Anti-Neu5Gc Antibody Development Service
Creative Biolabs stands at the forefront of glyco-immunology, offering a comprehensive suite of solutions for the generation of high-affinity reagents against complex carbohydrate antigens. Through our specialized Non-Human Glycan Antibody Development platform, we have established a dedicated pipeline for the development of antibodies targeting N-glycolylneuraminic acid (Neu5Gc). Neu5Gc is a prominent xenoantigen with profound implications in oncology, xenotransplantation, and biotherapeutic safety. Due to the extreme structural similarity between Neu5Gc and the human-native N-acetylneuraminic acid (Neu5Ac), generating antibodies that can strictly discriminate between these two sialic acids is a formidable challenge. Our service leverages genetically engineered host models and advanced subtractive screening technologies to deliver monoclonal antibodies with exquisite specificity, empowering researchers to decipher the role of xenosialitis in human disease without the interference of cross-reactivity.
Background: The Neu5Gc Enigma
Sialic acids are a diverse family of nine-carbon alpha-keto acids that typically terminate the glycan chains on the cell surface of vertebrates. They play critical roles in cell-cell recognition, signaling, and pathogen interaction. In most mammals, including great apes, the two most common forms of sialic acid are N-acetylneuraminic acid (Neu5Ac) and its hydroxylated derivative, N-glycolylneuraminic acid (Neu5Gc). The conversion of Neu5Ac to Neu5Gc is catalyzed by the enzyme cytidine monophosphate-N-acetylneuraminic acid hydroxylase (CMAH). However, in the human lineage, an inactivating mutation in the CMAH gene occurred approximately two to three million years ago, rendering humans unable to synthesize Neu5Gc endogenously.
Structural Comparison: Neu5Ac vs. Neu5Gc
The structural distinction between these two molecules is incredibly subtle, yet it has massive immunological consequences. This single oxygen atom difference is the sole basis for the antigenicity of Neu5Gc in humans.
| Feature | Neu5Ac (Human Native) | Neu5Gc (Xenoantigen) |
|---|---|---|
| C5 Substituent | N-acetyl group (-NHCOCH3) | N-glycolyl group (-NHCOCH2OH) |
| Biosynthesis in Humans | Yes (Endogenous) | No (CMAH gene inactive) |
| Dietary Source | Synthesized by body | Red meat, dairy products |
| Immunogenicity | Non-immunogenic (Self) | Highly immunogenic (Non-self) |
Dietary Incorporation and Xenosialitis
Despite the genetic loss, Neu5Gc is not entirely absent from the human body. It acts as a dietary-derived xenoantigen that integrates into human glycans:
- Metabolic Incorporation: Exogenous Neu5Gc from dietary sources (especially red meat) is taken up by human cells and incorporated into surface glycoconjugates.
- Antibody Response: Humans produce circulating "xeno-autoantibodies" against Neu5Gc due to exposure to dietary sources and certain bacteria early in life.
- Chronic Inflammation: The interaction between these circulating antibodies and Neu5Gc-laden tissues triggers a chronic inflammatory state known as xenosialitis. This process is increasingly linked to the promotion of carcinomas and the aggravation of atherosclerosis.
Critical Pain Points in Current Research Reagents
The development of reagents to study Neu5Gc has been historically plagued by specificity issues. The chemical difference between the human-native Neu5Ac and the xenoantigen Neu5Gc is miniscule—a single oxygen atom at the C5 position of the pyranose ring. This subtle structural variation represents a significant immunological hurdle for any antibody development service provider.
Polyclonal Cross-Reactivity
Traditional immunization often yields polyclonal sera that recognize the conserved core structure of sialic acids rather than the specific N-glycolyl modification. This results in significant binding to Neu5Ac, causing high background noise and false positives.
Immunological Tolerance
Because Neu5Gc is a self-antigen in many common host species (e.g., wild-type mice), the immune system eliminates high-affinity B-cell clones through tolerance mechanisms. This makes it difficult to raise a robust immune response using standard hybridoma protocols.
Endogenous Interference
Detecting Neu5Gc in human clinical samples requires reagents that can compete effectively with endogenous human xeno-autoantibodies. Many commercial antibodies lack the affinity to displace these naturally occurring competitors.
