohydrate-active enzymes, as part of our premier Anti-Glycan Related Enzyme Antibody Development Service. This specific module focuses on the development of antibodies targeting bacterial endoglycosidases (such as EndoE, EndoS, and EndoF) and other related glycoside hydrolases. These enzymes are critical tools in structural biology for dissecting N-glycan processing mechanisms and in the engineering of antibodies and Fc-fusion proteins.
Targeting Endoglycosidases: Key to Glyco-Engineering Tools
Bacterial endoglycosidases, particularly those from the glycoside hydrolase families GH18 and GH20, possess unique capabilities to cleave the glycosidic bonds within the chitobiose core of N-linked glycans. Enzymes like EndoE (from Enterococcus faecalis), EndoS (from Streptococcus pyogenes), and EndoF variants are widely utilized to deglycosylate IgG antibodies or remodel glycans on therapeutic proteins. Understanding the multi-modular structure of these enzymes and their specific interaction with complex or high-mannose glycans is essential for optimizing their use in biotechnology. High-affinity antibodies against these enzymes serve as pivotal reagents for monitoring enzyme expression, verifying purity during production, and detecting residual enzyme contamination in downstream processing.
Challenges in Anti-Endoglycosidase Antibody Generation
Developing specific antibodies against these enzymes presents distinct challenges. Many endoglycosidases share conserved catalytic domains, leading to potential cross-reactivity that complicates the differentiation of specific homologs (e.g., distinguishing EndoF1 from EndoF3). Furthermore, these enzymes are often used in complex reaction mixtures containing mammalian proteins, requiring antibodies that do not cross-react with host serum components. Creative Biolabs addresses these issues through precise antigen design and rigorous negative screening protocols.
Structural Homology
High sequence identity within glycoside hydrolase families (e.g., GH18) makes it difficult to generate isoform-specific antibodies without expert epitope design.
Assay Sensitivity
Detecting trace amounts of residual endoglycosidase in a deglycosylated product requires antibodies with exceptional affinity and low background.
Complex Matrices
Antibodies must function reliably in diverse buffers and in the presence of cleaved glycans or denatured IgG fragments.
Our Service Portfolio
We offer a streamlined workflow from antigen preparation to antibody validation, specifically tailored for anti-glycosidase antibody development. Our platform supports the generation of polyclonal and monoclonal antibodies against a wide array of targets.
Anti-EndoE & Anti-EndoS Antibody Development
Targeting enzymes involved in IgG glycan hydrolysis. We develop antibodies that recognize the specific domains of EndoE and EndoS, facilitating studies on their mechanism of action in immune evasion and their application in antibody drug conjugate (ADC) manufacturing.
Anti-EndoF & Anti-PNGase F Antibody Development
We provide specific reagents for EndoF1, EndoF2, EndoF3, and PNGase F. These antibodies are crucial for quality control in glycan release workflows and for verifying enzyme removal after the deglycosylation of glycoproteins.
Anti-Neuraminidase (Sialidase) Antibody Development
Beyond endoglycosidases, we cover exoglycosidases. Our service includes the generation of anti-neuraminidase antibodies targeting viral neuraminidase (e.g., Influenza) or bacterial sialidase antibodies (e.g., NEU1, NEU2, NEU3, NEU4) for inhibition assays and structural studies.
Anti-Mannosidase & Glycosyltransferase Antibody Development
We extend our capabilities to other carbohydrate active enzyme antibodies, including mannosidase antibody, chitinase antibody, and transferases like sialyltransferase antibody (ST3GAL, ST6GAL1) and fucosyltransferase antibody (FUT8, FUT1).
Service Highlights
High Specificity
Distinguish between closely related isoforms (e.g., EndoE vs. EndoS) using domain-specific immunogens.
Versatile Applications
Validated for Western Blotting, ELISA (residual enzyme detection), and Inhibition Assays.
Multiple Species
Available in mouse, rabbit, llama (VHH), and recombinant formats to suit your experimental design.
Analytic Support
Supported by our Glycosylation Analysis and Glycosyltransferase & Glycosidase Substrate Microarray services.
Inquire for Your Custom Project
Published Data
Elucidating the precise molecular interplay between modifying enzymes and therapeutic antibodies is fundamental to advancing glyco-engineering strategies. A groundbreaking 2023 study utilizing Cryo-Electron Microscopy (Cryo-EM) provided the first detailed visualization of the bacterial endoglycosidase EndoS complexed with its target, the IgG1 Fc fragment. The resulting structural data revealed a distinctive "V"-shaped architecture where the enzyme physically anchors itself to the antibody through a specific protein-protein interface, rather than relying solely on carbohydrate recognition. Specifically, the non-catalytic β-sandwich domain of the enzyme binds tightly to the CH2-CH3 junction of the Fc region, acting as a molecular clamp. This interaction stabilizes the complex and perfectly positions the catalytic domain to access and hydrolyze the conserved N-glycans at Asn297. Furthermore, kinetic analysis coupled with structural modeling identified a sequential deglycosylation mechanism, where the enzyme processes one heavy chain at a time. These findings highlight the critical role of high-affinity, domain-specific antibodies in validating enzyme integrity and expression levels for structural biology campaigns. For researchers developing glyco-engineered therapeutics or studying immune evasion, accessing validated reagents that target these specific enzymatic domains is essential for quality control and mechanistic characterization.
