Fucosyltransferases (FUTs) and ABO Blood Group Antigens
Creative Biolabs supports glycan-focused research on fucosyltransferase function, histo-blood group antigen biosynthesis, and glycan-recognition tools through our Anti-Fucosyltransferase (FUT) Antibody Development Service. For investigators studying how the fut gene family controls ABO blood group synthesis and Lewis antigens, a clear view of enzymatic order, substrate preference, and tissue distribution is essential before assay design, target validation, or glycan profiling.
Why FUT Enzymes Matter in Blood Group Glycobiology
ABO and Lewis antigens are carbohydrate determinants assembled stepwise on precursor chains. They are not produced by a single enzyme. Instead, their final structures reflect coordinated activity among specific fucosyltransferases and the ABO glycosyltransferases. This is why the same individual may express distinct blood group related glycans on erythrocytes, epithelial surfaces, and secretions.
For research teams, FUT biology matters at three levels. First, it explains how H antigens are generated and then converted into A or B structures. Second, it clarifies why Lewis phenotypes depend strongly on FUT2 and FUT3 status. Third, it provides a mechanistic bridge between normal glycobiology and altered fucosylation patterns reported in inflammation, host-microbe interaction, and cancer-associated glycan remodeling.
| Key Concept | Main Enzyme or Gene | Primary Substrate Context | Major Product Outcome |
|---|---|---|---|
| H antigen initiation | FUT1 or FUT2 | Type 2 or type 1 precursor chain | H type 2 or H type 1 structure |
| Lewis epitope formation | FUT3 | Type 1 or type 2 precursor, or H-modified chain | Lea, Leb, Lex, or Ley |
| ABO conversion | ABO glycosyltransferases | H antigen backbone | A or B antigen formation |
| Secretor phenotype control | FUT2 | Mucosal tissues and body fluids | Secreted H, A, B, and related Lewis-active glycans |
Core Background on Precursor Chains and Fucose Linkage
Understanding FUT-dependent blood group synthesis starts with the precursor chain. The core acceptor is typically a lactosamine-related structure that can be extended and then decorated. Two chain classes are especially important.
- Type 1 chain: Galβ1-3GlcNAc. This chain is highly relevant in secretions and mucosal tissues.
- Type 2 chain: Galβ1-4GlcNAc. This chain is prominent on erythrocytes and many cell-surface glycoconjugates.
The distinction is not cosmetic. It changes enzyme preference and therefore the final antigen spectrum. FUT1 preferentially acts on type 2 precursors, whereas FUT2 is more active on type 1 precursors. FUT3 can install fucose in α1,4 linkage on type 1 chains and in α1,3 linkage on type 2 chains, enabling Lewis antigen formation.
In practical terms, this means that the same biosynthetic system can generate different outputs depending on chain type, cell lineage, and gene status. A laboratory analyzing a glycoprotein sample, a glycolipid fraction, and a mucosal secretion may therefore detect related but not identical antigen repertoires.
What FUT1 Primarily Does
FUT1 adds α1,2-linked fucose mainly to type 2 precursors. This step is central to H antigen generation on erythroid cells and supports downstream A or B antigen formation on type 2 chains.
What FUT2 Primarily Does
FUT2 also creates α1,2-fucosylated H structures, but it is especially important on type 1 chains in secretory epithelia and body fluids. This is why FUT2 strongly influences secretor status.
What FUT3 Primarily Does
FUT3 is the major Lewis-active enzyme. It can create Lea or Lex directly from precursor chains and can also act on H-bearing chains to yield Leb or Ley.
Where ABO Enzymes Fit In
ABO transferases do not create the initial H determinant. They modify preformed H antigen by adding terminal sugars that define A or B specificity on the appropriate backbone.
The FUT Gene Family
The broader human FUT family contains several enzymes with distinct linkage specificities and biological settings. In the context of ABO and Lewis biosynthesis, three members are the principal focus: FUT1, FUT2, and FUT3. Their combined action explains much of the classic histo-blood group diversity seen across tissues.
