Anti-Hyaluronidase Antibody Development Service
Hyaluronidases are a specialized class of glycosidases responsible for the enzymatic cleavage of hyaluronic acid (HA), a major component of the extracellular matrix. These enzymes play pivotal roles in diverse physiological and pathological processes, including fertilization, embryonic development, angiogenesis, and tumor metastasis. At Creative Biolabs, we offer a premier custom development service dedicated to generating high-affinity antibodies against specific hyaluronidase isoforms. As a core component of our broader Anti-Glycan Related Enzyme Antibody Development Service, this platform provides researchers with precision tools to dissect the complex biological functions of these enzymes in cellular microenvironments.
Background: Hyaluronidases in Health and Disease
The human genome encodes six hyaluronidase-like genes (HYAL1, HYAL2, HYAL3, HYAL4, PH-20/SPAM1, and HYALP1), clustered on chromosomes 3p21.3 and 7q31.3. Among these, HYAL1 and HYAL2 are the major somatic hyaluronidases. HYAL1 is predominantly lysosomal, active at acidic pH, and instrumental in degrading HA into small fragments. In contrast, HYAL2 is anchored to the plasma membrane via a GPI anchor, generating intermediate-sized HA fragments. The sperm adhesion molecule 1 (SPAM1 or PH-20) exhibits neutral pH activity and is essential for fertilization.
In oncology, aberrant expression of hyaluronidases correlates with tumor progression. For instance, elevated HYAL1 levels in bladder and prostate cancers are associated with high-grade disease and metastasis, while its downregulation in other contexts may impede tumorigenesis. Therefore, reliable anti-glycosidase antibody tools are indispensable for determining the precise localization, expression levels, and functional status of these enzymes in various tissues. However, the high sequence homology between isoforms and their post-translational modifications pose significant hurdles for antibody generation.
Challenges in Anti-Hyaluronidase Antibody Development
High Homology
The HYAL family shares significant structural similarity. Antibodies raised against full-length proteins often cross-react with multiple isoforms (e.g., distinguishing HYAL1 from HYAL2), confounding experimental results.
Low Immunogenicity
As endogenous mammalian enzymes, hyaluronidases are often highly conserved across species, leading to immune tolerance in standard host animals and resulting in low-titer or low-affinity antibodies.
Conformational Epitopes
Many applications, such as neutralization assays or flow cytometry, require antibodies that recognize the native, folded structure of the enzyme, which peptide immunogens may fail to mimic.
Our Comprehensive Service Portfolio
Creative Biolabs addresses these challenges through a robust pipeline that integrates advanced antigen design with high-throughput screening. We provide end-to-end solutions for the development of polyclonal, monoclonal, and recombinant antibodies targeting specific hyaluronidase members.
Isoform-Specific Antigen Design
We analyze the crystal structures and sequence alignments of the HYAL family to identify unique, surface-exposed epitopes. We utilize both peptide synthesis (for linear epitopes) and recombinant protein expression (for conformational epitopes) to ensure the generated anti-glycan enzyme antibody binds exclusively to the target isoform.
Hybridoma & Phage Display Platforms
Clients can choose between traditional hybridoma technology for high-affinity IgG production or our advanced phage display library screening. The latter is particularly effective for generating antibodies against conserved antigens or for selecting binders with specific functional properties, such as enzyme inhibition.
Functional Validation
Beyond standard binding assays (ELISA, WB), we offer specialized functional validation. This includes hyaluronidase inhibition assays to identify neutralizing antibodies and IHC screening on tissue arrays to confirm specific staining patterns in relevant cancer or normal tissue samples.
Recombinant Antibody Engineering
We can engineer the resulting antibodies into various formats, including scFv, Fab, or full-length IgGs with modified Fc regions. This flexibility allows researchers to adapt the anti-glycosidase antibody for specific applications, such as imaging or creating antibody-drug conjugates for research use.