Context Dependency
The presentation of Neu5Gc can vary based on the underlying glycan tree (linkage type, branching). Poorly characterized antibodies may bind Neu5Gc only in specific contexts, leading to inconsistent data across different tissue types.
Precision Engineering Workflow
To address these challenges, we utilize a specialized workflow that combines genetic engineering with high-throughput screening. By using Cmah-/- knockout mice or phylogenetically distant species like chickens, we bypass immunological tolerance to generate high-affinity binders.
Anti-Neu5Gc Antibody Development Services
Creative Biolabs focuses on the development of high-affinity, low cross-reactivity antibodies specifically targeting the N-glycolylneuraminic acid (Neu5Gc) xenoantigen. Our comprehensive service platform supports advanced research into red meat-associated inflammation, tumor microenvironment (TME) characterization, and immune response monitoring in humanized animal models.
High-Affinity Monoclonal Antibody Generation
Leveraging Cmah-/- knockout mice, we generate monoclonal antibodies (mAbs) with sub-nanomolar affinity. These reagents are designed to detect trace levels of Neu5Gc in human tissues, essential for studying red meat-induced inflammation and TME accumulation without interference from native Neu5Ac.
Humanized Model Research Reagents
We develop specialized antibodies validated for use in humanized mouse models (e.g., Neu5Gc-deficient mice engrafted with human tumors). These tools are critical for tracking the immune clearance of Neu5Gc-positive grafts and modeling human xenosialitis in vivo.
Recombinant & Therapeutic Engineering
For projects aiming to exploit Neu5Gc as a therapeutic target, we offer antibody humanization and affinity maturation services. We engineer antibodies with reduced immunogenicity for potential use in targeting Neu5Gc-rich tumors via ADCC or ADC mechanisms.
Biotherapeutic Quality Control (QC) Tools
We produce highly sensitive antibodies for the detection of Neu5Gc contamination in recombinant biologics produced in non-human cells (CHO). These reagents serve as robust QC tools to ensure the safety of biopharmaceuticals intended for human use.
Inquire about Anti-Neu5Gc Antibodies
Key Research Applications
Our specificity-validated antibodies are empowering cutting-edge research across multiple disciplines, from nutritional epidemiology to cancer immunology.
Red Meat-Associated Inflammation
Neu5Gc is the primary molecule linking red meat consumption to increased cancer risk and atherosclerosis. Our antibodies enable:
- Precise immunohistochemical staining of Neu5Gc accumulation in colon and vascular tissues.
- Quantification of dietary Neu5Gc incorporation in long-term feeding studies.
- Investigation of "Xenosialitis" pathways activated by Neu5Gc-antibody immune complexes.
Tumor Microenvironment (TME) Profiling
Neu5Gc accumulates metabolically in rapidly growing tumors, often exceeding levels in normal tissue. Our reagents support:
- Differentiation of tumor vs. normal tissue glycosylation patterns.
- Targeting Neu5Gc as a specific tumor-associated carbohydrate antigen (TACA).
- Mapping the distribution of Neu5Gc in hypoxic regions of the TME.
Why Choose Creative Biolabs?
Exclusive Specificity
Discrimination between Neu5Gc and Neu5Ac at the single-atom level.
Advanced Hosts
Utilization of Cmah-/- knockout mice and avian systems to bypass tolerance.
Data Ownership
Full IP transfer with hybridoma sequencing and validation reports.
Multi-Platform Validation
Rigorous testing via SPR, Glycan Microarray, and cell-based binding assays.
Published Data
Recent advancements in glyco-immunology have validated the therapeutic potential of targeting the tumor-specific antigen GM3(Neu5Gc) using humanized antibodies. A pivotal study addressed the challenge of evaluating these therapies given the natural absence of the functional cytidine monophospho-N-acetyl-neuraminic acid hydroxylase (CMAH) enzyme in human cells. By stably transfecting the murine Cmah gene into human ovarian carcinoma cells, researchers successfully engineered a cell line that expresses surface GM3(Neu5Gc), thereby mimicking the accumulation of this xenoantigen seen in human tumors due to metabolic incorporation. Flow cytometric analysis confirmed the exclusive binding of the humanized antibody, 14F7hT, to these Cmah-expressing cells, demonstrating exquisite specificity without cross-reactivity to native acetylated gangliosides. Furthermore, the study provided compelling evidence of biological activity: the antibody induced robust antibody-dependent cell-mediated cytotoxicity (ADCC) with human effector cells. In a decisive in vivo assessment, systemic administration of the antibody significantly suppressed tumor growth in mice bearing these engineered human xenografts. This data underscores the efficacy of high-affinity anti-Neu5Gc antibodies in arresting tumor progression and highlights the strategic value of using Cmah-engineered models to rigorously screen and validate xenoantigen-specific immunotherapeutics for clinical development.
