Neu5Gc vs. Neu5Ac: Evolution, Diet, and Cancer Risk
Creative Biolabs supports Neu5Gc-focused research with specialized anti-Neu5Gc antibody development services for scientists investigating glycan discrimination, xeno-autoantigen biology, chronic inflammation, and tumor-associated sialylation. In the neu5gc vs neu5ac discussion, the core issue is not simply one extra oxygen atom. It is how a small structural difference became a defining feature of human sialic acid evolution, shaped host biology, altered immune recognition, and created a research framework linking diet-derived glycans to inflammation and cancer-associated processes.
Why Neu5Gc and Neu5Ac Matter in Glycan Research
Sialic acids are terminal acidic monosaccharides commonly found on glycoproteins, glycolipids, and other glycoconjugates. Because they occupy outermost positions on cell surfaces and secreted molecules, they influence receptor binding, molecular half-life, immune recognition, cell-cell interaction, and pathogen engagement. Among the many sialic acid variants identified in nature, Neu5Ac and Neu5Gc are the two major mammalian forms most often discussed in biomedical literature.
Neu5Ac is the predominant human sialic acid. Neu5Gc is widely present in most mammals but is not synthesized in normal human cells because the human lineage lost a functional CMAH enzyme. That difference has broad implications for glycobiology, immunology, oncology, nutrition research, and antibody development.
| Feature | Neu5Ac | Neu5Gc |
|---|---|---|
| Full name | N-acetylneuraminic acid | N-glycolylneuraminic acid |
| Human endogenous synthesis | Yes | No in normal humans |
| Structural difference | Acetyl group at C-5 | Glycolyl group at C-5 |
| Key enzyme relationship | Substrate pool retained in humans | Normally generated from Neu5Ac by CMAH in most mammals |
| Immunological status in humans | Self-associated sialic acid | Non-human glycan and potential xeno-autoantigen |
| Typical research interest | Human glycosylation, signaling, receptor biology | Dietary incorporation, chronic inflammation, tumor glycans, anti-Neu5Gc immunity |
Sialic Acid Evolution and the Human Loss of Neu5Gc
A central event in sialic acid evolution was the inactivation of the human CMAH gene. In most mammals, CMAH converts CMP-Neu5Ac into CMP-Neu5Gc. In humans, this pathway is disrupted, so Neu5Ac became the dominant endogenous sialic acid. This evolutionary shift changed the molecular composition of the human glycocalyx and has been discussed as relevant to human-specific immune recognition and other aspects of glycobiology.
From a mechanistic standpoint, the neu5gc vs neu5ac comparison is therefore a comparison between a retained human self-glycan and a lost ancestral mammalian glycan. The difference is subtle at the chemical level yet highly meaningful at the biological level.
What Changed During Human Evolution
Humans lost the ability to synthesize Neu5Gc because of a disabling mutation in the CMAH gene. This made Neu5Ac the dominant sialic acid form on human cells and secreted glycoconjugates.
Why the Change Matters
Once Neu5Gc was no longer a normal self-structure, it became immunologically foreign when introduced from external sources. That creates the foundation for anti-Neu5Gc antibody responses and xeno-autoantigen biology.
Research Implications of This Evolutionary Split
- Human-specific glycan biology cannot be inferred directly from standard mammalian systems.
- Neu5Gc-containing reagents, sera, tissues, and biomaterials may introduce non-human glycan variables.
- Antibody specificity studies must distinguish Neu5Gc from closely related Neu5Ac-bearing structures.
- Tumor glycan analysis requires careful interpretation when non-human sialic acid is detected.
Dietary Neu5Gc Exposure and Tissue Incorporation
Although humans do not synthesize Neu5Gc, they can acquire it from exogenous sources. The literature1 most often discusses red meat and certain dairy products as dietary sources. In this context, Neu5Gc is studied as one diet-derived glycan that can be metabolically incorporated into human glycoconjugates.
