Non-Human Glycans and Xenotransplantation Challenges
Creative Biolabs offers Non-Human Glycan Antibody Development for research teams studying glycan-driven immune recognition in xenotransplantation. When donor tissues carry glycans that are absent or immunologically foreign in humans, these structures can function as xenoantigens and trigger antibody binding. Reliable glycan-specific reagents are therefore useful for profiling donor materials, comparing engineered cell lines, and investigating mechanisms of xenotransplantation rejection.
Why Non-Human Glycans Are a Major Xenotransplantation Barrier
Xenotransplantation is being explored to help address the shortage of human donor organs. Among potential donor species, pigs remain the most extensively studied because their organ size, breeding features, and genome engineering compatibility make them practical research models.
However, cross-species transplantation is not limited by anatomy alone. A major challenge lies at the cell surface. Donor tissues may display carbohydrate structures that humans do not produce. These non-human glycans can be recognized immediately by circulating human antibodies.
This early immune recognition is one reason glycan incompatibility remains central in pig-to-human and pig-to-primate transplantation studies. Once antibodies bind donor endothelial cells, they can initiate complement activation, inflammation, vascular injury, and coagulation abnormalities. In other words, the glycan layer is not a minor detail. It is often one of the first molecular interfaces between graft and host immunity.
Rejection Stages in Xenotransplantation
Before discussing individual glycan targets, it is helpful to understand how xenograft rejection develops across different stages.
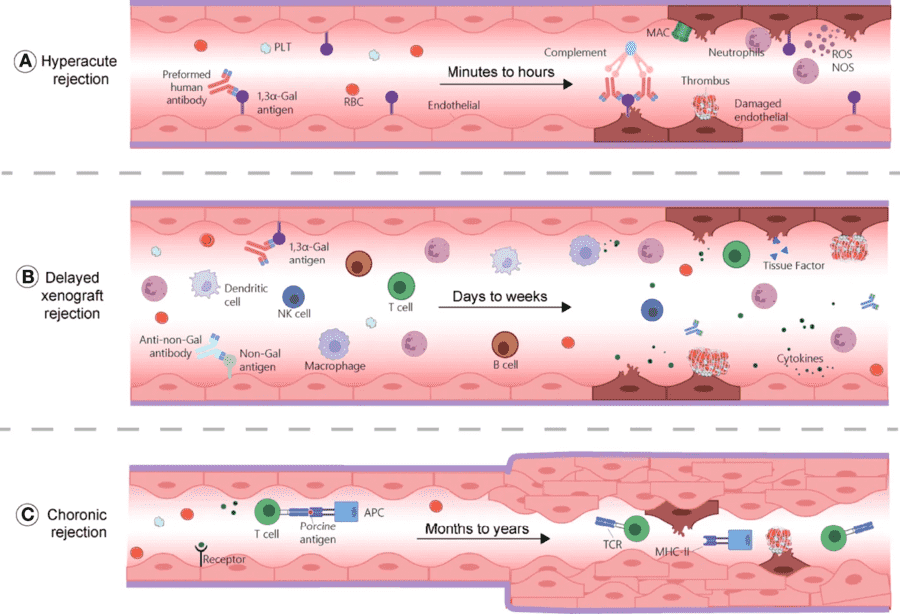 Fig.1 Mechanisms of xenotransplantation rejection.1
Fig.1 Mechanisms of xenotransplantation rejection.1
As shown above, hyperacute rejection, delayed xenograft rejection, and chronic rejection are mechanistically distinct but biologically connected. This overview creates a clear transition into the discussion of alpha-Gal, Neu5Gc, and other non-Gal xenoantigens.
Highly Exposed
Glycans are abundant on donor cell surfaces, especially on endothelial cells that directly contact recipient blood.
Naturally Immunogenic
Humans can carry pre-existing antibodies against certain non-human glycans, making early graft injury more likely.
Broad Tissue Distribution
The same xenoantigen may appear on multiple glycoproteins and glycolipids across donor tissues.
Difficult to Ignore
Even after gene editing, residual or non-dominant glycan signals can still contribute to antibody-mediated damage.
Why This Topic Extends Beyond a Single Antigen
Early xenotransplantation work often focused on one dominant glycan barrier. That view was useful, but it is no longer sufficient. As donor engineering improves, the field increasingly recognizes that glycan-mediated incompatibility is layered. Reducing one antigen may uncover the contribution of others.
This is why modern xenotransplantation studies often examine a panel of xenoantigens rather than relying on a single marker. Researchers now evaluate how multiple glycans, together with species-specific protein incompatibilities, shape overall graft immunogenicity.
