Plant Glycans and Allergy: The Role of Cross-Reactive Carbohydrate Determinants (CCDs)
Creative Biolabs supports plant glycan and allergy research with integrated solutions for antibody generation, glycan profiling, plant glycoprotein characterization, and N-glycan mapping. For groups investigating cross-reactive carbohydrate determinants, our Anti-Plant and Algal Glycan Antibody Development service provides a practical starting point for building selective research tools against plant-associated glycoepitopes. These services are supplied for research use only and are not intended for clinical diagnosis or treatment.
Why Plant Glycans Matter in Allergy Research
Plant allergens are often discussed as proteins, yet many plant allergens are glycoproteins. Their attached glycans can influence how serum IgE binds in immunoassays, how broad apparent sensitization patterns look, and how difficult it becomes to interpret positive in vitro findings. In this context, cross-reactive carbohydrate determinants, often abbreviated as CCDs, remain one of the most important topics in glycoallergy research.
CCDs are carbohydrate motifs carried by glycoproteins from plants and several non-mammalian sources. They are widely recognized because they can bind IgE from sensitized individuals across many unrelated allergen extracts. This broad recognition pattern explains why one serum sample may react with pollen, plant foods, and other glycoprotein-containing materials even when the underlying protein backbones are not closely related.
For researchers, the key question is not simply whether IgE binds. The important question is what the IgE is binding to. If the signal is driven mainly by shared glycan motifs rather than protein-specific epitopes, the biological interpretation changes. This is why plant glycan structure analysis, careful reagent selection, and glycan-focused validation are essential.
Shared Across Sources
CCD-bearing glycans are not restricted to one allergen family. Similar motifs can occur on many plant glycoproteins, which increases the chance of broad IgE binding patterns.
Relevant to Assay Readouts
A positive in vitro signal may reflect recognition of a carbohydrate determinant rather than a unique protein allergen epitope.
Driven by Non-Human Glycofeatures
Plant complex N-glycans contain motifs absent from mammalian glycoproteins, which helps explain why they can be immunologically visible.
Important for Specificity Control
Glycan-aware assay design reduces misinterpretation and supports more rigorous comparison between extracts, purified glycoproteins, and glycan-defined materials.
Plant Glycan Structure Behind CCD Recognition
The most relevant plant CCD motifs are usually discussed in the context of plant complex N-glycans. Two structural features are especially important: core α1,3-fucose and β1,2-xylose. These residues are attached to the conserved N-glycan core and distinguish many plant glycoproteins from mammalian counterparts.
In practical terms, a plant glycoprotein can carry a shared glycan signature that becomes the target of IgE. Once serum IgE recognizes that signature, similar reactivity may appear across unrelated glycoproteins that carry comparable N-glycan decorations. This is the structural basis of much observed IgE cross-reactivity linked to CCDs.
| Structural feature | Typical context | Why it matters in CCD research | Research implication |
|---|---|---|---|
| Core α1,3-fucose | Plant complex N-glycans | Frequently described as a major non-self determinant in plant and insect glycoproteins | Important for mapping serum binding and assay interference |
| β1,2-xylose | Plant complex N-glycans | Commonly accompanies plant-specific glycan cores and contributes to shared glycoepitope patterns | Useful when comparing glycoprotein panels and glycan-defined arrays |
| MMXF and related motifs | Representative CCD-bearing glycan structures | Provide a defined model for studying IgE recognition of plant-type glycans | Suitable for targeted glycan microarray and antibody validation workflows |
| Plant glycoprotein presentation | Pollen, foods, extracts, purified proteins | Glycan density and molecular context can influence apparent binding intensity | Supports side-by-side analysis of structure and presentation format |
Key Points for Interpreting Plant Glycoprotein Reactivity
- Not every positive IgE result points to a protein-specific response.
- Shared glycan motifs can create broad patterns of apparent polysensitization.
- Plant extracts vary in glycoprotein composition and glycan presentation.
- Defined glycan and glycoprotein tools are more informative than crude extract comparison alone.
How CCDs Contribute to IgE Cross-Reactivity
IgE cross-reactivity occurs when one antibody population recognizes structurally similar epitopes on different molecules. In CCD allergy research, the shared epitope is not primarily the peptide backbone but the glycan signature. If serum IgE recognizes plant complex N-glycans carrying core α1,3-fucose and, in many cases, β1,2-xylose, the same serum may react with multiple glycoproteins from distinct botanical sources.
