Anti-β-Gal Antibody Development Service
Glycobiology is a rapidly expanding frontier in biomedical research, revealing critical insights into cell signaling, immune recognition, and disease progression. Among the diverse array of carbohydrate structures, β-galactose (β-Gal) residues—specifically terminal structures like Galβ1-4GlcNAc (LacNAc) or Galβ1-3GlcNAc—serve as vital markers in physiological and pathological contexts. These epitopes are often implicated in lysosomal storage diseases (e.g., GM1 gangliosidosis) and are emerging as robust biomarkers for cellular senescence (SA-β-gal). At Creative Biolabs, we leverage our deep expertise in glyco-immunology to provide specialized antibody development services. Drawing on the capabilities of our Non-Human Glycan Antibody Development platform, we offer precision-engineered solutions for targeting β-Gal epitopes, facilitating advanced research into glycosylation pathways and age-related pathologies.
Understanding the Beta-Gal Epitope in Research
The beta-gal epitope typically refers to terminal galactose residues linked via a beta-glycosidic bond to an underlying sugar, most commonly N-acetylglucosamine (GlcNAc). This creates the Type 2 (Galβ1-4GlcNAc) or Type 1 (Galβ1-3GlcNAc) lactosamine units. In healthy physiology, these structures are often capped by sialic acid or fucose. However, in specific disease states—such as lysosomal dysfunction or cellular senescence—these terminal β-Gal residues can become exposed or accumulated.
Key Research Applications
- Lysosomal Storage Disorders: In conditions like GM1 gangliosidosis or Morquio B disease, deficiency in the enzyme β-galactosidase (GLB1) leads to the toxic accumulation of β-galactose-terminated substrates (e.g., GM1 ganglioside, keratan sulfate) within lysosomes.
- Senescence-Associated β-Galactosidase (SA-β-gal): While traditionally detected via enzymatic activity at pH 6.0, the upregulation of lysosomal β-galactose is also accompanied by changes in surface glycan signatures, making anti-glycan antibodies valuable tools for identifying senescent cells in flow cytometry or immunofluorescence assays.
- Glycan Biosynthesis Analysis: Monitoring the expression of poly-N-acetyllactosamine (poly-LacNAc) chains, which act as scaffolds for selectin ligands, is crucial for understanding cell adhesion and trafficking.
The Challenge: Developing Specific Anti-Glycan Antibodies
Generating a high-affinity anti-glycan antibody against β-Gal structures presents unique obstacles that traditional protein-based immunization strategies often fail to overcome.
Low Immunogenicity
Carbohydrates are T-cell independent antigens. They typically induce a weak IgM response with no affinity maturation or memory B-cell formation, making it difficult to generate high-affinity IgG antibodies.
Structural Similarity
The β-Gal epitope (Galβ1-4GlcNAc) is structurally similar to the α-Gal epitope (Galα1-3Gal). Antibodies must be rigorously screened to ensure they do not cross-react with alpha-gal or other abundant galactose-containing glycans.
Carrier Dependence
The presentation of the glycan matters. A simple sugar hapten behaves differently than when it is presented on a lipid (glycolipid) or protein (glycoprotein) backbone, requiring specialized conjugation strategies for immunization.
Our Custom Antibody Development Solutions
Creative Biolabs addresses these challenges through a proprietary glycan immunization platform. We offer a comprehensive suite of services to generate antibodies that specifically recognize the spatial conformation of β-Gal terminal structures.
Monoclonal Antibody Development
We utilize specialized hybridoma technology optimized for carbohydrate antigens. By using potent adjuvants and glycan-peptide conjugates (e.g., KLH-LacNAc), we can drive the immune system to produce high-affinity IgG isotypes. We also employ electrofusion to increase hybridoma yield.
Polyclonal Antibody Production
For broader detection needs, we offer polyclonal antibody services in rabbits, goats, or chickens. This is often a cost-effective solution for initial screening or for assays like agglutination where multivalent binding is advantageous.
Custom Antibody via Phage Display
To bypass immune tolerance mechanisms completely, we offer synthetic human or naïve phage display library screening. This allows us to select binders (scFv, Fab, or VHH) against specific glycan targets in vitro, ensuring precise epitope mapping without animal immunization.