Key Features and Benefits
High Specificity
Guaranteed discrimination between HYAL1, HYAL2, and PH-20 isoforms.
Extensive Validation
Validated for IHC, WB, and ELISA to ensure performance in your assay.
Diverse Hosts
Options for mouse, rabbit, rat, and llama (VHH) antibody production.
Cost-Effective
Optimized workflows to deliver high-quality results within budget and timeline.
Start Your Project
Collaborating with Creative Biolabs allows you to leverage our extensive expertise in the field of carbohydrate active enzyme antibody development. Whether you require a research reagent to detect HYAL1 in serum or an inhibitor to study HA degradation in the tumor microenvironment, our team is ready to design a solution tailored to your scientific needs. We ensure transparency, rigorous quality control, and scientific support throughout the project lifecycle.
Request a Quote
Published Data
Understanding the catabolism of hyaluronan (HA) in the skin is pivotal for dermatological research and therapeutic development. A recent comprehensive study utilized specific polyclonal antibodies to map the spatial distribution of key HA-degrading proteins—HYAL1, HYAL2, TMEM2, and CEMIP—within healthy human full-thickness skin. Through the application of immunohistochemistry (IHC) and immunohistofluorescence (IHF), researchers achieved high-resolution localization of these enzymes, distinguishing their specific roles in the extracellular matrix. The results revealed distinct compartmentalization: HYAL1 was predominantly found in the intracellular lysosomes of epidermal keratinocytes, particularly in the stratum spinosum and granulosum. In contrast, HYAL2 showed a broader distribution throughout the epidermis and dermis. Notably, the study offered the first in situ visualization of TMEM2 and CEMIP in human skin, identifying intracellular TMEM2 in keratinocytes and fibroblasts, and restricting CEMIP expression to the basal epidermal layer. These findings emphasize the necessity of high-affinity, isoform-specific antibodies for dissecting enzymatic pathways in tissue microenvironments. Utilizing such precise reagents is essential for investigating skin aging mechanisms, wound healing processes, and the longevity of HA-based dermal fillers.
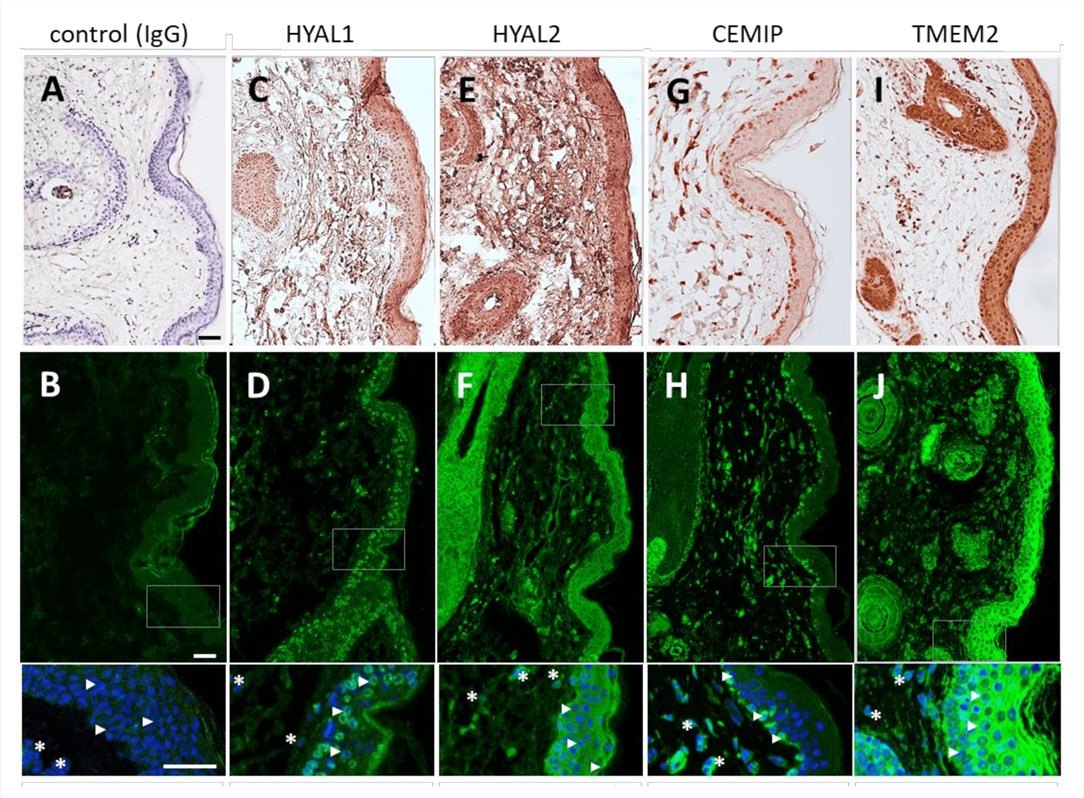 Fig.1
Immunohistochemical and immunofluorescent localization of hyaluronan-degrading enzymes (HYAL1, HYAL2, CEMIP, and TMEM2) in human skin tissue using specific antibodies.1
Fig.1
Immunohistochemical and immunofluorescent localization of hyaluronan-degrading enzymes (HYAL1, HYAL2, CEMIP, and TMEM2) in human skin tissue using specific antibodies.1
FAQs
How do you ensure the antibody differentiates between HYAL1 and HYAL2?
