Anti-Sialidase (Neuraminidase) Antibody Development Service
Sialidases, also commonly referred to as neuraminidases, are pivotal glycoside hydrolases that cleave terminal sialic acid residues from glycoproteins, glycolipids, and oligosaccharides. These enzymes play a dominant role in the pathogenesis of various infectious diseases, particularly in the life cycle of the influenza virus, where neuraminidase (NA) facilitates the release of progeny virions from infected host cells. At Creative Biolabs, we leverage our comprehensive Anti-Glycan Related Enzyme Antibody Development Service to provide high-specificity antibody solutions targeting various sialidase families. Our specialized platform is designed to generate monoclonal and polyclonal antibodies that effectively inhibit enzymatic activity, block viral transmission, and serve as critical tools for vaccine efficacy evaluation.
Scientific Background: Sialidases and Their Biological Impact
Sialidases are a diverse group of enzymes found across viruses, bacteria, protozoa, and mammals. They are biologically categorized into different families within the Carbohydrate-Active Enzymes (CAZy) database, primarily glycoside hydrolase families GH33 (bacteria, eukaryotic sialidases) and GH34 (viral neuraminidases).
Influenza Neuraminidase (NA)
The influenza neuraminidase is a homotetrameric glycoprotein anchored in the viral envelope. It functions by cleaving sialic acid receptors on the host cell surface, preventing the aggregation of newly formed viruses and enabling their spread to uninfected cells. Because the NA active site is relatively conserved compared to the highly variable hemagglutinin (HA) head, an influenza neuraminidase antibody that targets these conserved regions can offer broad protection. Research indicates that antibodies inhibiting NA enzymatic activity (neuraminidase inhibition, NAI) can reduce viral shedding and severity of disease, making them a significant focus for universal vaccine strategies.
Mammalian and Bacterial Sialidases
Beyond viruses, mammalian sialidases (NEU1, NEU2, NEU3, NEU4) regulate cellular signaling and catabolism. For instance, specific detection with a NEU1 antibody or NEU3 antibody is essential for studying lysosomal storage diseases and cancer progression. Similarly, a bacterial sialidase antibody is critical for investigating the virulence mechanisms of pathogens like Clostridium perfringens or Streptococcus pneumoniae, which use sialidases to strip host mucosal defenses.
Antigenic Drift
The surface proteins of influenza viruses constantly mutate. Developing a viral neuraminidase antibody that maintains efficacy across drifting strains requires targeting structurally conserved epitopes.
Structural Stability
Recombinant NA antigens often differ structurally from the native viral tetramer. Antibodies raised against unstable monomers may fail to recognize the functional tetramer on the virus surface.
Assay Complexity
Traditional Hemagglutination Inhibition (HAI) assays do not measure NA-based immunity. Specialized Enzyme-Linked Lectin Assays (ELLA) are required to properly validate anti-neuraminidase antibody functionality.
Sequence Homology
Distinguishing between human endogenous sialidases and pathogen-derived enzymes requires rigorous counter-screening to prevent off-target binding in diagnostic or therapeutic development research.
Comprehensive Anti-Sialidase Antibody Development Services
Creative Biolabs offers a modular service pipeline capable of generating antibodies against a wide spectrum of sialidase targets. Whether you require a sialidase antibody for structural biology, a neutralizing antibody for viral research, or a detection reagent for bacterial pathogenesis, our team provides tailored solutions.
Anti-Influenza Neuraminidase Antibody Development
We specialize in developing antibodies targeting Influenza A (N1-N9 subtypes) and Influenza B NA. Our strategy involves using tetrameric stabilized antigens or Virus-Like Particles (VLPs) to ensure the antibodies recognize the native quaternary structure. We can generate broadly reactive antibodies capable of inhibiting enzymatic activity across multiple viral strains, aiding in the evaluation of next-generation influenza vaccines.
Mammalian Sialidase Antibody Production
Our platform supports the generation of highly specific antibodies against human neuraminidases, including NEU1, NEU2, NEU3, and NEU4 antibody development. These reagents are validated for use in flow cytometry, western blotting, and immunohistochemistry (IHC) to study sialidase expression patterns in oncology and metabolic disease research.
Bacterial & Parasitic Sialidase Antibodies
We provide development services for antibodies against microbial sialidases (e.g., NanA, NanB from streptococci). These anti-glycosidase antibody products are crucial for dissecting the role of sialidase in biofilm formation, host colonization, and immune evasion mechanisms.