Analytical & Validation Services
We don't just deliver a tube of antibody; we deliver confidence. We validate our anti-β-Gal antibodies using glycan microarrays to profile specificity against hundreds of related structures, ensuring no cross-reactivity with blood group antigens or other gal-terminating glycans.
Core Advantages
High Specificity
Rigorous counter-screening against Alpha-Gal and other isomers to ensure precise target recognition.
Diverse Formats
Full flexibility to deliver IgG, IgM, Fab fragments, or scFv depending on your assay requirements.
Data-Driven
Every antibody comes with a comprehensive COA, including ELISA, Dot Blot, or Flow Cytometry data.
Expert Support
PhD-level project managers guide you from antigen design to final validation.
Inquire Now
Project Workflow
Published Data
The lysosomal enzyme β-galactosidase (GLB1), distinctly recognized as Senescence-Associated β-Galactosidase (SA-β-gal) when active at pH 6.0, has transcended its traditional role in cell biology to become a pivotal biomarker in diagnostic pathology. A systematic 2022 review rigorously evaluates the biological mechanism and clinical applications of this enzyme, highlighting its specific accumulation in lysosomes during cellular senescence. The analysis reveals that SA-β-gal expression is significantly upregulated in a diverse array of pathological conditions, ranging from precancerous lesions to invasive carcinomas of the colon, ovary, and prostate. Importantly, the literature elucidates the phenomenon of therapy-induced senescence (TIS), where chemotherapeutic agents trigger a surge in SA-β-gal activity, marking it as a vital indicator for monitoring treatment efficacy and tumor dormancy. While conventional enzymatic assays are limited by the requirement for fresh tissue and strict acidity controls, the distinct overexpression of GLB1 protein and its associated glycan structures in senescent cells provides a stable foundation for immunological detection. This supports the utility of specialized anti-glycan antibodies that can precisely target these senescence-associated epitopes in fixed tissue specimens, facilitating the visualization of tumor margins and the assessment of senescence-associated secretory phenotypes (SASP) without the constraints of metabolic activity assays.
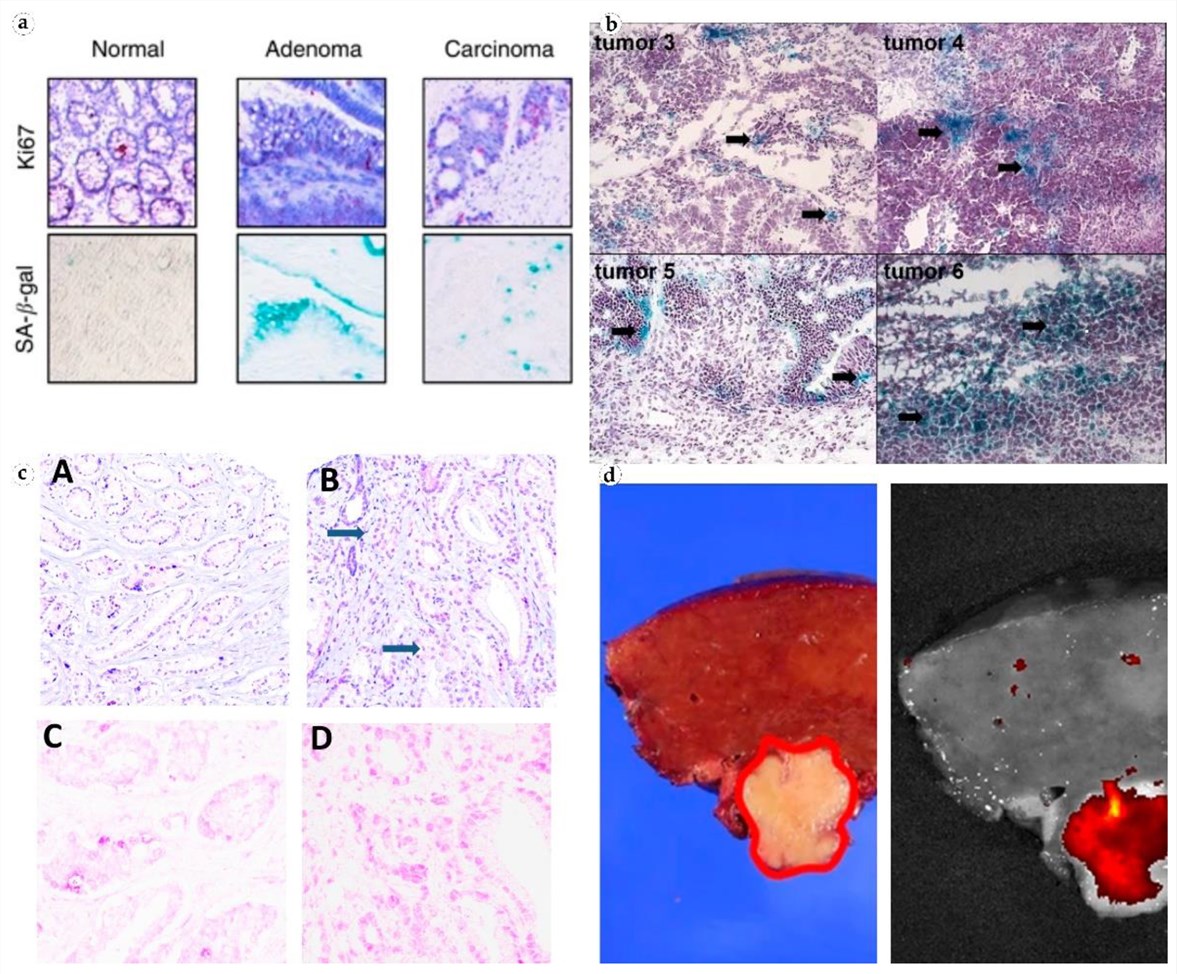 Fig.1
Multi-tissue visualization of Senescence-Associated β-Galactosidase (SA-β-gal) accumulation
distinguishing senescent neoplastic cells from normal tissue in colorectal, ovarian, and
prostate malignancies.1
Fig.1
Multi-tissue visualization of Senescence-Associated β-Galactosidase (SA-β-gal) accumulation
distinguishing senescent neoplastic cells from normal tissue in colorectal, ovarian, and
prostate malignancies.1
FAQs