Key Non-Human Glycans in Xenotransplantation Research
Among the known glycan barriers, alpha-Gal and Neu5Gc are the most widely discussed. Both are absent in humans but present in many mammals, including pigs. Non-Gal antigens such as SDa-related structures are also important in current xenotransplantation research, particularly when major donor-engineering strategies reduce dominant glycan signals.
| Target | Full Name | Why Humans Lack It | Why It Matters | Research Relevance |
|---|---|---|---|---|
| alpha-Gal | Galactose-alpha-1,3-galactose | Humans do not express active alpha-1,3-galactosyltransferase | Frequently recognized by natural human antibodies | Classical glycan xenoantigen in pig-derived tissues |
| Neu5Gc | N-glycolylneuraminic acid | Humans have an inactive CMAH gene | Can be recognized as foreign in sialylated glycan contexts | Important non-Gal target in engineered donor studies |
| SDa-related glycans | SDa-associated carbohydrate structures | Not absent from humans as a class, but porcine B4GALNT2-dependent SDa-related expression can contribute to xenogeneic mismatch | May contribute to non-Gal antibody binding | Often evaluated in studies of B4GALNT2 editing and residual xenoantigenicity |
Alpha-Gal as a Classical Xenoantigen
Alpha-Gal has historically been the best-known glycan barrier in xenotransplantation. Because humans do not express this epitope, natural anti-alpha-Gal antibodies are common. When these antibodies bind donor vascular endothelium, complement activation may follow rapidly.
This helps explain why alpha-Gal was such an early priority in donor-gene editing strategies. It also explains why alpha-Gal remains a standard benchmark in donor-cell characterization.
The same glycan is also relevant in alpha-gal syndrome. Although alpha-gal syndrome is not the same as xenotransplantation rejection, both settings illustrate the strong immunological visibility of this non-human glycan.
Neu5Gc Immunogenicity in Donor Materials
Neu5Gc immunogenicity has become increasingly important in studies that move beyond alpha-Gal. Neu5Gc differs subtly from the human sialic acid Neu5Ac, but that small structural difference can still be immunologically meaningful.
One challenge is that Neu5Gc can occur in multiple glycoconjugate settings. It is not limited to a single uniform epitope. Its abundance, tissue distribution, and molecular presentation all affect how it is detected and how strongly it may contribute to serum reactivity.
For this reason, Neu5Gc-focused research often requires highly selective antibodies, glycan-array validation, and side-by-side testing against related human-compatible sialylated structures.
How Glycan Xenoantigens Contribute to Xenotransplantation Rejection
Glycan-mediated injury is best understood as part of a broader rejection network rather than a single isolated event. Antibody binding to donor glycans can initiate early vascular damage, but the consequences extend well beyond the first antibody-graft contact.
Early Antibody Binding
Pre-existing recipient antibodies can recognize alpha-Gal, Neu5Gc, or other xenoantigens on donor endothelial cells soon after exposure.
Complement Activation
Bound antibodies may activate complement and promote membrane injury, endothelial dysfunction, and amplification of inflammatory signaling.
Cellular Recruitment
Innate immune cells such as macrophages, neutrophils, and natural killer cells can contribute to ongoing graft injury after initial antibody binding.
Coagulation Dysregulation
Endothelial damage and cross-species incompatibilities in hemostatic control can promote thrombosis and progressive graft dysfunction.
In practice, this means xenotransplantation rejection is not explained by a single assay readout. Antibody binding data, glycan profiling, complement-related markers, and tissue-level analysis all contribute to a more reliable interpretation of graft immunogenicity.
What Researchers Commonly Need to Evaluate
- Whether a donor sample still expresses detectable alpha-Gal or other non-human glycans
- Whether Neu5Gc-containing structures remain after donor engineering
- Whether antibody binding is target-specific or reflects broad cross-reactivity
- Whether engineered cells show lower serum reactivity than parental controls
- Whether the assay format is suitable for screening, validation, or mechanistic studies
Our Research Solutions for Non-Human Glycan Studies
Creative Biolabs supports glycan-focused xenotransplantation research with antibody development, glycan characterization, and analytical validation services. Our workflows are designed for scientific research use only and can help investigators move from broad immunogenicity questions to specific, testable assay strategies. These services are intended for assay development, specificity assessment, and sample characterization rather than clinical transplantation decision-making.
Non-Human Glycan Antibody Development
Custom antibody generation against alpha-Gal, Neu5Gc-related targets, and other non-human glycans for donor profiling and xenoantigen studies.
Anti-Glycan Antibody Development
Broader glycan-targeted antibody support for research groups that need monoclonal, polyclonal, or recombinant reagents for glycoimmunology applications.
Glycan Microarray
Binding profiling for specificity analysis, cross-reactivity assessment, and comparative recognition of target versus off-target glycans.
Glycosylation Analysis
Structural and compositional characterization of glycosylated samples to support donor-material evaluation and glycan verification.
SPR-Based Binding Analysis
Affinity and kinetics measurements for candidate antibodies or glycan-binding interactions in comparative research workflows.
Service Advantages for This Topic
- Targeted support for non-human glycans and related xenoantigens
- Specificity-focused screening against structurally similar glycans
- Flexible validation options for flow cytometry, ELISA, IF, and array studies
- Useful for donor-cell characterization and mechanistic glycoimmunology research
Start a Discussion About Your Project
If you are studying non-human glycans, donor engineering, or serum reactivity in xenotransplantation models, a well-designed glycan assay strategy can make downstream interpretation much clearer.
