Anti-Hexosaminidase Antibody Development Service
Effective monitoring of host cell proteins (HCPs) is pivotal for ensuring the safety and stability of therapeutic biologics. At Creative Biolabs, we leverage our extensive expertise in Anti-Glycan Related Enzyme Antibody Development Service to provide specialized solutions for the detection of lysosomal enzymes. Our custom anti-hexosaminidase antibody development service is designed to support researchers in identifying and characterizing residual hexosaminidase B (HexB) and related isoforms in bioprocessing workflows, helping to assess their potential impact on product N-glycan integrity.
Understanding Hexosaminidases in Bioprocessing
Hexosaminidases are lysosomal glycosidase enzymes responsible for the hydrolysis of terminal N-acetyl-D-hexosamine residues from N-acetyl-β-D-hexosaminides. In humans and other mammals, the system is primarily composed of two major isoenzymes: Hexosaminidase A (Hex A), a heterodimer of α and β subunits, and Hexosaminidase B (Hex B), a homodimer of β subunits. These enzymes play a critical biological role in the degradation of gangliosides and other glycoconjugates within the lysosome.
In the context of biopharmaceutical manufacturing, particularly when using Chinese Hamster Ovary (CHO) cells or other mammalian expression systems, these intracellular enzymes can be released into the culture supernatant upon cell lysis. Even trace amounts of residual glycosidases can pose a significant risk to the quality of therapeutic monoclonal antibodies (mAbs). Specifically, residual HexB activity can cleave terminal N-acetylglucosamine (GlcNAc) residues from the N-glycans of the antibody product during storage. This degradation can alter the drug's pharmacokinetic profile, reduce efficacy, or increase immunogenicity. Therefore, the development of high-affinity anti-glycan enzyme antibodies capable of detecting these specific HCPs is essential for process validation and stability studies.
Custom Anti-Hexosaminidase Antibody Development Workflow
Start Your Project
Our Custom Antibody Development Services
We offer a comprehensive suite of services to generate antibodies targeting specific subunits of the hexosaminidase system. Whether you require a polyclonal antibody for broad detection or a monoclonal glycosidase antibody for specific isoform quantification, our platform is adaptable to your research needs.
Anti-Hexosaminidase B (HexB) Antibody Development
Hexosaminidase B is often the primary concern in mAb stability studies due to its stability and activity at neutral pH. We develop antibodies specifically targeting the β-subunit homodimer. These reagents are critical for establishing sensitive ELISAs to monitor HexB clearance during downstream processing (DSP) and to verify the purity of the final drug substance.
Anti-Hexosaminidase A (HexA) Antibody Development
For researchers investigating lysosomal storage disorders or specific cellular pathways, distinguishing Hex A from Hex B is vital. We utilize subtractive screening strategies to isolate clones that recognize the α-subunit unique to Hex A, minimizing cross-reactivity with the β-subunit. This allows for precise immunodetection in complex biological samples.
Anti-Hexosaminidase S (HexS) Antibody Development
Hexosaminidase S, a homodimer of α-subunits, is typically found in pathological conditions or specific developmental stages. We provide custom antibody generation for this rare isoform, facilitating advanced research into hexosaminidase biology and subunit assembly mechanics.
Assay Development Support
Beyond antibody generation, we support the integration of these reagents into functional assays. We can assist in pairing antibodies for sandwich ELISA development, optimizing conditions for Western Blot detection of trace enzymes, or validating reagents for immunoprecipitation to isolate active enzymes from supernatants.
Why Choose Our Anti-Hexosaminidase Antibodies?
High Sensitivity
Engineered to detect low-abundance lysosomal glycosidase antibodies in complex HCP mixtures.
Subunit Specificity
Precise targeting of α or β subunits to distinguish between Hex A, Hex B, and Hex S isoforms.
Validated Performance
Verified performance in relevant assays like Western Blot and ELISA for consistent results.
Rapid Turnaround
Optimized workflows to deliver your custom reagents within competitive timelines.
How We Work with Your Team
Our collaborative process begins with a detailed consultation to understand the scope of your HCP monitoring or enzymatic research. We guide you through antigen selection—whether to use a specific peptide sequence to avoid homology with the therapeutic protein or a full-length enzyme for maximum immunogenicity. Throughout the project, we provide regular updates and data reports, ensuring the final anti-glycosidase antibody meets your specifications for specificity and affinity.
Inquire Now
Published Data
Reliable immunodetection is pivotal for evaluating the pharmacokinetics and structural stability of therapeutic enzymes in preclinical models. In a study addressing GM2 gangliosidosis, researchers engineered a protease-resistant modified human β-hexosaminidase B to overcome the instability of conventional enzyme replacement therapies. Utilizing specific anti-hexosaminidase antibodies, the investigation combined immunohistochemical staining with immunoprecipitation analysis to comprehensively track the enzyme following intracerebroventricular administration. The antibodies successfully visualized the uptake of the therapeutic protein into key brain regions, such as the hippocampal formation, confirming its penetration into neural parenchyma. Furthermore, immunoblotting analysis enabled the precise differentiation between the mature enzyme and its proteolytic fragments, providing conclusive evidence that the optimized variant maintained its structural integrity significantly better than previous iterations across the forebrain, midbrain, and hindbrain. This robust antibody-based validation was essential to correlate the enzyme’s enhanced biostability with the observed reduction in accumulated glycolipids and subsequent motor function improvement. These findings highlight the critical value of high-specificity antibodies in monitoring the delivery, stability, and therapeutic efficacy of biologic drug candidates within complex biological matrices.