After uptake, exogenous Neu5Gc can enter salvage pathways and become incorporated into cell surface and secreted glycans. In human tissues, incorporated Neu5Gc is then displayed in a foreign context: it is chemically mammalian, but immunologically non-self to humans. That is why Neu5Gc is often described as a xeno-autoantigen.
Key Points About Dietary Incorporation
- Humans lack normal endogenous Neu5Gc synthesis.
- Neu5Gc can still appear in human tissues through external uptake.
- Red meat is the most commonly discussed dietary source.
- Incorporated Neu5Gc may be detected on glycoproteins and glycolipids.
- Accumulation can be more evident in some tumors than in surrounding normal tissues.
How Neu5Gc May Contribute to Chronic Inflammation and Cancer-Related Processes
The most discussed disease-oriented model involves a two-part interaction. First, Neu5Gc from exogenous sources becomes incorporated into human glycoconjugates. Second, many humans carry anti-Neu5Gc antibodies of variable repertoire and intensity. When these antibodies encounter Neu5Gc-bearing tissues, the result may be a low-grade immune response rather than an efficient acute clearance event. This concept is commonly described as a chronic inflammation mechanism.
In this framework, Neu5Gc functions as a xeno-autoantigen: foreign in biosynthetic origin, yet displayed on human tissues. That combination makes it biologically unusual and particularly relevant to inflammation-associated disease models.
Why the Neu5Gc Cancer Discussion Persists
Cancer biology already involves altered glycosylation, immune remodeling, metabolic stress, and changes in the tumor microenvironment. Neu5Gc fits into this setting because tumors may show preferential accumulation of Neu5Gc-containing glycoconjugates compared with many normal tissues. Several mechanisms have been discussed in the literature:
- enhanced uptake of exogenous Neu5Gc by malignant cells
- hypoxia-associated increases in transport or incorporation pathways
- tumor-associated hypersialylation and altered ganglioside composition
- persistent interaction with circulating anti-Neu5Gc antibodies
- immune modulation through Neu5Gc-bearing gangliosides and shed glycoconjugates
What This Means for Diet-Associated Cancer Research
Any discussion of diet-associated cancer risk should remain multifactorial. Neu5Gc is better treated as a mechanistic factor under investigation than as a stand-alone explanation. Its value lies in linking dietary exposure, human-specific immune recognition, and inflammation-associated tumor biology within a testable experimental framework.
| Research Question | Why It Matters | Useful Experimental Tools |
|---|---|---|
| Is Neu5Gc present in a sample? | Confirms whether non-human sialic acid incorporation occurred | Specific antibodies, glycan analysis, orthogonal validation workflows |
| Is the signal truly Neu5Gc and not Neu5Ac? | Prevents misinterpretation caused by structurally similar epitopes | Selective antibody screening, competition assays, array-based profiling |
| Does Neu5Gc correlate with inflammation-associated phenotypes? | Supports mechanistic work on xeno-autoantigen biology | Cell assays, immune binding studies, tissue staining panels |
| Do tumors enrich Neu5Gc-bearing glycoconjugates? | Informs biomarker and target discovery research | IHC-ready antibodies, glycolipid and glycoprotein characterization |
Common Pitfalls in Neu5Gc vs Neu5Ac Studies
- using antibodies without robust cross-reactivity controls
- treating animal-derived culture components as biologically neutral
- assuming all sialic acid signals reflect human-endogenous Neu5Ac
- overstating causal claims where only association has been shown
- failing to separate glycoprotein-associated and glycolipid-associated Neu5Gc pools
Our Research Solutions for Neu5Gc-Focused Projects
Creative Biolabs offers research-use support for groups working on sialic acid evolution, xeno-autoantigen biology, glycan immunology, and tumor-associated Neu5Gc detection. We design research workflows for specificity, reproducibility, and downstream usability rather than one-size- fits-all reagent generation.
