Anti-Galactosyltransferase (GalT) Antibody Development Service
Galactosyltransferases (GalTs) encompass a large and diverse family of enzymes responsible for transferring galactose from an activated donor (typically UDP-galactose) to various acceptor substrates, including glycoproteins, glycolipids, and proteoglycans. These enzymes, particularly the beta-1,4-galactosyltransferase (B4GALT) and core 1 synthase, glycoprotein-N-acetylgalactosamine 3-beta-galactosyltransferase (C1GALT1) families, are pivotal in defining the glycan structures that mediate cell-cell interactions, immune responses, and signal transduction. As a global leader in antibody engineering, Creative Biolabs leverages its proprietary platform to offer comprehensive Anti-Glycan Related Enzyme Antibody Development Services. We specialize in overcoming the immunogenic challenges of these highly conserved enzymes to deliver high-affinity anti-GalT antibodies for advanced structural and functional analysis.
Background of Galactosyltransferases
Galactosyltransferases are categorized into several subfamilies based on the linkage they catalyze (e.g., beta-1,3, beta-1,4, or alpha-1,3). The B4GALT family, comprising seven members (B4GALT1–7), is perhaps the most extensively studied, playing essential roles in N-linked glycosylation and the synthesis of lactose and lactosamine disaccharides. Specifically, B4GALT1 is a key component of the lactose synthase complex. Other significant members include the C1GALT1, which initiates core 1 O-glycan synthesis (T-antigen formation), and various alpha-galactosyltransferases responsible for synthesizing the Gal-alpha-1,3-Gal epitope.
Dysregulation of these enzymes is frequently observed in pathological conditions. For instance, altered expression of B4GALT isoforms has been linked to the metastatic potential of cancer cells and the modulation of immune receptor signaling. However, distinguishing between these closely related isoforms using traditional reagents remains a significant hurdle due to their high sequence homology in catalytic domains. The development of specific anti-glycosyltransferase antibodies is therefore critical for dissecting their individual contributions to disease mechanisms.
Challenges in Developing Antibodies Against GalT
Generating reliable antibodies against galactosyltransferases presents unique difficulties compared to standard protein targets. These enzymes are often residents of the Golgi apparatus, meaning they are membrane-bound and may require specific detergents to maintain native conformation during immunization or screening. Furthermore, as Carbohydrate-Active Enzymes (CAZymes), many GalTs share conserved catalytic domains, leading to high cross-reactivity if the immunogen is not carefully designed. The distinction between a beta-galactosidase antibody target and a galactosyltransferase target also requires precise epitope mapping to avoid confusion in downstream assays.
Our Anti-GalT Antibody Development Services
Creative Biolabs provides a modular service package tailored to the specific needs of glyco-biology researchers. Whether you require a broad-spectrum CAZyme antibody or an isoform-specific reagent, our platform ensures optimal results.
Custom Polyclonal & Monoclonal Antibody Production
We offer end-to-end production of both polyclonal and monoclonal antibodies targeting specific GalT family members. Our team utilizes advanced antigen design strategies, such as peptide synthesis derived from unique variable regions or recombinant protein expression, to minimize cross-reactivity with other glycosyltransferase antibodies.
Hybridoma and Phage Display Technologies
To achieve high affinity and specificity, we employ robust hybridoma development and phage display library screening. This is particularly effective for generating recombinant antibodies against difficult targets like membrane-bound B4GALT or C1GALT1, ensuring the resulting anti-glycan enzyme antibody functions effectively in diverse applications.
Target-Specific Development
Our expertise covers the full spectrum of galactosyltransferases. We can develop antibodies specific to B4GALT1 through B4GALT7, C1GALT1, and alpha-1,3-galactosyltransferases. We also provide comparative development services for related enzymes, such as anti-sialyltransferase antibodies or fucosyltransferase antibodies, to support comprehensive pathway analysis.
Application-Oriented Validation
We validate all generated antibodies for their intended use. Whether you need a glycosyltransferase expression antibody for Western Blotting, Immunohistochemistry (IHC), or Immunofluorescence (IF) to localize enzymes within the Golgi, we tailor our screening process to match your experimental conditions.
Development Workflow
Inquire about GalT Antibodies
Service Highlights
High Specificity
Distinguish between highly homologous isoforms like B4GALT1 and B4GALT2 with precision.
Comprehensive Validation
Validated for WB, IHC, and IF to ensure functionality in native and denatured states.
Diverse Species
Antibodies available from mouse, rabbit, rat, and alternative hosts like llama or chicken.
Broad CAZyme Coverage
Capabilities extend to other enzyme families including anti-sialyltransferase and anti-fucosyltransferase.
Published Data
Recent investigations into the progression of early-stage lung adenocarcinoma (LUAD) have illuminated the critical role of glycosyltransferases in tumor progression and immune evasion. A pivotal study identified Beta-1,4-galactosyltransferase 1 (B4GALT1) as a significant driver of the transition from minimally invasive to invasive phenotypes. Comprehensive immune profiling and immunohistochemical analyses revealed a stark inverse correlation between B4GALT1 expression and the infiltration of functional, Granzyme B-positive CD8+ T cells within the tumor microenvironment. Mechanistically, B4GALT1 was found to robustly upregulate Programmed Death-Ligand 1 (PD-L1) levels via two distinct pathways: post-transcriptionally, by directly mediating its N-linked glycosylation to enhance protein stability and prevent ubiquitin-proteasomal degradation, and transcriptionally, by stabilizing the upstream regulator TAZ. This enzymatic activity effectively creates an immunosuppressive barrier that facilitates tumor invasion. Crucially, in vivo xenograft experiments demonstrated that targeting B4GALT1 not only suppressed tumor growth but also significantly potentiated the therapeutic efficacy of anti-PD-1 checkpoint blockade, successfully restoring T-cell cytotoxicity against cancer cells. These findings underscore the potential of B4GALT1 as a novel therapeutic target and highlight the essential value of high-specificity antibodies for characterizing glycosylation-dependent immune checkpoints and dissecting complex tumor-immune interactions.