FUT1 and FUT2 both encode α1,2-fucosyltransferases, but their acceptor preference and tissue distribution are not interchangeable. FUT1 is closely tied to erythrocyte-associated H antigen synthesis. FUT2 is the main secretor pathway enzyme and shapes blood group related glycans in saliva, gastrointestinal secretions, and epithelial surfaces. FUT3 provides α1,3/4 fucosyltransferase activity required for the most widely discussed Lewis antigens.
This division of labor also explains common genotype-phenotype questions in glycan research. A functional ABO locus alone does not guarantee broad A or B expression in secretions. Without FUT2 activity, type 1 H antigen formation in secretory tissues is impaired. Likewise, Lewis antigen output depends strongly on FUT3 functionality and on whether α1,2-fucosylated intermediates are available for further conversion.
Research Points Frequently Examined
- Secretor versus non-secretor status: usually studied through FUT2 genotype or enzyme activity.
- Lewis-positive versus Lewis-negative patterns: dependent on FUT3 functionality and influenced by whether FUT2-generated H intermediates are available.
- Substrate competition and sequence: whether FUT2 or FUT3 acts first can alter the final glycan set.
- Tissue specificity: erythrocytes and epithelial tissues do not present identical precursor availability.
- Fucosylation in cancer: altered FUT expression can reshape glycan presentation beyond classical blood group contexts.
How FUTs Determine ABO and Lewis Antigen Synthesis
The simplest way to frame the pathway is as an ordered branching system.
- A precursor chain is present in either type 1 or type 2 form.
- FUT1 or FUT2 adds α1,2-linked fucose to produce an H antigen, depending on substrate type and tissue context.
- The ABO enzyme can then convert H into A or B antigen by adding the corresponding terminal monosaccharide.
- FUT3 can act either on the original precursor chain or on an H-modified intermediate, generating Lewis-active structures.
This branching logic explains the canonical relationships:
- Type 1 precursor + FUT3 can yield Lea.
- Type 1 precursor + FUT2 yields H type 1, which can be converted by FUT3 to Leb.
- Type 2 precursor + FUT3 can yield Lex.
- Type 2 precursor + FUT1 yields H type 2, which can be converted to A or B type 2 by ABO enzymes and can also support Ley-related structures through FUT3 activity.
Because Lewis synthesis depends on both precursor access and upstream fucosylation status, phenotype interpretation often requires more than one gene. This is particularly relevant in projects that compare glycan phenotypes across donor cohorts, tissue models, organoids, engineered cell lines, or cancer samples.
Why This Topic Extends Beyond Classical Blood Typing
FUT-dependent glycans are not limited to transfusion biology. The same enzymatic logic influences mucosal colonization, host-pathogen recognition, and tissue-specific glycan landscapes. In cancer research, altered expression of FUT genes can change terminal fucosylation patterns on glycoproteins and glycolipids, sometimes increasing Lewis-related epitopes or other fucosylated determinants that affect adhesion, signaling, or immune recognition.
For this reason, researchers often study FUT pathways in settings such as:
- cell line engineering for secretor and non-secretor models
- glycoprotein and glycolipid antigen discovery
- lectin or antibody specificity evaluation
- host-microbe interaction studies involving mucosal glycans
- comparative profiling of normal and tumor-associated fucosylation
Importantly, the services discussed on this page are provided for research use only. They are intended to support mechanistic, analytical, and discovery-oriented studies rather than clinical diagnosis or treatment.
Our Solutions for FUT and Blood Group Antigen Research
Creative Biolabs offers a focused workflow for teams working on FUT enzymes, glycan epitope mapping, and blood group related carbohydrate analysis. Depending on your project goal, we can support target reagent generation, glycan profiling, or gene-to-glycome correlation studies.
Anti-Fucosyltransferase (FUT) Antibody Development
This service is well suited for teams studying FUT protein expression, localization, isoform-specific detection, or assay development around FUT1, FUT2, FUT3, and related family members. It is especially useful when commercial reagents do not match the required species, epitope region, or application format.
Glycosylation Analysis Service
For investigators who need direct structural evidence, this service supports profiling of fucosylated glycans, H antigen-related structures, and glycosylation shifts across samples. It is valuable for mapping how enzyme perturbation changes the final glycome.
