N-Acetyllactosamine (LacNAc) Repeats and Poly-LacNAc
Creative Biolabs supports glycan-focused research with anti-N-acetyllactosamine antibody development services for investigators studying linear and branched LacNAc motifs, poly-LacNAc extension, galectin binding, and carbohydrate antigens such as the i-antigen and I-antigen. N-acetyllactosamine repeats are not just structural segments on glycoconjugates. They contribute to lectin recognition, can influence cell-surface organization, and provide glycan frameworks that may be further remodeled into biologically relevant epitopes. For research teams working on glycan recognition, cell interaction, or antigen discovery, a clear view of poly-LacNAc biology is often useful before assay development starts.
Why LacNAc Repeats Matter in Glycan Biology
N-acetyllactosamine, often abbreviated as LacNAc, is the disaccharide Galβ1-4GlcNAc. Repetition of this unit generates extended chains known as poly-N-acetyllactosamine or poly-LacNAc. These chains are widely found on N-glycans, O-glycans, and glycolipids, where they act as elongation scaffolds rather than isolated terminal motifs. In practice, that means the same LacNAc building block can support several layers of glycan function:
- extension of complex glycans on membrane proteins and lipids
- presentation of internal LacNAc motifs to galectins
- generation of developmental and hematologic carbohydrate antigens
- substrate formation for further fucosylation, sialylation, or branching
- modulation of receptor organization at the cell surface
Because poly-LacNAc often appears on biologically active glycoproteins, its relevance extends far beyond structural glycobiology. It affects how cells interact with their environment, how glycan-binding proteins form lattices, and how antigenic signatures emerge during differentiation.
| Feature | Core Description | Research Relevance | Typical Context |
|---|---|---|---|
| LacNAc unit | Galβ1-4GlcNAc disaccharide | Basic recognition element for multiple lectins | Glycoproteins, glycolipids, complex glycans |
| Poly-LacNAc chain | Repeated LacNAc extension | Creates extended glycan scaffolds | Cell surface and secreted glycoconjugates |
| Linear i-antigen | Unbranched poly-LacNAc-rich sequence | Developmental and hematologic antigen biology | Fetal and immature erythroid systems |
| Branched I-antigen | Branched poly-LacNAc structure | Changes antigen display and recognition | Adult erythrocytes and differentiated cells |
| Galectin ligand platform | Internal LacNAc repeats on extended glycans | Can support lattice formation and receptor retention | Immune, epithelial, and tumor-associated cells |
N-Acetyllactosamine Structure and Chain Organization
The n-acetyllactosamine structure is simple at the disaccharide level, but its biological behavior depends heavily on context. A single LacNAc unit may sit near the nonreducing end of a glycan, while extended chains create internal recognition sites that are not equivalent to terminal epitopes. Poly-LacNAc can remain linear or become branched, and that architectural difference changes how proteins engage the chain.
Linear Extension
Alternating addition of galactose and N-acetylglucosamine builds repeated LacNAc units. Linear extension is especially relevant to the i-antigen concept because repeated unbranched sequences can dominate antigen presentation.
Branched Extension
Introduction of branch points into poly-LacNAc changes chain density, spatial display, and accessibility. This conversion underlies formation of the I-antigen and can alter lectin recognition patterns.
Internal Versus Terminal Motifs
Galectins often prefer internal LacNAc motifs rather than a simple terminal residue. For that reason, chain length and neighboring sugar composition can be as important as the presence of LacNAc itself.
Carrier Dependence
The same glycan chain can behave differently on glycoproteins and glycolipids. Surface density, molecular mobility, and steric exposure all influence downstream binding and assay performance.
From an analytical perspective, this is why researchers often need tools that distinguish short LacNAc motifs from extended poly-LacNAc and linear sequences from branched forms. Antibodies, lectins, and glycan arrays are each informative, but they answer different questions.
Poly-LacNAc Function in Cell Adhesion and Antigen Formation
Poly-LacNAc function is often best understood as a platform effect. Rather than acting only as an isolated antigen, the chain can influence how other molecules are organized at the cell surface. Several research themes are especially important.
Galectin Binding and Surface Organization
Galectins recognize β-galactoside-containing glycans, and poly-LacNAc is a well-studied scaffold because repeated LacNAc units can increase binding opportunities. This matters for receptor retention, clustering, and lattice formation at the plasma membrane. In experimental systems, longer or multivalent LacNAc-containing ligands often show stronger galectin engagement than simple disaccharides. That relationship is one reason poly-LacNAc is frequently discussed in connection with membrane organization and signaling-related studies.