Tell us your target, sample type, intended assay, and validation goals. We can help you define a practical research workflow for glycan detection and antibody-based analysis.
Request a Project Consultation
Published Data
A 2022 review article in Frontiers in Immunology summarizes delayed xenograft rejection and notes that, beyond the classical alpha-Gal response, pre-existing antibodies against non-Gal epitopes such as Neu5Gc and the SDa blood group are implicated in rejection.
The review also presents delayed xenograft rejection as a broader process that includes acute humoral injury, cellular xenograft rejection, and coagulation dysregulation. This context is useful when interpreting how residual xenoantigenicity may remain relevant after major donor-engineering efforts reduce dominant glycan signals.
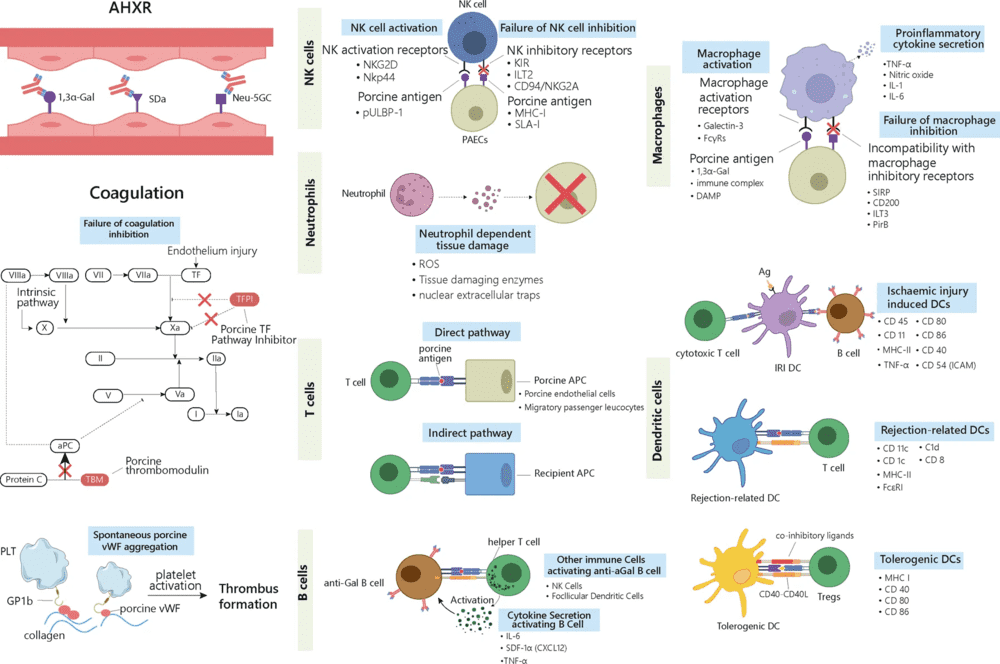 Fig.2 Non-Gal xenoantigens in delayed xenograft rejection.1
Fig.2 Non-Gal xenoantigens in delayed xenograft rejection.1
In particular, Figure 2 labels Neu5Gc and SDa as representative non-Gal epitopes in delayed xenograft rejection and places them within a wider framework that also includes innate immune activation, adaptive immune responses, and thrombotic changes in the graft.
FAQs
Why are non-human glycans important in xenotransplantation studies?
Because humans do not produce certain donor glycans, these structures can behave as xenoantigens and trigger antibody binding, complement activation, and graft injury.
Is alpha-Gal still relevant when donor pigs are genetically engineered?
Yes. Alpha-Gal remains a key benchmark antigen. Even after its reduction, other non-Gal targets may still contribute to residual serum reactivity.
Why is Neu5Gc immunogenicity difficult to evaluate?
Neu5Gc is structurally close to human Neu5Ac, and it can appear in different glycan contexts. This makes high-specificity reagents and careful controls especially important.
Does alpha-gal syndrome mean the same thing as xenotransplantation rejection?
No. They are different immune settings, but both involve recognition of alpha-Gal as a non-human glycan.
Can you develop antibodies against a specific xenoantigen?
Yes. We support custom antibody development against defined non-human glycan targets for research applications.
Which assay formats are useful for this type of project?
Common options include glycan arrays, ELISA, flow cytometry, immunostaining, and binding-kinetics analysis, depending on the research question.
Are these services intended for clinical transplantation decisions?
No. These services are for scientific research use only and are not intended for clinical diagnosis or treatment.
Reference:
- Zhou, Qiao, Ting Li, Kaiwen Wang, Qi Zhang, Zhuowen Geng, Shaoping Deng, Chunming Cheng, and Yi Wang. Current status of xenotransplantation research and the strategies for preventing xenograft rejection. Frontiers in Immunology 13 (2022): 928173. Distributed under Open Access license CC BY 4.0, without modification. https://doi.org/10.3389/fimmu.2022.928173