ABH-Glycan Microarray
This platform helps characterize binding preferences toward ABH-related glycans and closely related blood group structures. It is useful for antibody screening, lectin profiling, receptor interaction studies, and cross-reactivity assessment across blood group motifs.
Glyco-gene Microarray
When a project focuses on the fut gene family rather than glycan output alone, this service supports expression-level investigation of glyco-genes that contribute to terminal glycan biosynthesis. It is useful for correlating gene signatures with antigen presentation patterns.
How These Services Fit Common Project Designs
- Protein-focused study: antibody development plus glyco-gene expression analysis
- Structure-focused study: glycosylation analysis plus ABH-glycan microarray
- Mechanism-focused study: FUT antibody generation plus glycan profiling before and after gene perturbation
- Cancer glycan study: comparative profiling of FUT expression and terminal fucosylated epitopes across model systems
Talk to Us About Your FUT Project
If your team is planning a study on FUT enzyme specificity, secretor-related glycan expression, Lewis antigen mapping, or blood group associated glycoanalysis, send us your target, sample type, preferred application, and expected readout. We can help align antibody development, microarray screening, and glycosylation analysis into a practical research workflow with clear deliverables.
Request a Quote for FUT Research Support
Published Data
A 2022 open-access review in Frontiers in Microbiology provides a concise overview of histo-blood group antigen biosynthesis, showing that FUT1 and FUT2 install α1,2-linked fucose on type 2 and type 1 precursor chains, respectively, to generate H determinants that can be further converted into A or B antigens by ABO glycosyltransferases. The review also outlines how FUT3 acts on precursor or H-modified chains to produce Lewis-related structures, including Lea, Leb, Lex, and Ley. The figure below serves as a useful visual summary for researchers studying secretor status, mucosal glycobiology, and glycan-dependent host-microbe interactions because it places type 1 and type 2 precursor chains within one framework and clarifies where FUT1, FUT2, FUT3, and ABO enzymes act.
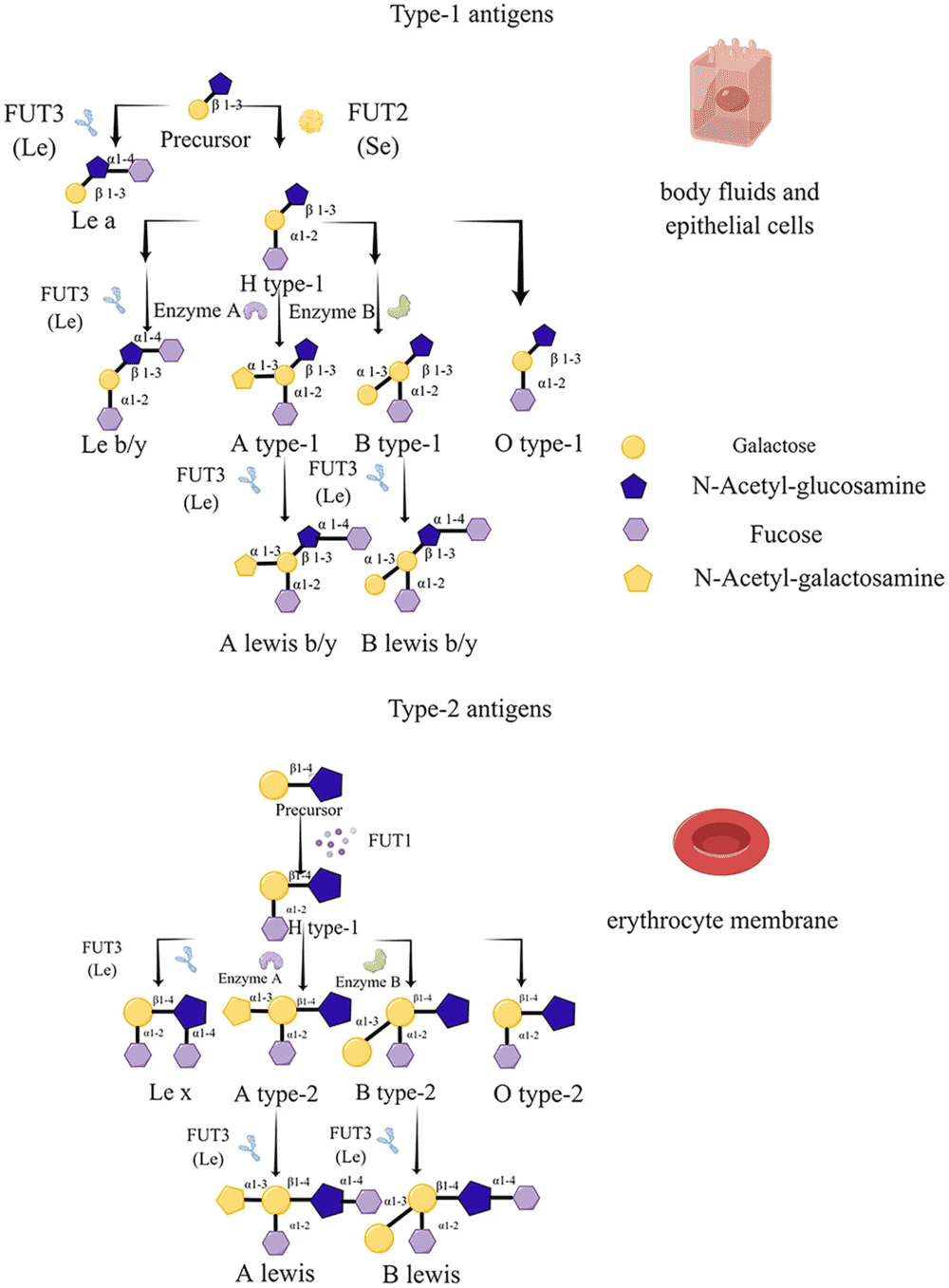 Fig.1 FUT-mediated biosynthesis routes of ABO and Lewis antigens.1
Fig.1 FUT-mediated biosynthesis routes of ABO and Lewis antigens.1
FAQs