Functional Assay Development
Beyond antibody generation, we offer downstream characterization services. This includes neuraminidase inhibition (NAI) assays using ELLA, kinetic analysis of enzymatic blockage, and transmission blocking assays in relevant animal models (e.g., guinea pig transmission models for influenza).
Development Workflow
Inquire About Sialidase Antibodies
Why Choose Creative Biolabs?
Broad Reactivity
Expertise in generating antibodies that target conserved epitopes across different viral subtypes or enzyme families.
Functional Guarantee
Screening processes specifically designed to identify clones with high Neuraminidase Inhibition (NAI) titers.
Advanced Platforms
Utilization of Hybridoma, Phage Display, and Single B Cell sorting technologies for diverse antibody formats (IgG, Fab, VHH).
Data-Driven Insights
Comprehensive characterization including kinetic affinity, epitope binning, and cross-protection analysis.
Published Data
A recent study highlights the critical role of structure-based antibody engineering in overcoming antigenic drift in influenza neuraminidase (NA). The research focused on a broadly reactive human monoclonal antibody, Z2B3, which was originally isolated from a patient infected with H7N9. While Z2B3 effectively neutralized diverse N1 and N9 subtypes, it lost potency against contemporary H1N1 seasonal strains carrying a specific K432E mutation in the NA active site. Comprehensive structural analysis of the antibody-antigen complex revealed that this specific loss of function was primarily due to the disruption of a key salt bridge interaction between residue D102 of the antibody's heavy chain and residue 432 of the NA active site rim. By leveraging this precise structural insight, researchers engineered a targeted variant of the antibody, designated Z2B3-D102R, to reverse the charge complementarity and re-establish the critical salt bridge. This single amino acid modification successfully restored high-affinity binding and enzymatic inhibition against the drifted N1 targets. Furthermore, in vivo prophylactic studies demonstrated that while the wild-type antibody failed to protect subjects from the variant virus, the engineered Z2B3-D102R variant conferred 100% protection against lethal challenge. This compelling data underscores the immense potential of rational design strategies in developing next-generation anti-sialidase antibodies capable of adapting to continuous viral evolution.
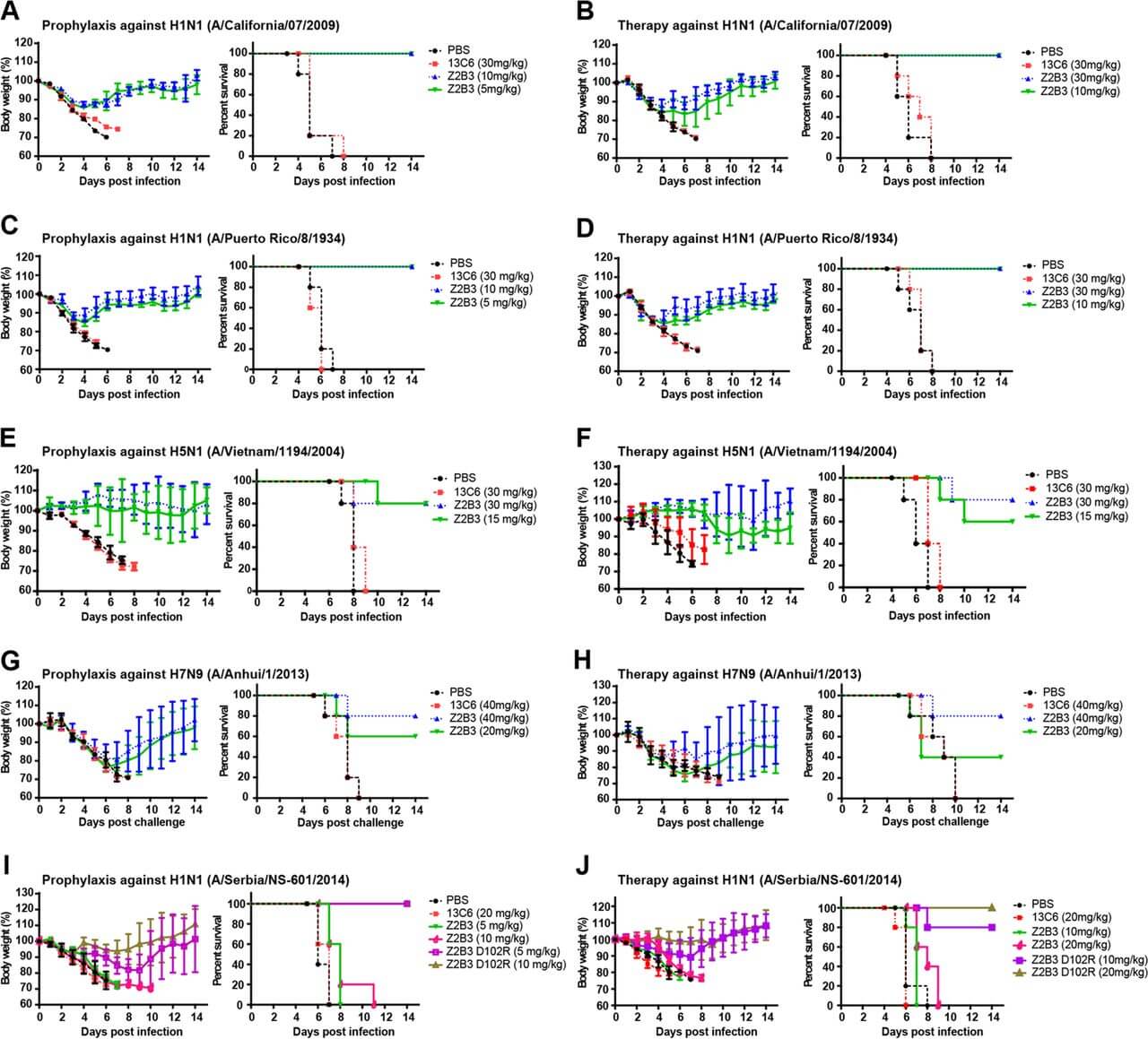 Fig.1
Restoration of protective efficacy against drifted H1N1 virus by structure-based antibody modification.1
Fig.1
Restoration of protective efficacy against drifted H1N1 virus by structure-based antibody modification.1
FAQs
What is the difference between HAI and NAI antibodies?
HAI (Hemagglutination Inhibition) antibodies target the Hemagglutination (HA) head and block receptor binding. They are the standard correlate of protection but are strain-specific. NAI (Neuraminidase Inhibition) antibodies target the NA enzyme, preventing viral release (budding) rather than entry. NAI antibodies reduce disease severity and viral shedding and are often more broadly reactive across drifted strains.