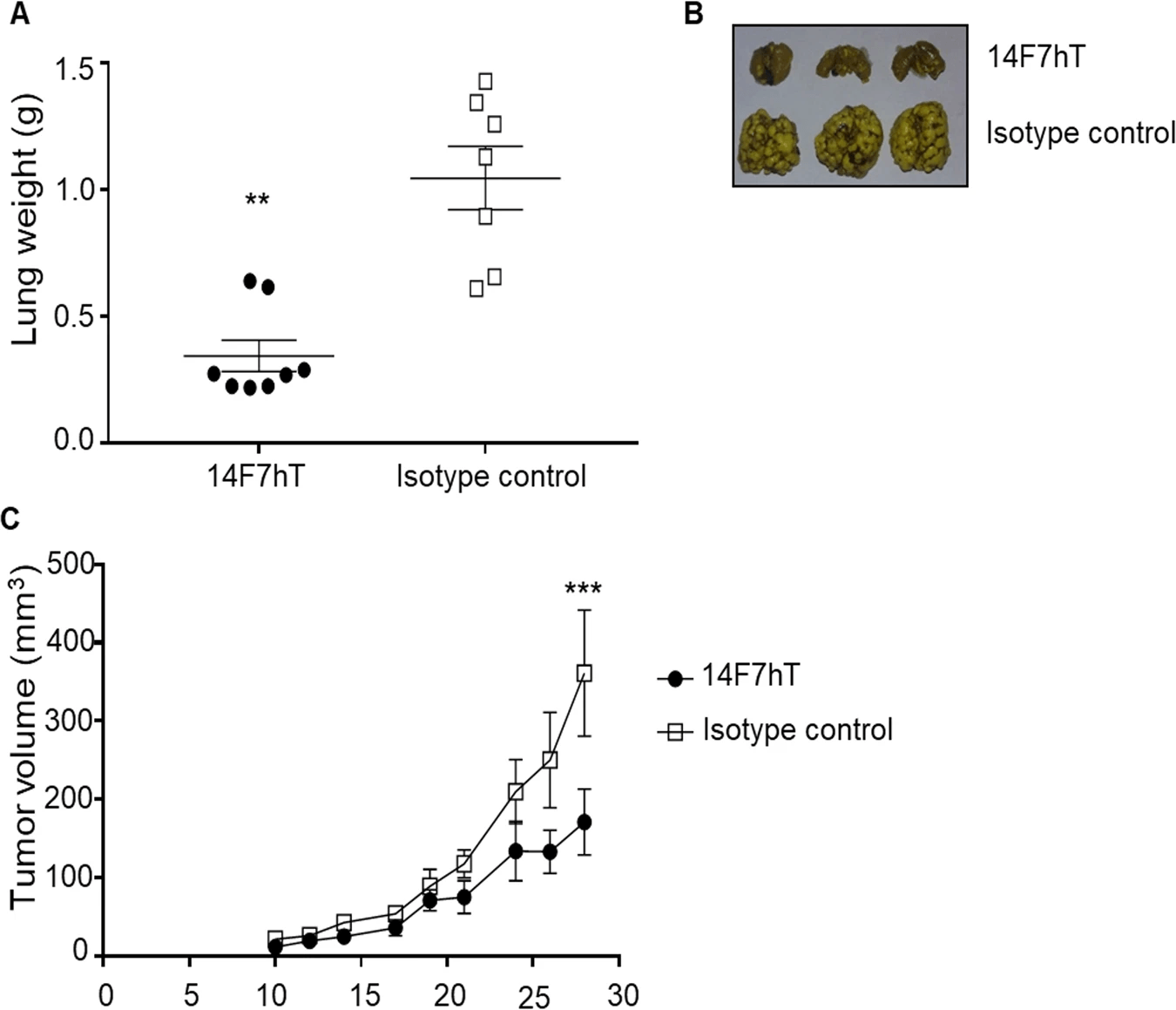
Fig.1 In vivo antitumor effect of the humanized anti-Neu5Gc antibody (14F7hT)
against Cmah-transfected human ovarian cancer cells.1
FAQs
Why is it necessary to use Cmah-/- mice for immunization?
