Beta-Galactoside Binding Proteins and Galectins
Creative Biolabs supports glycan-focused research on beta-galactoside-binding proteins and galectins with tailored reagent development and analytical services. For projects involving beta-gal-containing glycans, galectin ligands, or related interaction networks, our Anti-Beta-Gal Antibody Development service can support the generation of research antibodies for assay development, target validation, and mechanistic studies. Because galectin biology is strongly shaped by glycan presentation on extracellular matrix components, immune cells, and tumor cells, well-defined reagents and orthogonal analytical strategies are often important for study design.
Why Beta-Galactoside Binding Proteins Matter
Beta-galactoside-binding proteins are an important group of lectins that recognize glycans containing terminal or subterminal beta-galactoside motifs. Galectins are the best-characterized family within this area, although individual members can differ in fine specificity. Their biology is shaped by glycan presentation rather than peptide sequence alone, which makes them highly relevant in glycobiology, tumor microenvironment research, inflammatory signaling, cell adhesion, immune regulation, and extracellular matrix remodeling.
In practical terms, galectin biology links glycan display to cell behavior. A cell may express the same protein backbone under different biological states, yet altered glycosylation can change galectin binding, receptor clustering, and downstream signaling. This is why beta-gal structure and lectin-glycan interaction are widely studied in cancer progression and chronic inflammation.
Glycan Recognition
Most galectins recognize beta-galactoside-containing glycoconjugates through a conserved carbohydrate recognition domain.
Receptor Organization
Multivalent binding can cluster glycoproteins at the cell surface and reshape signaling output.
Immune Regulation
Galectins influence T cells, myeloid cells, cytokine balance, and inflammatory cell recruitment.
Tumor Biology
Dysregulated galectin activity is associated with immune escape, invasion, angiogenesis, and metastatic spread in many cancer settings.
Beta-Gal Structure, Galectin Classes, and Binding Logic
In this context, beta-gal structure usually refers to glycan motifs that expose galactose in a beta linkage, often within LacNAc-related or more complex branched glycans. Recognition is not determined by galactose alone. Binding can be influenced by neighboring sugars, branching, sialylation, fucosylation, linker context, glycan density, and whether ligands are displayed on glycoproteins, glycolipids, or extracellular matrix components.
Galectins share a conserved carbohydrate recognition domain, but their architecture differs. This structural diversity determines valency, oligomerization behavior, and receptor crosslinking capacity.
| Class | Representative Members | Structural Feature | Functional Implication |
|---|---|---|---|
| Prototype | Galectin-1, -2, -7, -13, -14 | One carbohydrate recognition domain per subunit, often forming homodimers | Efficient ligand crosslinking at the cell surface |
| Chimera | Galectin-3 | One carbohydrate recognition domain plus a non-lectin N-terminal domain | Oligomerization and lattice formation with broad signaling effects |
| Tandem-repeat | Galectin-4, -8, -9, -12 | Two distinct carbohydrate recognition domains connected by a linker peptide | Simultaneous engagement of different glycan ligands or receptors |
Key determinants of lectin-glycan interaction
- Accessibility of beta-galactoside motifs on the cell surface
- Extension into poly-N-acetyllactosamine sequences
- Terminal modification by sialic acid or fucose
- Local ligand density and multivalent display
- Galectin dimerization or higher-order assembly
- Cell type-specific glycosylation state
Why the same galectin can behave differently across models
Galectin function is context-dependent. The same galectin may promote cell death in one immune subset but support survival, motility, or receptor retention in another setting. This variability often reflects changes in glycan presentation, ligand accessibility, and cell state rather than galectin abundance alone. For experimental design, that usually means combining a defined glycan reagent strategy with orthogonal validation methods.
Major Galectins Studied in Cancer and Inflammation
Galectin-1
Frequently studied as an immunoregulatory lectin. It can shape T-cell fate, support immune tolerance in the tumor microenvironment, and influence stromal interactions.
Galectin-3
The only chimera-type galectin in mammals. It is widely linked to receptor clustering, inflammatory signaling, fibrosis-related pathways, and multiple aspects of cancer progression.
Galectin-8
A tandem-repeat galectin with roles in adhesion signaling, stress responses, and selective receptor interactions that depend on glycan context.
Galectin-9
Often evaluated in immune regulation and tumor-associated immune suppression, especially in relation to T-cell function and checkpoint-related biology.
How Galectins Participate in Cancer Progression
Cancer-associated glycosylation remodeling changes the availability of galectin ligands on tumor cells, endothelial cells, platelets, fibroblasts, and infiltrating immune cells. This creates a dense communication network in which galectins can alter receptor residence time, cluster glycoconjugates, and influence signaling programs linked to tumor progression.