Which FUT enzymes are most directly involved in ABO and Lewis antigen synthesis?
FUT1, FUT2, and FUT3 are the key fucosyltransferases in this setting. FUT1 and FUT2 generate H determinants through α1,2 fucosylation, while FUT3 provides the α1,3 or α1,4 fucosylation needed for major Lewis antigens.
Why is FUT2 so important for secretor status?
FUT2 is the major α1,2-fucosyltransferase acting on type 1 precursor chains in secretory tissues and body fluids. When FUT2 is inactive, secreted H, A, and B related structures are reduced or absent despite preserved erythrocyte blood group expression driven by other pathways.
How does FUT3 affect the Lewis phenotype?
FUT3 introduces the α1,3 or α1,4 fucose linkages required for major Lewis antigens. Its activity is essential for Lea, Leb, Lex, and Ley formation, although the final Lewis pattern can also depend on whether FUT2-generated H intermediates are available.
Do ABO enzymes and FUT enzymes perform the same reaction?
No. FUT enzymes transfer fucose, whereas ABO glycosyltransferases transfer the terminal sugar that defines A or B specificity. ABO conversion depends on a pre-existing H determinant, which is why FUT activity is upstream of final ABO antigen formation.
Which service is most useful if I need to profile terminal fucosylated glycan structures?
Our Glycosylation Analysis Service is usually the best starting point for direct structural profiling. It can be complemented by ABH-Glycan Microarray analysis when binding specificity or cross-reactivity across blood group related glycans also needs to be tested.
Can your team support studies on FUT expression changes in cancer models?
Yes. We can support FUT-focused antibody development, glyco-gene expression studies, and glycosylation profiling for projects investigating altered terminal fucosylation in tumor cell lines, engineered models, or comparative sample sets.
Are these services intended for clinical blood typing or diagnosis?
No. The services described on this page are provided for research use only and are designed for scientific investigation of glycobiology, glycan analysis, and reagent development.
Reference:
- Hu, Mingyang, Xiyun Zhang, Jinze Li, Luotong Chen, Xiaolin He, and Tingting Sui. Fucosyltransferase 2: A Genetic Risk Factor for Intestinal Diseases. Frontiers in Microbiology 13 (2022): 940196. Distributed under Open Access license CC BY 4.0, without modification. https://doi.org/10.3389/fmicb.2022.940196