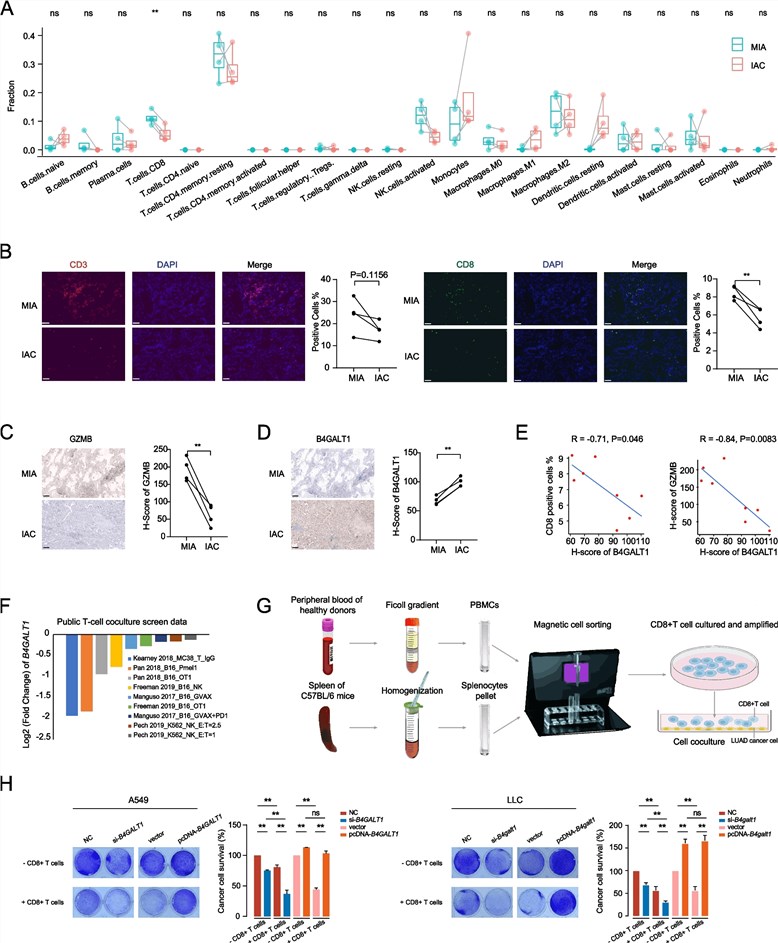 Fig.1
B4GALT1 expression negatively regulates CD8+ T-cell infiltration and cytotoxic activity in lung adenocarcinoma.1
Fig.1
B4GALT1 expression negatively regulates CD8+ T-cell infiltration and cytotoxic activity in lung adenocarcinoma.1
FAQs
How do you ensure your Anti-GalT antibodies do not cross-react with other glycosyltransferases?
We utilize rigorous antigen design, selecting peptide sequences that are unique to the specific GalT isoform (e.g., B4GALT1 vs. B4GALT2) and located away from the highly conserved catalytic domain found in many carbohydrate active enzymes. We also employ negative screening against related proteins during the clone selection process.
Can you develop antibodies against other glycan-related enzymes besides GalT?
Yes, our platform covers a wide range of enzymes. We offer development services for anti-sialyltransferase antibody, fucosyltransferase antibody, anti-neuraminidase antibody, mannosidase antibody, and more.
Are your antibodies suitable for detecting native enzymes in flow cytometry?
Many of our antibodies are suitable for flow cytometry, but this depends on the antigen design. For detection of native conformation, we recommend our cell-based immunization or whole-protein immunization strategies. Please specify your application needs when inquiring.
What is the difference between an anti-glycosidase antibody and an anti-glycosyltransferase antibody?
Glycosyltransferases (like GalT) synthesize glycans by transferring sugar residues, whereas glycosidases (like beta-galactosidase or sialidase) break them down. Antibodies against these two classes target completely different enzymes with distinct structures and functions.
Do you provide services for bacterial or viral glycan enzymes?
Yes, we can develop antibodies against non-mammalian targets, such as bacterial sialidase antibody, viral neuraminidase antibody (including influenza neuraminidase antibody), and other pathogen-derived enzymes.
What Our Customers Say
"We needed a specific antibody for B4GALT1 that wouldn't cross-react with other family members for our IHC studies. Creative Biolabs delivered a highly specific clone that worked perfectly on our tissue arrays."
"The team was very knowledgeable about CAZymes. Their customized antigen design strategy for our rare glycosyltransferase target was impressive, and the resulting antibodies had excellent affinity."
Reference:
- Cui, Y., Li, J., Zhang, P. et al. "B4GALT1 promotes immune escape by regulating the expression of PD-L1 at multiple levels in lung adenocarcinoma." Journal of Experimental & Clinical Cancer Research 42 (2023): 146. Distributed under Open Access license CC BY 4.0. https://doi.org/10.1186/s13046-023-02711-3
Supports
- Glycosyltransferase & Glycosidase Substrate Microarray
- Glycosylation Analysis
- Custom Glycosylation of Biomolecules