Immune Escape
Several galectins suppress effective anti-tumor immunity by altering T-cell activation, survival, trafficking, and cytokine balance. This shifts the microenvironment toward tolerance rather than productive immune clearance.
Adhesion, Migration, and Metastatic Fitness
Galectins can regulate cell-cell and cell-matrix interactions, helping tumor cells adapt to invasion, circulation, and distant-site colonization. Their effect often reflects changes in glycan architecture rather than protein abundance alone.
Inflammation and Thromboinflammation
Galectin function also extends to inflammatory recruitment and platelet biology. In cancer-associated settings, this can help sustain a pro-inflammatory and pro-thrombotic environment associated with tumor persistence and dissemination.
Research questions commonly addressed on this topic
- Which glycan motifs drive galectin binding in a specific tumor model
- Whether galectin-1 and galectin-3 contribute differently to immune regulation
- How altered glycosylation changes receptor clustering and signaling kinetics
- Whether a candidate reagent can distinguish closely related beta-gal-containing structures
- How galectin-associated inflammation correlates with cancer progression markers
| Biological Context | Representative Galectin Function | Common Readout | Useful Research Tool |
|---|---|---|---|
| Tumor microenvironment | Immune modulation and receptor lattice formation | Flow cytometry, cytokine profiling, tissue staining | Selective anti-glycan or anti-galectin antibodies |
| Cell adhesion studies | Crosslinking of surface glycoconjugates | Adhesion assay, migration assay, live-cell imaging | Defined glycan probes and binding assays |
| Inflammatory signaling | Modulation of leukocyte activation and cytokine response | Immune cell phenotyping, pathway analysis | Validated antibodies and glycoanalytical support |
Research Solutions and Service Details
Creative Biolabs provides research-use support for teams studying beta-galactoside binding, galectin function, and glycan-dependent target biology. We focus on reagent quality, specificity control, and assay compatibility rather than one-size-fits-all workflows. The services below are positioned for research applications and should be selected according to the target class, glycan context, and assay objective.
Anti-Beta-Gal Antibody Development
We develop customized antibodies against beta-gal-associated glycan targets for researchers who need selective recognition, low background, and reproducible performance across assay formats. This option is most relevant when the study requires direct detection of beta-gal-containing glycans or closely related glycan epitopes in ELISA, flow cytometry, immunostaining, glycan profiling, or related research assays.
Glycoarray and Binding Analysis Support
We help characterize lectin-glycan interaction patterns with glycoarray-based profiling and complementary analytical strategies. This is especially useful when closely related glycans must be distinguished experimentally or when ligand preference needs to be profiled at the panel level.
Glycosylation Analysis
For studies linking galectin biology to glycan remodeling, our glycosylation analysis services can support ligand characterization, comparative profiling, and structure-function interpretation across different biological states or sample types.
Affinity and Kinetics Evaluation
When a project requires quantitative comparison of interaction strength or reagent ranking, binding characterization can be incorporated through suitable affinity and kinetics evaluation workflows.
What we can align with your project
- Target definition around beta-gal-containing glycans or galectin-relevant ligands
- Specificity strategies for structurally similar glycans
- Antibody format selection for different research assays
- Analytical design for ligand profiling and validation
- Documentation suitable for internal research decision-making
Request a Quote for Your Study
If your team is studying galectin-mediated signaling, beta-gal structure, tumor-associated glycans, or inflammatory glycan biology, share your target, sample type, preferred application, and validation goals with Creative Biolabs. We can help translate a broad glycobiology question into a practical reagent and assay plan for research use.
Discuss Your Beta-Gal and Galectin Project
Published Data
A 2023 open-access review summarized how galectins function in cancer-associated inflammation and thrombosis. The authors described galectins as carbohydrate-binding proteins that regulate proliferation, adhesion, migration, and phagocytosis, and noted that galectin dysregulation is linked to tumor development, immune cell recruitment, modulation of neutrophils, monocytes, and lymphocytes, as well as platelet-related pro-thrombotic events. The review also outlined the structural classification of mammalian galectins into prototype, chimera, and tandem-repeat forms, which helps frame how galectin architecture contributes to ligand crosslinking and cell-surface organization.
For researchers studying cancer, inflammation, or thrombosis, this paper links galectin architecture with biological behavior. Structure is not an isolated biochemical detail here. The number and arrangement of carbohydrate recognition domains influence how galectins oligomerize, crosslink ligands, and reshape signaling in complex tissue environments.