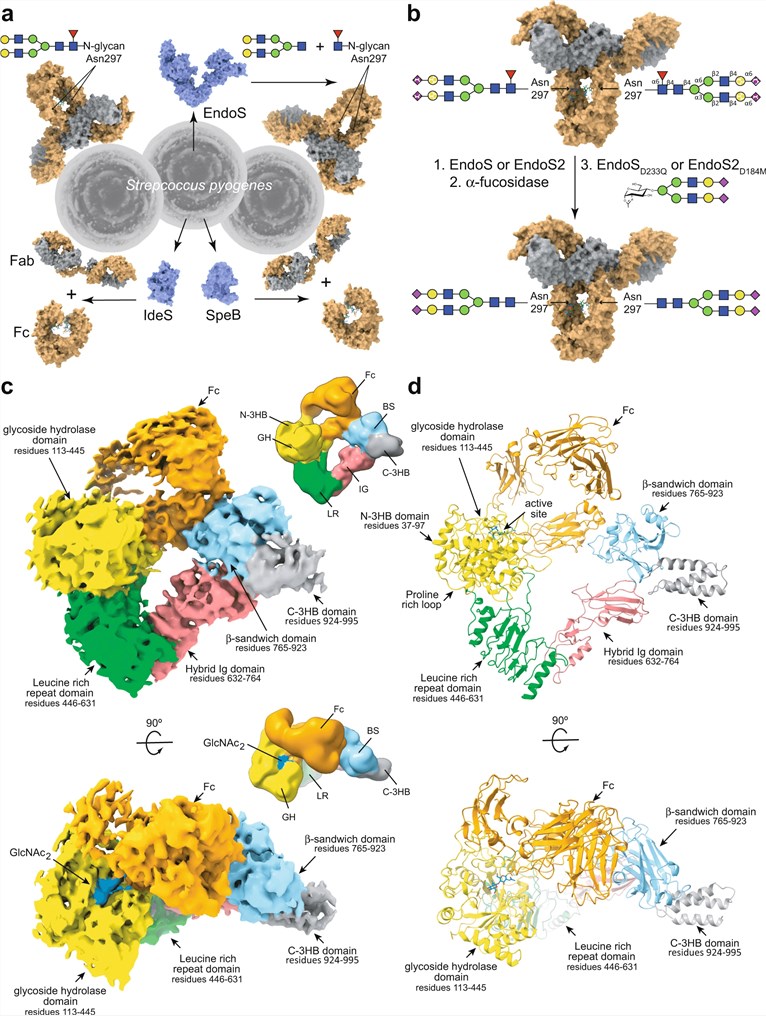 Fig.1
Cryo-EM reconstruction of the EndoS-Fc complex revealing the V-shaped clamping mechanism and protein-specific recognition sites.1
Fig.1
Cryo-EM reconstruction of the EndoS-Fc complex revealing the V-shaped clamping mechanism and protein-specific recognition sites.1
FAQs
Do your antibodies cross-react between EndoH and EndoF?
EndoH and EndoF share some structural similarities, but their specific epitopes differ. We can design screening strategies to identify clones that are specific to either EndoH or EndoF, or clones that are cross-reactive, depending on your research needs. We recommend specifying the desired cross-reactivity profile during the project planning phase.
Can these antibodies be used to detect residual enzymes in therapeutic antibody preparations?
Yes, one of the primary applications of our anti-glycan enzyme antibodies is the detection of residual endoglycosidases (like EndoS or IdeS) in drug substances. We can develop high-sensitivity ELISA pairs or HCP (Host Cell Protein) assay reagents to ensure your final product is free of processing enzymes.
Do you offer antibodies against mammalian sialidases or transferases?
Yes, our service covers a broad range of carbohydrate-active enzymes. We develop antibodies against mammalian targets such as NEU1 antibody, ST6GAL1 antibody, and FUT8 antibody. These are valuable for studying glycosylation pathways in cell biology and disease models.
What host species are available for immunization?
We offer standard host species such as mouse, rat, and rabbit. For targets that are highly conserved or toxic, we also provide custom antibody generation in llamas (to produce VHH) or chicken (IgY), which can often yield higher specificity against bacterial enzymes.
Are these reagents suitable for immunoprecipitation (IP)?
Yes, we can validate antibodies for IP applications. This is particularly useful for co-IP studies to investigate the interaction between glycosyltransferases (e.g., MGAT, B3GNT) and their protein partners within the Golgi apparatus.
What Our Customers Say
"The anti-EndoE antibody we ordered has been essential for our glyco-engineering projects. We needed a reliable way to check for enzyme clearance after the reaction, and this antibody showed zero cross-reactivity with our target protein. Excellent sensitivity in ELISA."
"Working with bacterial sialidases can be tricky due to antibody specificity issues. Creative Biolabs generated a monoclonal antibody that perfectly distinguished our recombinant enzyme from endogenous background. The validation data provided was very thorough."
"We requested antibodies against FUT8 for a knockout validation study. The project management was smooth, and the antibodies worked great for Western blotting on cell lysates. It saved us a lot of time compared to testing commercial reagents of unknown quality."
"Our team needed to monitor the expression of a novel glycosyltransferase. The custom polyclonal package was cost-effective and delivered high-titer serum. The technical support team was very knowledgeable about carbohydrate-active enzymes."
Reference:
- Trastoy, B., et al. "Mechanism of antibody-specific deglycosylation and immune evasion by Streptococcal IgG-specific endoglycosidases." Nature Communications 14.1 (2023): 1705. Distributed under Open Access license CC BY 4.0, without modification. https://doi.org/10.1038/s41467-023-37215-3
Supports
- Glycosylation Analysis Services
- Custom Glycosylation of Biomolecules
- Glycosyltransferase & Glycosidase Substrate Microarray
