Sialic Acids: The "Face" of the Cell
Sialic acid-focused reagent development is often a useful starting point when a study centers on terminal glycan recognition, Siglec engagement, or immune regulation. At Creative Biolabs, we support glycan-focused research through our custom anti-sialic acid antibody development service, glycosylation analysis, sialoside binding profiling, and membrane glycoengineering solutions tailored to different research goals. This page explains why sialic acids are important in cell-cell recognition, how Siglec receptors read sialylated structures, and which experimental strategies are useful when a project requires more precise analysis of sialylated glycoconjugates.
Why Sialic Acids Matter on the Cell Surface
Sialic acids are a family of acidic nine-carbon monosaccharides typically found at the non-reducing ends of glycans on glycoproteins and glycolipids. Because they occupy terminal positions, they are among the first molecular features encountered during contact between cells, proteins, pathogens, antibodies, lectins, and extracellular matrices. This terminal location gives them strong biological influence. Even a modest change in sialylation can alter charge, steric presentation, receptor access, and downstream signaling.
In mammalian systems, sialic acids are major components of the glycocalyx. They affect how cells are recognized, how long some serum glycoproteins persist, how immune cells distinguish self-associated surfaces, and how receptors remain exposed or masked. In this sense, sialylation often helps define the outermost biochemical signature available for interaction.
| Topic | Key Point | Research Relevance |
|---|---|---|
| Terminal position | Sialic acids cap many glycan chains on proteins and lipids. | Strong impact on binding accessibility and recognition events. |
| Negative charge | They contribute electrostatic repulsion and surface hydration. | Important for membrane behavior, receptor clustering, and trafficking. |
| Structural diversity | Different backbones, linkages, and modifications generate distinct ligands. | Critical for selective binding by Siglec receptors, antibodies, and pathogens. |
| Immune interpretation | Sialylated glycans can function as self-associated molecular patterns. | Relevant to immune homeostasis, tolerance, and inflammatory control. |
Core Concepts in Sialic Acid Biology
Common Molecular Forms
The two most discussed mammalian sialic acids are N-acetylneuraminic acid and N-glycolylneuraminic acid. In humans, N-acetylneuraminic acid is dominant because humans do not synthesize N-glycolylneuraminic acid endogenously. Beyond the core monosaccharide identity, biological behavior also depends on linkage context, including alpha2,3, alpha2,6, and alpha2,8 linkages, as well as additional modifications such as O-acetylation.
Major Structural Contexts
- Sialylated N-glycans on membrane and secreted glycoproteins
- Sialylated O-glycans, especially on mucin-type proteins
- Gangliosides and other glycolipids in plasma membranes
- Polysialylated structures in neural and developmental systems
What Sialylation Changes Functionally
Sialylation is not a decorative modification. It can influence ligand affinity, receptor exposure, half-life in circulation, susceptibility to enzymatic trimming, and the threshold for immune activation. It also helps determine whether a glycan motif is interpreted as a tolerated self-associated pattern or as a signaling surface with functional consequences. In practical terms, the same glycoprotein or cell surface can behave differently after terminal sialylation is altered.
Recognition
Terminal sialylation changes how lectins, antibodies, and receptors contact a glycan-bearing surface.
Signaling
Receptor engagement by specific sialoglycans can amplify or dampen intracellular signaling cascades.
Trafficking
Sialylation influences serum persistence, endocytosis, and interaction with glycan-binding partners.
Immune Tone
The sialylated glycocalyx helps shape how immune cells interpret a surface in different biological contexts.
Sialic Acids and Cell-Cell Recognition
Cell-cell recognition is influenced by more than proteins alone. Glycans provide an additional regulatory layer, and sialic acids are especially important because they are both chemically prominent and biologically interpretable. When two cells meet, terminal sialylation can facilitate engagement with a compatible lectin or shield underlying epitopes from interaction. This matters in immune surveillance, leukocyte trafficking, host-pathogen adhesion, and receptor organization at the membrane.
In some systems, sialylation creates a direct ligand. In others, it limits access to a subterminal glycan or protein determinant. This dual role helps explain why sialic acid biology is highly context dependent. Increased sialylation does not always mean increased binding. Depending on the system, it may create recognition, reduce recognition, or shift recognition toward a different receptor class.