Cell Adhesion Molecules and Cell Surface Retention
Many cell adhesion molecules and receptors carry complex glycans that can be extended with poly-LacNAc. When galectins bind these glycans, they may promote surface residency, stabilize receptor assemblies, and alter how cells interact with extracellular partners. This does not reduce adhesion biology to one mechanism, but it does explain why poly-LacNAc is regularly evaluated in studies of epithelial integrity, immune cell behavior, and tumor-associated glycan remodeling.
i-Antigen and I-Antigen Biology
The i-antigen is classically associated with linear poly-LacNAc-rich structures, whereas the I-antigen reflects branched forms generated during development and differentiation. This transition is particularly well known in erythroid biology. For researchers, the i-antigen/I-antigen system provides a useful model for understanding how a modest change in chain topology can reshape antigenicity without replacing the underlying LacNAc backbone. It also illustrates why structural definition matters when choosing probes for glycan detection.
Scaffold for Additional Epitopes
Poly-LacNAc is also a foundation for further modification. Fucosylation, sialylation, and branching can convert LacNAc-based chains into additional glycan determinants. In this sense, poly-LacNAc is not only a ligand platform but also an intermediate framework that supports downstream epitope diversification.
| Biological Theme | Role of Poly-LacNAc | What Researchers Often Measure | Useful Tools |
|---|---|---|---|
| Galectin binding | Presents repeated internal LacNAc motifs | Affinity, selectivity, multivalency effects | Glycan arrays, binding assays, SPR |
| Cell adhesion molecules | Supports glycan-dependent receptor organization | Surface retention, clustering, migration response | Flow cytometry, imaging, glycoprotein analysis |
| i-antigen formation | Reflects linear poly-LacNAc presentation | Developmental antigen expression | Specific antibodies, glycan profiling |
| I-antigen formation | Reflects branched chain topology | Branch-sensitive recognition | Defined glycans, targeted antibody screening |
| Epitope diversification | Acts as precursor scaffold | Downstream fucosylation or sialylation patterns | Mass spectrometry, glycoarrays |
Why This Topic Can Be Experimentally Difficult
- closely related LacNAc-containing glycans may cross-react with the same probe
- linear and branched forms are not interchangeable in binding studies
- short motifs do not always predict behavior of extended chains
- presentation on proteins and lipids can shift accessibility
- multivalency strongly affects apparent affinity
These variables are exactly why poly-LacNAc projects often require customized reagents rather than off-the-shelf detection tools.
Research Solutions for LacNAc and Poly-LacNAc Studies
Creative Biolabs provides research-use services for scientists who need selective tools for LacNAc-related glycans. Our workflow is designed for projects involving linear or branched poly-LacNAc, galectin-binding studies, i-antigen-focused antigen recognition, or glycan profiling across defined sample sets.
Anti-N-Acetyllactosamine Antibody Development
We develop custom antibodies against LacNAc-related motifs, including projects that require discrimination between short LacNAc epitopes and extended poly-LacNAc presentations. Screening strategies can be adapted for chain length, branching sensitivity, and intended assay format.
Glycoarray-Based Specificity Profiling
When the key question is recognition breadth rather than antibody generation alone, glycoarray analysis helps define whether a binder prefers linear LacNAc, extended poly-LacNAc, branched I-antigen-like motifs, or related glycans with terminal modifications.
Glycosylation Analysis
For projects centered on endogenous glycan remodeling, we support structural characterization of glycoproteins and other glycoconjugates. This is useful when poly-LacNAc is suspected but not yet confirmed in the target system.
Binding and Kinetics Evaluation
For galectin binding or glycan-protein interaction studies, kinetic analysis can help separate weak motif recognition from strong multivalent engagement. This is particularly useful when comparing different LacNAc chain presentations.
What We Can Help You Define
- whether your target contains LacNAc or extended poly-LacNAc
- whether a binder is selective for linear or branched motifs
- whether chain extension changes galectin recognition
- whether a probe performs in ELISA, flow cytometry, staining, or array-based assays
- whether cross-reactivity with related glycans is acceptable for your study goal
All services are provided for research use only and are not intended for clinical diagnosis or treatment.
Start a LacNAc-Focused Project
If your team is evaluating n-acetyllactosamine structure, poly-LacNAc function, galectin binding, or i-antigen-related glycan display, a well-defined reagent strategy can reduce ambiguity early in the project. We can work from a target glycan, a known binding protein, a cell model, or an application requirement. Typical starting inputs include the glycan class of interest, preferred assay format, chain topology concerns, and any related glycans that should be excluded during screening.
Request a Custom LacNAc Research Solution
Published Data
A useful example of how poly-LacNAc architecture can affect glycan recognition comes from an open-access study that analyzed human galectin-3 binding to multivalent poly-LacNAc neo-glycoproteins. The investigators compared a panel of BSA-displayed glycans and measured their interactions with galectin-3 by ELISA and surface plasmon resonance. In this model system, the tetra- to heptasaccharide neo-glycoproteins bound much more strongly than the disaccharide conjugates. In the SPR experiments, the strongest binders were the LacdiNAc-capped tetrasaccharide and hexasaccharide neo-glycoproteins, with reported dissociation constants of 14 pM and 26 pM, respectively, whereas the asialofetuin control showed weaker binding in the nanomolar range. Within this Gal-3 binding system, the data support the conclusion that glycan extension and presentation strongly influence binding strength.