Anti-Neu5Gc Antibody Development
We develop custom antibodies against Neu5Gc-bearing targets with emphasis on discriminating Neu5Gc from Neu5Ac and related sialylated structures. This is valuable for tissue staining, glycan mapping, assay development, and mechanistic studies involving xeno-autoantigen recognition and tumor-associated sialylation.
Glycoarray Platforms
Our glycoarray workflows support fine specificity profiling against closely related glycan panels. This helps identify cross-reactivity early and improves confidence in neu5gc vs neu5ac discrimination.
Glycosylation Analysis
We support glycoconjugate characterization for proteins, lipids, and complex sample types, helping researchers define where Neu5Gc is present and how it is displayed.
Affinity and Kinetics Measurement by SPR
For projects that require quantitative binding evaluation, we provide affinity and kinetics assessment to support lead selection, assay transfer, and analytical confidence.
What We Can Help You Evaluate
- Neu5Gc-specific detection in tissue and cell samples
- discrimination between Neu5Gc and Neu5Ac-containing epitopes
- screening against broader sialylated glycan backgrounds
- reagent suitability for ELISA, flow cytometry, IF, IHC, and array workflows
- project designs relevant to chronic inflammation and tumor glycan studies
Request a Quote for Your Neu5Gc Project
If your team is studying the neu5gc vs neu5ac relationship in evolution, diet, inflammation, or cancer-associated glycans, send us your target format, preferred application, sample type, and validation priorities. We can help you define a practical reagent strategy for research use only, including specificity control design and downstream assay planning.
Discuss Your Neu5Gc Research Needs
Published Data
A recent open-access review summarized clinical and experimental evidence showing that Neu5Gc-containing glycoconjugates preferentially accumulate in a wide range of human tumors, whereas expression in normal adult tissues is limited and is mainly seen in cells with high turnover. The article explains that Neu5Gc differs from Neu5Ac by a single oxygen atom and that humans do not endogenously synthesize Neu5Gc because the CMAH pathway is inactive. It further discusses dietary uptake, hypoxia-enhanced incorporation, and tumor-associated metabolic changes as plausible contributors to Neu5Gc accumulation in malignant tissues. The review also describes a proposed model in which incorporated Neu5Gc may interact with circulating anti-Neu5Gc antibodies and participate in chronic inflammation and other tumor-associated processes, while noting that the precise biological implications remain incompletely understood.
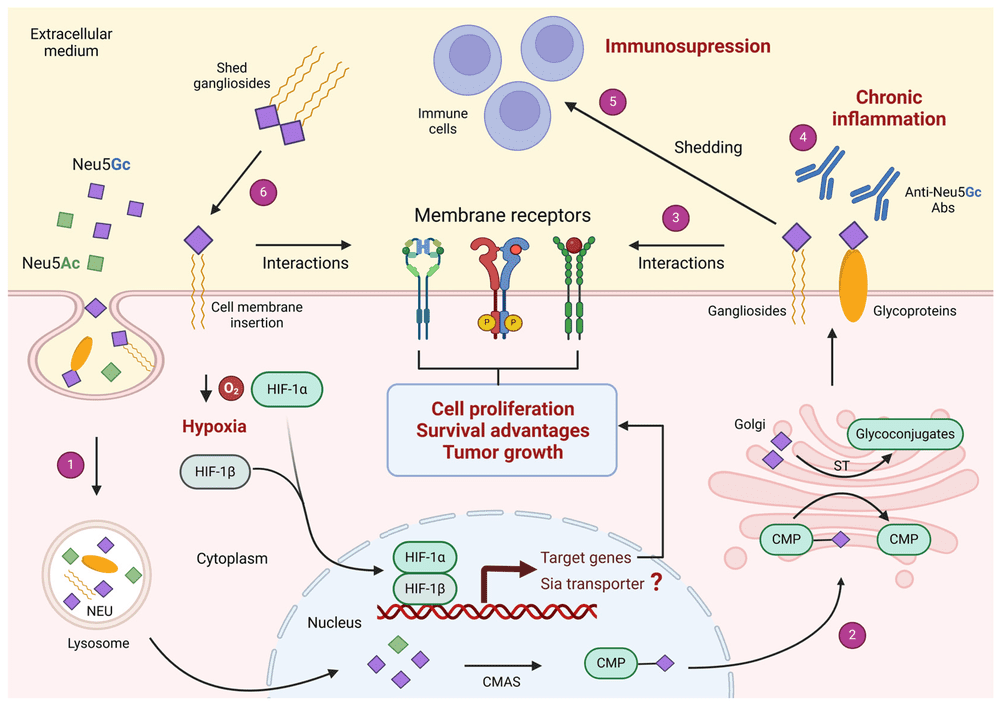 Fig. 1 Proposed role of Neu5Gc-containing glycoconjugates in cancer progression.1
Fig. 1 Proposed role of Neu5Gc-containing glycoconjugates in cancer progression.1