Representative Interaction Modes
- Direct recognition by Siglec receptors on immune cells
- Modulation of selectin-mediated adhesion through sialylated glycan motifs
- Masking of subterminal glycan determinants and protein epitopes
- Control of membrane spacing and glycocalyx architecture
Siglec Receptors and Immune Regulation
Siglec receptors are sialic acid-binding immunoglobulin-like lectins expressed mainly on immune cells. They recognize sialoglycans in linkage- and context-dependent ways and often function as regulators of immune activation. Many Siglecs contain immunoreceptor tyrosine-based inhibitory motifs, while some activating family members signal through associated adaptors such as DAP12.
This makes the sialic acid-Siglec axis one of the clearest examples of glycan-mediated immune regulation. In broad terms, sialylated surfaces can be interpreted as self-associated molecular patterns. When inhibitory Siglecs engage these ligands, they can help restrain excessive immune responses and support tissue homeostasis. Altered sialylation may therefore reshape innate and adaptive immune behavior. This is why Siglec receptors are frequently discussed in studies of inflammation, autoimmunity, cancer biology, and host-pathogen interaction.
| Feature | General Principle | Why Researchers Measure It |
|---|---|---|
| Cell type expression | Different Siglecs are enriched on different immune cell populations. | Helps explain cell-specific responses to the same sialoglycan pattern. |
| Ligand selectivity | Binding depends on glycan linkage, density, and surrounding scaffold. | Essential for array design and antibody validation. |
| Cis and trans effects | Siglecs may bind ligands on the same cell or an opposing cell. | Important when modeling immune synapses and membrane organization. |
| Checkpoint-like signaling | Many Siglecs tune immune activation thresholds. | Useful in mechanistic studies of immune regulation. |
Where Sialic Acids Shape Experimental Outcomes
Sialic acid biology intersects with many experimental questions. Some projects begin with a descriptive question, such as whether a cell line carries a highly sialylated surface. Others begin with a functional question, such as why an immune receptor responds differently after glycoengineering. In both cases, strong study design depends on matching the analytical method to the biological question.
- Profiling terminal glycan composition on proteins, antibodies, and membrane-associated biomolecules
- Comparing alpha2,3 and alpha2,6 sialylation patterns across samples
- Testing receptor or antibody binding to defined sialosides
- Evaluating how membrane glycoediting changes recognition or signaling
- Generating custom anti-sialic acid reagents for detection and mechanistic studies
A recurring challenge is that sialic acid function depends on more than presence or absence. Linkage, density, local scaffold, neighboring sugars, and membrane presentation all matter. For this reason, one assay is rarely enough on its own. Higher-confidence studies often combine analytical characterization with functional binding assays and targeted reagent development.
Our Research Solutions for Sialic Acid Studies
Creative Biolabs provides research services that align with common experimental needs in sialic acid biology. These services are intended for investigators working on cell surface glycans, Siglec receptors, immune regulation, and cell-cell recognition in non-clinical research settings.
Anti-Sialic Acid Antibody Development
This service is suitable for projects that need selective reagents against sialylated targets or defined sialic acid-containing motifs. It can support detection, localization, comparative profiling, and mechanistic studies where uncontrolled glycan cross-reactivity would weaken interpretation.
Glycosylation Analysis Service
A suitable option for defining global or target-specific sialylation patterns. This service helps clarify whether the observed biology is associated with altered terminal glycan composition, glycan occupancy, or structure-dependent differences in sialylation.
Sialoside Microarray
Particularly relevant for receptor, lectin, or antibody binding studies. It enables high-content mapping against defined sialoside structures and is useful when binding specificity depends on linkage, branching, or nearby glycan context.
Custom Glycosylation of Cell Membranes
This service supports functional experiments that require controlled remodeling of membrane glycans. It is useful when researchers need to test how cell surface sialylation affects immune contact, ligand access, or receptor signaling in a more native-like setting.
Tell Us What You Need to Analyze
If your study involves sialylated glycoproteins, glycolipids, glycocalyx remodeling, or Siglec receptor binding, a focused service plan can save time during assay design. Share your sample type, target class, preferred readout, and whether your main goal is profiling, reagent generation, or functional validation. Our team can help match your project with a suitable research workflow.
Request a Sialic Acid Research Solution
Published Data
An open-access review published in 2021 summarized the role of the sialic acid-Siglec axis in human immune regulation. The authors described Siglecs as sialic acid-binding immunoglobulin-like lectins that are mainly expressed on blood cells in a cell type-specific manner, and noted that sialoglycans on mammalian cell surfaces contribute to self-recognition and immune regulation. The review also discussed the involvement of this axis in autoimmunity, cancer, and related therapeutic strategies.