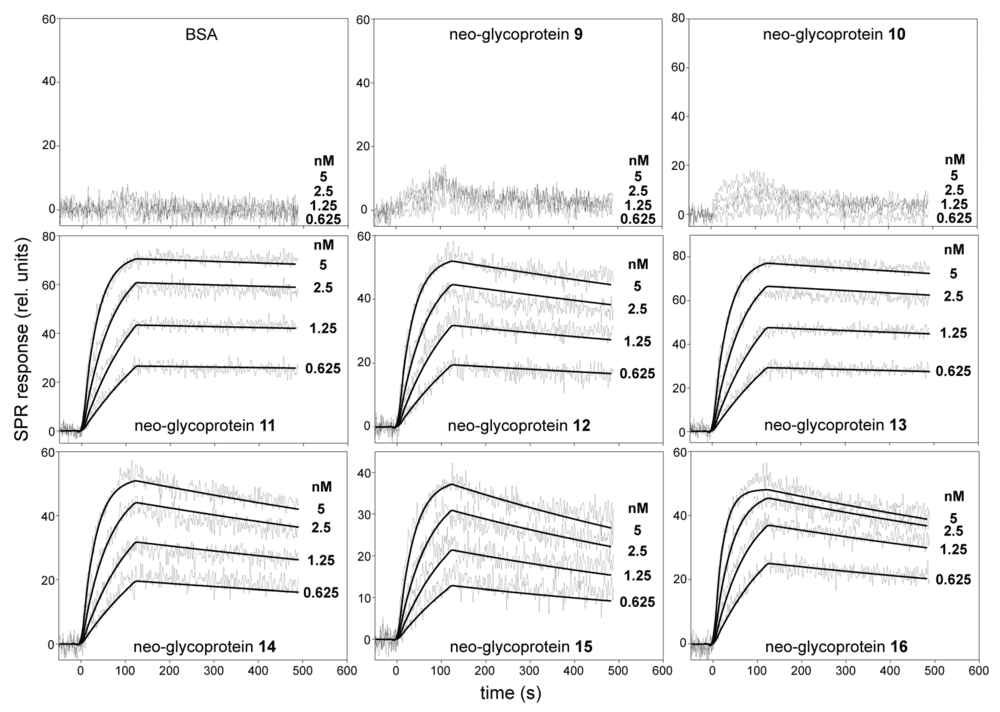 Fig. 1 SPR Kinetic Binding Analysis of Galectin-3 Interactions with Poly-LacNAc Neo-Glycoproteins.1
Fig. 1 SPR Kinetic Binding Analysis of Galectin-3 Interactions with Poly-LacNAc Neo-Glycoproteins.1
This figure is particularly relevant because it visualizes the measurable difference between short and extended glycan ligands in a defined Gal-3 binding system. For investigators studying galectin binding, receptor organization, or glycan-dependent cell interactions, it underscores the value of testing extended structures directly rather than inferring behavior from minimal motifs alone.
FAQs
What is the difference between LacNAc and poly-LacNAc?
LacNAc is a single Galβ1-4GlcNAc disaccharide. Poly-LacNAc is an extended chain built from repeated LacNAc units. The repeated form creates extended internal motifs and different recognition behavior.
Why is poly-LacNAc important for galectin binding?
Many galectins, especially galectin-3, bind more effectively to extended LacNAc-containing glycans than to a single disaccharide. Chain length, internal motif exposure, and multivalent presentation can all strengthen binding.
How is the i-antigen related to poly-LacNAc?
The i-antigen is associated with linear poly-LacNAc-rich structures. It is part of a classic developmental antigen system used to study how glycan chain architecture changes during differentiation.
What distinguishes the I-antigen from the i-antigen?
The key difference is topology. The i-antigen reflects more linear poly-LacNAc presentation, while the I-antigen reflects branched structures. That branching change can alter antigen recognition even when the LacNAc backbone remains central.
Can antibodies distinguish short LacNAc motifs from extended poly-LacNAc chains?
They can, but only when screening is carefully designed. Chain length, branching, neighboring sugars, and presentation format all affect selectivity, so validation against defined glycan panels is important.
Which assays are useful for LacNAc repeat research?
Common options include glycan arrays, ELISA, flow cytometry, histological staining, glycoprotein analysis, and kinetic binding studies such as SPR. The most informative assay depends on whether the question is structural, functional, or application-driven.
Reference:
- Bumba, Lubor, et al. Poly-N-Acetyllactosamine Neo-Glycoproteins as Nanomolar Ligands of Human Galectin-3: Binding Kinetics and Modeling. International Journal of Molecular Sciences 19.2 (2018): 372. Distributed under Open Access license CC BY 4.0, without modification. https://doi.org/10.3390/ijms19020372