For researchers, this paper brings the main threads together without overstating them: human-specific loss of Neu5Gc synthesis, dietary incorporation, xeno- autoantigen recognition, chronic inflammation, and tumor enrichment. It also reinforces why high-specificity detection tools are necessary when studying Neu5Gc-bearing glycoconjugates in complex biological systems.
FAQs
What is the main difference between Neu5Gc and Neu5Ac?
Neu5Gc and Neu5Ac are closely related sialic acids. The structural difference is a single oxygen atom at the C-5 position. Even so, that small change can strongly affect immune recognition, glycan binding behavior, and experimental specificity.
Why do humans make Neu5Ac but not Neu5Gc?
Humans lost a functional CMAH enzyme during evolution. In most mammals, CMAH helps generate Neu5Gc from Neu5Ac. Because the human CMAH pathway is inactive, Neu5Ac is the dominant endogenous human sialic acid.
How can Neu5Gc still appear in human tissues if humans do not synthesize it?
Neu5Gc can be acquired from external sources and incorporated into human glycoconjugates through salvage and metabolic reuse pathways. Dietary exposure is the most widely discussed explanation.
Why is Neu5Gc described as a xeno-autoantigen?
It is foreign in biosynthetic origin because humans do not normally make it, yet it can become displayed on human tissues after incorporation. That unusual combination allows host antibodies to recognize a non-human glycan in a human tissue context.
How is Neu5Gc linked to chronic inflammation?
A proposed mechanism is that incorporated Neu5Gc interacts with circulating anti-Neu5Gc antibodies, producing a persistent low-grade immune response rather than rapid elimination. That ongoing immune engagement is why chronic inflammation is frequently discussed in this field.
Does Neu5Gc alone explain diet-associated cancer risk?
No. Diet-associated cancer risk is multifactorial. Neu5Gc is better viewed as one mechanistic factor under investigation because it links dietary exposure, non-human glycan incorporation, and inflammation-associated biology within a broader experimental framework.
Why are Neu5Gc-specific antibodies difficult to develop?
The challenge comes from the close structural similarity between Neu5Gc and Neu5Ac, the diversity of carrier glycans, and the risk of cross-reactivity with related sialylated epitopes. Rigorous screening against relevant controls is essential.
What should I provide when requesting a Neu5Gc-related service quote?
Please share your target type, sample source, intended application, preferred antibody format, and any required cross-reactivity exclusions. That information helps define a more accurate research-use workflow.
Reference:
- Blanco, Rancés, and Juan P. Muñoz. The role of non-human sialic acid Neu5Gc-containing glycoconjugates in human tumors: A review of clinical and experimental evidence. Biomolecules 15.2 (2025): 253. Distributed under Open Access license CC BY 4.0, without modification. https://doi.org/10.3390/biom15020253