This is one reason why bromelain and other glycoprotein-rich materials have historically been used as reference tools when discussing CCD reactivity. They help illustrate that the carbohydrate determinant itself can explain broad in vitro binding. For researchers building assays, the lesson is direct: specificity controls must include glycan-aware negative and positive comparators.
It is also important to distinguish analytical cross-reactivity from biological relevance. In the literature, anti-CCD IgE is widely recognized as a cause of extensive in vitro cross-reactivity and as a source of complexity in serological interpretation. At the same time, anti-CCD IgE is generally considered to have low clinical relevance when CCDs are assessed in isolation. The structure, carrier, density, and detection format still matter for data interpretation.
At the Glycan Level
Shared plant-type N-glycans create repeated carbohydrate motifs across otherwise unrelated glycoproteins.
At the Assay Level
ELISA, blotting, and multiplex formats may all detect IgE binding to carbohydrate determinants unless carefully controlled.
At the Interpretation Level
Structure-resolved workflows help separate plant glycan reactivity from protein-specific recognition and support cleaner conclusions.
What Researchers Often Need to Clarify
- Which glycan motifs are present on the study material
- Whether IgE binding follows plant glycan structure rather than allergen family
- Whether a plant glycoprotein panel reproduces the same reactivity pattern as a defined glycan panel
- Whether anti-glycan antibodies or lectin-based tools can verify the suspected glycofeatures
Our Research Solutions for Plant Glycans and CCD Studies
Creative Biolabs provides a focused service portfolio for groups studying cross-reactive carbohydrate determinants, plant glycoprotein characterization, and glycan-mediated IgE cross-reactivity. The following services are especially suitable for this topic because they move from epitope recognition to structural confirmation and platform-based comparison.
Anti-Plant and Algal Glycan Antibody Development
This service is highly relevant when your project needs selective binders against plant-associated glycoepitopes. It is useful for reagent generation, glycan-focused assay development, comparative screening, and mechanistic studies involving plant glycoprotein recognition.
100 N-Glycan Microarray
A defined N-glycan array is well suited to dissect plant glycan structure and identify whether serum or antibody binding tracks with plant-type complex glycan motifs. It supports comparative profiling of CCD-associated recognition patterns.
Glycoprotein Microarray
This platform is valuable when you need to test recognition in a more native presentation format. It helps compare plant glycoprotein samples side by side and determine whether observed IgE cross-reactivity remains consistent at the glycoprotein level.
N-glycosylation Analysis of Proteins
Structural confirmation remains essential in CCD research. This service supports mapping of plant-type N-glycans, helping researchers verify the presence of core fucose, xylose-containing motifs, and other features relevant to glycan-based cross-reactivity.
How These Services Work Together
- Start with plant glycoprotein or defined glycan selection
- Profile recognition patterns with glycan or glycoprotein microarrays
- Confirm structural features by targeted N-glycosylation analysis
- Develop glycan-selective antibodies for downstream verification and tool building
Tell Us What You Need to Compare
If your study involves pollen glycoproteins, plant food glycoallergens, reference CCD reagents, or custom glycan-binding antibody generation, our team can help define a research workflow around your sample type and assay objective. You can send us your target material, preferred readout, and the level of structural resolution you need. We will suggest a practical combination of microarray, glycosylation, and antibody-development options.
Request a Plant Glycan Research Strategy
Published Data
A relevant open-access review for this topic is the 2020 Frontiers in Immunology article The History of Carbohydrates in Type I Allergy. Although the paper discusses carbohydrate allergens more broadly, it specifically states that many allergens, especially those from the plant kingdom, possess common N-glycosidically linked immunogenic carbohydrate determinants with IgE-binding properties. It also outlines the historical development of the CCD concept.
The figure below presents the representative non-self CCD structures MMXF and MUXF. It shows the conserved N-glycan core together with the two plant-associated modifications most often discussed in CCD research, core α1,3-fucose and β1,2-xylose. This figure is useful for illustrating the structural basis of broad in vitro IgE cross-reactivity associated with plant-type N-glycans.
The review also notes that anti-CCD IgE is a major source of analytical cross-reactivity, while generally being considered of limited clinical relevance when CCDs are assessed alone. That distinction is important when interpreting plant glycoprotein binding data and related serological readouts.