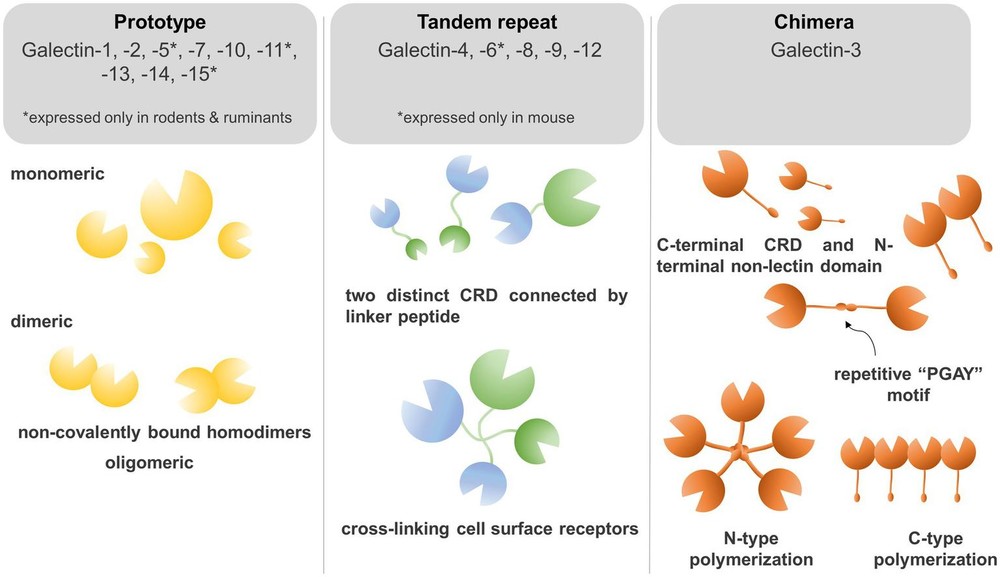 Fig.1 Galectin classification in beta-galactoside binding proteins.1
Fig.1 Galectin classification in beta-galactoside binding proteins.1
The figure highlights three major galectin classes. Prototype galectins contain one carbohydrate recognition domain and often function as homodimers. Galectin-3 is the only chimera-type member and carries a non-lectin N-terminal region that supports oligomerization. Tandem-repeat galectins contain two distinct carbohydrate recognition domains connected by a linker peptide. The review also notes that not every galectin shows identical glycan preference, so structural class should not be treated as a complete predictor of ligand specificity. This framework nonetheless helps explain why different galectins can display different binding geometries and different effects on receptor organization.
FAQs
What are beta-galactoside binding proteins?
They are proteins that recognize glycans containing beta-galactoside motifs. Galectins are the best-known family in this area, and most members engage glycans through a conserved carbohydrate recognition domain.
Why are galectins important in cancer research?
Galectins can affect immune regulation, adhesion, migration, stromal communication, and other processes that shape cancer progression. Their activity often reflects both galectin expression and tumor-associated glycosylation changes.
Is beta-gal structure alone enough to predict galectin binding?
Not usually. Binding also depends on glycan extension, branching, terminal modification, ligand density, and molecular presentation on proteins, lipids, or matrix components.
How do galectin-1 and galectin-3 differ experimentally?
Galectin-1 is a prototype galectin that commonly functions as a homodimer, whereas galectin-3 is the only chimera-type galectin and can oligomerize through its N-terminal domain. That difference can lead to different receptor clustering behavior and biological outcomes.
Can Creative Biolabs support studies on lectin-glycan interaction rather than only antibody generation?
Yes. In addition to anti-beta-gal antibody development, we can support glycoarray-based profiling, glycosylation analysis, and related binding characterization for research projects focused on glycan recognition.
Are these services intended for clinical diagnosis or treatment?
No. The services and discussion on this page are for scientific research use only and are not intended for clinical diagnosis or therapeutic use.
What information should I provide when requesting a quote?
The most helpful starting points are your target or glycan motif of interest, sample type, preferred assay, required specificity profile, and any timeline or validation expectations for the research study.
Reference:
- Kruk, Linus, Attila Braun, Erika Cosset, Thomas Gudermann, and Elmina Mammadova-Bach. Galectin functions in cancer-associated inflammation and thrombosis. Frontiers in Cardiovascular Medicine 10 (2023): 1052959. Distributed under Open Access license CC BY 4.0, without modification. https://doi.org/10.3389/fcvm.2023.1052959
- Return to Glycan Science
- Anti-Beta-Gal Antibody Development
- Glycoarray Platforms
- Glycosylation Analysis