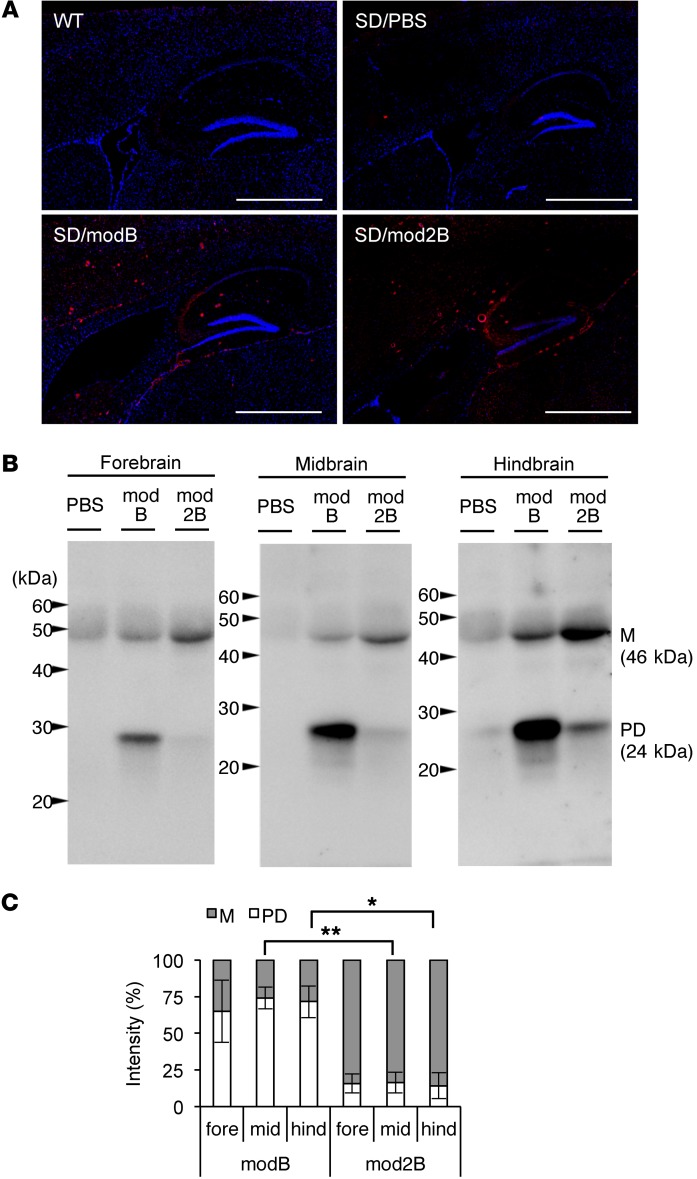 Fig.1
Analysis of biodistribution and protease resistance of modified HexB in brain tissue.1
Fig.1
Analysis of biodistribution and protease resistance of modified HexB in brain tissue.1
FAQs
Why is monitoring Hexosaminidase B important in monoclonal antibody production?
Hexosaminidase B is a robust lysosomal enzyme that can be released from CHO cells during culture. It has the ability to cleave terminal GlcNAc residues from the N-glycans of the secreted antibody. This enzymatic degradation can compromise the consistency of the drug product and potentially affect its safety and efficacy, making detection critical.
Can you generate antibodies that distinguish between Hex A and Hex B?
Yes. Hex A is a heterodimer (αβ) and Hex B is a homodimer (ββ). We can develop antibodies targeting the α-subunit to specifically detect Hex A, or target unique conformational epitopes on the ββ dimer to preferentially recognize Hex B, depending on the screening strategy.
What applications are these antibodies suitable for?
Our custom antibodies are typically validated for use in ELISA (for HCP quantification), Western Blot (for qualitative confirmation), and immunoprecipitation. We can also tailor the validation process for flow cytometry or immunohistochemistry if required for basic research.
Do you offer recombinant antigen production services for immunization?
Yes, we can produce high-quality recombinant human or CHO-derived hexosaminidase antigens. This ensures that the antibodies generated are raised against the correctly folded protein, increasing the likelihood of recognizing the native enzyme in your samples.
What is the typical timeline for a custom antibody project?
A standard monoclonal antibody development project using hybridoma technology typically takes 4-6 months, including antigen preparation, immunization, fusion, screening, and sub-cloning. Polyclonal projects are generally faster, taking approximately 3-4 months.
What Our Customers Say
"Detecting trace HexB in our CHO supernatant was a challenge until we used the custom antibodies from Creative Biolabs. The specificity for the beta-subunit was excellent, allowing us to validate our clearance steps with confidence."
"We needed antibodies to distinguish Hex A from Hex B for a lysosomal storage disease model. Their team designed a screening strategy that successfully isolated alpha-subunit specific clones. Highly professional service."
"The data package provided with the purified antibody was comprehensive. It included Western blot validation against both recombinant protein and cell lysates, which saved us a lot of time on internal QC."
"Our request was urgent due to a regulatory filing deadline. Creative Biolabs accelerated the polyclonal production timeline and delivered high-titer antibodies that worked on the first try in our ELISA setup."
Reference:
- Kitakaze, K., et al. "Protease-resistant modified human β-hexosaminidase B ameliorates symptoms in GM2 gangliosidosis model." The Journal of Clinical Investigation 126.5 (2016): 1691-1703. Distributed under Open Access license CC BY 4.0, without modification. https://doi.org/10.1172/JCI85300
Supports
- Custom Glycosylation of Biomolecules
- Glycosylation Analysis
- Glycosyltransferase & Glycosidase Substrate Microarray