Specific differentiation is achieved through rigorous epitope mapping and negative screening. We identify peptide sequences that are unique to the target isoform and not conserved in others. During the screening phase, we perform counter-screens against the non-target isoforms to eliminate cross-reactive clones.
Can you generate neutralizing antibodies that inhibit enzyme activity?
Yes. To generate neutralizing antibodies, we target the catalytic domain of the hyaluronidase enzyme. We can employ phage display strategies to select binders that block the active site, and we confirm their inhibitory potential using our established enzymatic activity assays.
What host species are available for antibody production?
We offer a wide range of host species, including mice and rats for monoclonal antibodies, and rabbits, goats, and sheep for polyclonal antibodies. We also provide camelid (llama/alpaca) immunization services for VHH development, which is advantageous for targeting cryptic epitopes.
What applications can these antibodies be validated for?
Our standard validation package typically includes ELISA and Western Blot. Upon request, we can extend validation to include Immunohistochemistry (IHC), Immunocytochemistry (ICC), Flow Cytometry, and functional inhibition assays, depending on the intended research application.
Do you provide the antigen for immunization?
Yes, we offer comprehensive antigen preparation services. We can synthesize specific peptides coupled to carriers (KLH/BSA) or express recombinant proteins in mammalian or bacterial systems to serve as high-quality immunogens for your project.
Is this service suitable for detecting hyaluronidase in serum samples?
Absolutely. We can develop high-sensitivity antibody pairs suitable for sandwich ELISA platforms, enabling the quantitative detection of circulating hyaluronidase levels in serum or plasma for research studies.
What Our Customers Say
"We required an antibody that could distinguish HYAL1 from HYAL2 for our bladder cancer research. Creative Biolabs designed a peptide antigen that worked perfectly. The resulting antibody showed clear specific staining in our IHC assays without cross-reactivity. Excellent work."
"Our lab focuses on ECM degradation, and we needed a neutralizing antibody for PH-20. The team at Creative Biolabs suggested phage display, and the binders they delivered effectively inhibited enzymatic activity in our in vitro models. Highly recommended for functional antibody development."
Reference:
- Žádníková, P., et al. "The Degradation of Hyaluronan in the Skin." Biomolecules 12.2 (2022): 251. Distributed under Open Access license CC BY 4.0. https://doi.org/10.3390/biom12020251
Supports
- Glycosylation Analysis
- Custom Glycosylation of Biomolecules
- Glycosyltransferase & Glycosidase Substrate Microarray