As shown in Figure 1 of the review, inhibitory Siglecs commonly signal through ITIM-dependent recruitment of SHP-1 and SHP-2, whereas activating CD33-related Siglecs can signal through DAP12 and Syk. This figure connects glycan recognition with downstream signaling logic in a concise visual format.
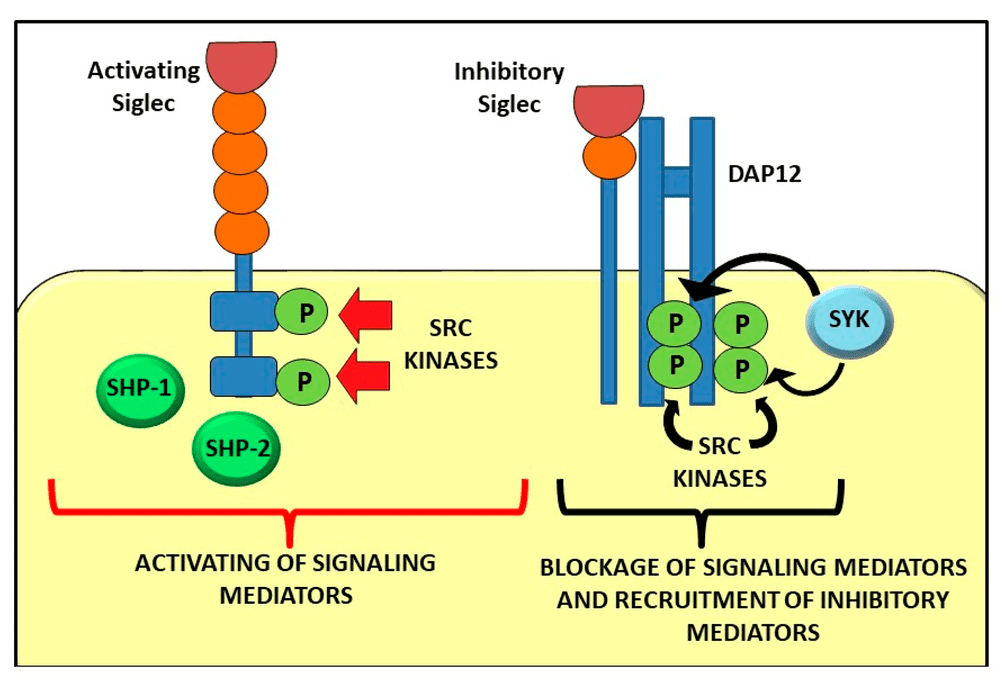 Fig.1 Signalling pathway mediated by activating and inhibitory Siglecs.1
Fig.1 Signalling pathway mediated by activating and inhibitory Siglecs.1
FAQs
Why are sialic acids often described as the face of the cell?
They often occupy terminal positions on glycans, so they are among the first molecular features encountered during cell-cell contact, receptor binding, and immune surveillance.
Why is terminal sialylation more influential than many internal glycan residues?
Because terminal residues directly affect surface charge, steric accessibility, and receptor engagement. A terminal change can alter recognition even when the rest of the glycan scaffold remains unchanged.
What makes Siglec receptors important in immune regulation?
Many Siglecs are expressed on immune cells and bind sialylated ligands in ways that tune activation thresholds. Inhibitory family members can help restrain excessive immune responses.
Is it enough to know that a sample is sialylated?
Usually not. Linkage type, density, scaffold, and neighboring sugars all influence biological behavior. Strong studies often combine structural and functional readouts.
When is a sialoside microarray the best choice?
It is especially useful when a project depends on distinguishing receptor or antibody binding among closely related sialylated glycan structures.
When should I choose glycosylation analysis instead of antibody development?
Choose glycosylation analysis when the main question is structural characterization of a sample. Choose antibody development when the main need is a custom detection or binding reagent for downstream experiments.
Can membrane glycoengineering help with functional studies?
Yes. Controlled remodeling of cell membrane glycans is valuable when you need to test whether altered sialylation changes receptor engagement, immune interaction, or cell-cell recognition in a biologically relevant format.
Reference:
- Gianchecchi, Elena, Andrea Arena, and Alessandra Fierabracci. "Sialic Acid-Siglec Axis in Human Immune Regulation, Involvement in Autoimmunity and Cancer and Potential Therapeutic Treatments." International Journal of Molecular Sciences 22.11 (2021): 5774. Distributed under Open Access license CC BY 4.0, without modification. https://doi.org/10.3390/ijms22115774