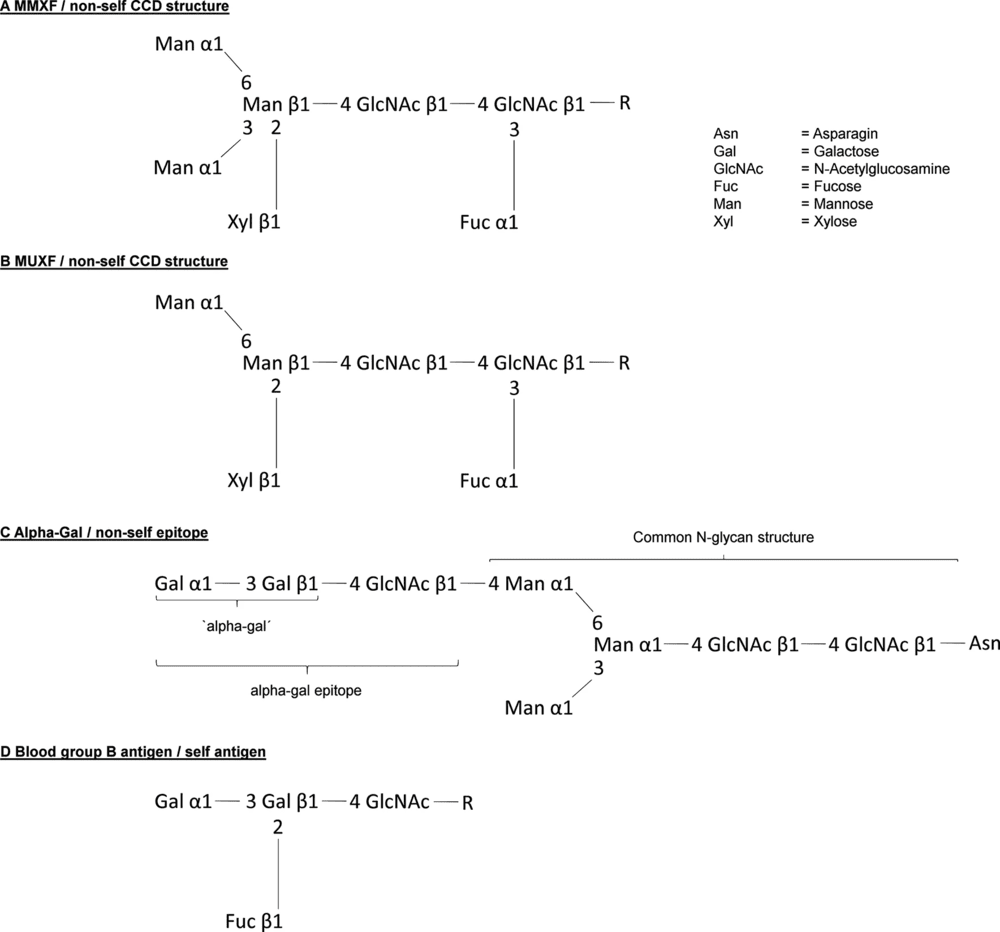 Fig.1 Representative CCD structures MMXF and MUXF on non-self N-glycans.1
Fig.1 Representative CCD structures MMXF and MUXF on non-self N-glycans.1
FAQs
What are cross-reactive carbohydrate determinants in plant allergy research?
Cross-reactive carbohydrate determinants are shared glycan motifs carried by glycoproteins from plants and other non-mammalian sources. They are important because serum IgE can recognize these motifs across many unrelated allergen preparations.
Why are core α1,3-fucose and β1,2-xylose discussed so often in CCD allergy?
These residues are characteristic features of many plant complex N-glycans and are central to the structural explanation of plant-associated CCD recognition.
Does a positive IgE signal to a plant extract always mean protein-specific sensitization?
No. A positive signal can be influenced by glycan recognition, especially when glycoprotein-rich materials carry shared carbohydrate determinants. This is why glycan-aware controls are important.
Which service is best if I want to map CCD-related binding patterns first?
A defined N-glycan microarray is usually the most direct option for early-stage binding pattern analysis because it separates glycan recognition from complex sample background.
When should I use glycoprotein microarrays instead of glycan microarrays?
Glycoprotein microarrays are useful when presentation context matters and you want to compare recognition of intact plant glycoproteins rather than isolated glycan motifs alone.
Can Creative Biolabs help validate whether my target material carries plant-type N-glycans?
Yes. Our N-glycosylation analysis workflow can support structural confirmation of plant-type glycan features relevant to CCD-focused studies.
Reference:
- Hils, Miriam, Florian Wölbing, Christiane Hilger, Jörg Fischer, Nils Hoffard, and Tilo Biedermann. The History of Carbohydrates in Type I Allergy. Frontiers in Immunology 11 (2020): 586924. Distributed under Open Access license CC BY 4.0, without modification. https://doi.org/10.3389/fimmu.2020.586924