How do you ensure the antibody doesn't cross-react with Alpha-Gal?
We employ a rigorous negative screening process. All candidate clones are tested against a panel of structurally similar glycans, including Galα1-3Gal (Alpha-Gal). Only clones that bind strongly to the β-Gal target and show no binding to Alpha-Gal are selected for development.
What is the difference between this antibody and an Anti-Beta-Galactosidase antibody?
An "Anti-Beta-Galactosidase antibody" typically targets the enzyme protein (GLB1). Our service develops "Anti-β-Gal antibodies" that target the carbohydrate structure (the sugar epitope) itself, regardless of which protein it is attached to. This is crucial for studying glycosylation patterns rather than just protein expression.
Can these antibodies be used for Flow Cytometry on live cells?
Yes. Since the β-Gal epitopes (like LacNAc) are often expressed on cell surface glycoproteins and glycolipids, our antibodies are suitable for flow cytometry. We can validate them using positive control cell lines known to express high levels of terminal β-galactose.
Do you offer IgM or IgG antibodies?
We can produce both. While natural anti-glycan antibodies are often IgM, our specialized immunization protocols and hybridoma screening can successfully isolate high-affinity IgG clones, which are generally more stable and easier to use in various applications.
What is the timeline for a custom project?
A typical monoclonal antibody development project takes approximately 4-6 months, including antigen synthesis, immunization, and screening. Polyclonal projects are faster, typically taking 2-3 months.
What Our Customers Say
"We needed a specific binder for the Type 2 LacNAc structure to distinguish it from Type 1 chains. Creative Biolabs delivered an IgG clone with excellent specificity. The dot blot data they provided was very convincing and saved us a lot of validation time."
"Targeting senescent cells via surface glycans was a new approach for us. The custom polyclonal antibody we ordered worked perfectly in our flow cytometry setup. It correlated beautifully with our SA-beta-gal enzymatic staining."
"The main issue with anti-glycan antibodies is usually low affinity. The monoclonal antibody developed by this team, however, has high avidity and works well even in low concentrations. Professional service and great communication."
"We utilized their phage display service to find a binder for a synthetic beta-gal derivative. The turnaround was impressive, and the absence of animal immunization protocols simplified our regulatory paperwork immensely."
Reference:
- Valieva, Y., et al. "Senescence-Associated β-Galactosidase Detection in Pathology." Diagnostics 12.10 (2022): 2309. Distributed under Open Access license CC BY 4.0. https://doi.org/10.3390/diagnostics12102309