Wild-type mice express Neu5Gc naturally as a "self" antigen. Therefore, their immune system is tolerant to it and will delete B-cells capable of producing high-affinity anti-Neu5Gc antibodies. Cmah-/- mice lack the enzyme to produce Neu5Gc, making them see it as a foreign antigen, which allows for a robust immune response.
How do you ensure the antibody doesn't cross-react with human Neu5Ac?
We employ a strict subtractive screening strategy (negative selection). After identifying binders to Neu5Gc, we counter-screen every clone against a Neu5Ac glycan array. Any clone showing binding to Neu5Ac above background levels is discarded, ensuring only highly specific antibodies are selected.
How does the specificity of your antibodies compare to lectins?
Lectins are often broad-spectrum binders that may recognize multiple sialic acid linkages or contexts. Our monoclonal antibodies are engineered to specifically recognize the N-glycolyl moiety, offering significantly higher specificity and affinity compared to traditional lectin-based detection, especially in complex biological matrices like tissue sections.
Do you offer matched antibody pairs for sandwich ELISA?
Yes, we can screen for matched pairs. Since Neu5Gc is a small hapten, standard sandwich formats are challenging unless the target is a multi-valent glycoprotein. We typically design competitive ELISA formats or screen for pairs that recognize distinct epitopes on Neu5Gc-containing glycoproteins if required for your specific assay.
Can you provide Neu5Gc-negative control cells for validation?
Yes, we can include validation data using CMAH-knockout human cell lines (true negatives) versus wild-type animal cells or human cells fed with exogenous Neu5Gc (positives). This ensures the antibody is truly detecting the antigen and not non-specific cellular components.
Is it possible to label the antibodies for direct imaging?
Absolutely. We offer conjugation services to label your custom anti-Neu5Gc antibodies with fluorophores (e.g., FITC, PE, APC), biotin, or HRP. This is particularly useful for flow cytometry or direct immunofluorescence studies in tumor microenvironment research.
Can these antibodies be used for clinical diagnostic purposes?
Our custom antibodies are developed for Research Use Only (RUO). However, we can perform antibody humanization and extensive validation to support your downstream development of In Vitro Diagnostic (IVD) kits or therapeutic candidates under appropriate regulatory frameworks.
What is the typical timeline for a custom monoclonal project?
A standard hybridoma development project using Cmah-/- mice typically takes approximately 4-6 months. This includes antigen preparation, immunization, fusion, screening, subcloning, and production. Expedited services may be available depending on project complexity.
Do you provide the antibody sequence?
Yes, for our custom development services, we can perform hybridoma sequencing and provide the full variable region sequences (VH and VL), granting you complete intellectual property ownership of the reagent.
Reference:
- Dorvignit, D.; et al. "Antitumor effects of the GM3(Neu5Gc) ganglioside-specific humanized antibody 14F7hT against Cmah-transfected cancer cells." Scientific Reports 9.1 (2019): 9921. Distributed under Open Access license CC BY 4.0. https://doi.org/10.1038/s41598-019-46148-1
What Our Customers Say
"We had struggled for years with commercial antibodies that cross-reacted with human sialic acids. The clones generated by Creative Biolabs showed clean separation in our flow cytometry assays. Their use of Cmah knockout mice really made the difference."
"The technical support team was incredibly knowledgeable about glycan chemistry. They helped us design an antigen strategy that resulted in highly specific IgY antibodies. The data package provided with the final product was comprehensive."
"Using their anti-Neu5Gc antibodies, we were finally able to visualize the accumulation of dietary Neu5Gc in our inflammation models. The staining was specific with virtually no background on control tissues. A fantastic tool for nutrition research."
"The humanized antibody variants provided by Creative Biolabs performed exceptionally well in our xenograft models. They showed potent ADCC activity against Neu5Gc-positive tumors. We are moving forward with these candidates."