Can you generate antibodies that distinguish between NEU1, NEU2, NEU3, and NEU4?
Yes. Mammalian sialidases share some homology but have distinct structural features. We utilize counter-screening strategies where candidate clones are tested against a panel of all four human sialidase recombinant proteins. Only clones that bind the specific target (e.g., NEU3 antibody) and do not cross-react with the others are selected for development.
Do you offer antibodies for bacterial sialidases?
Yes, we develop antibodies against bacterial sialidases such as those from Clostridium perfringens (NanI, NanJ) or Streptococcus pneumoniae (NanA). A bacterial sialidase antibody is useful for studying bacterial nutrition acquisition (sialic acid scavenging) and pathogenicity.
What assay formats do you use to validate anti-NA antibodies?
We primarily use the Enzyme-Linked Lectin Assay (ELLA), which is the gold standard for measuring neuraminidase inhibition titers. We also perform standard ELISA for binding affinity and can support plaque reduction assays or virus yield reduction assays to confirm biological neutralization.
Are your anti-sialidase antibodies suitable for therapeutic development?
Our custom antibody development services are primarily for research use. However, we can perform humanization and affinity maturation services to generate lead candidates that possess the characteristics required for therapeutic discovery and preclinical research (e.g., high affinity, low immunogenicity, broad breadth).
What Our Customers Say
"We needed a pair of antibodies for a sandwich ELLA to detect N1 neuraminidase. Creative Biolabs designed a strategy using recombinant tetramers that worked perfectly. The resulting antibodies had excellent sensitivity and no cross-reactivity with N2."
"The team helped us generate VHH antibodies against bacterial sialidases. The small size of the VHHs allowed them to access the catalytic cleft much better than conventional IgGs. Highly recommended for structural biology projects."
"Validating anti-NEU1 antibodies for flow cytometry is notoriously difficult due to specificity issues. Creative Biolabs performed rigorous knockout cell line validation for us, giving us total confidence in the data."
"Professional service with detailed reports. The breadth of reactivity analysis for our influenza B NA antibody was comprehensive, covering both Yamagata and Victoria lineages as requested."
Reference:
- Jiang, H., et al. "Structure-based modification of an anti-neuraminidase human antibody restores protection efficacy against the drifted influenza virus." mBio 11.5 (2020): e02315-20. Distributed under Open Access license CC BY 4.0, without modification. https://doi.org/10.1128/mBio.02315-20
Supports
- Glycosyltransferase & Glycosidase Substrate Microarray
- Glycosylation Analysis
- Custom Glycosylation of Biomolecules
