Anti-Heparanase Antibody Development Service
Heparanase (HPSE) acts as a critical modulator of the extracellular matrix (ECM), making it a high-value target in oncology and inflammatory research. As a leader in the field of glyco-biology, Creative Biolabs offers comprehensive solutions through our Anti-Glycan Related Enzyme Antibody Development Service. We specialize in generating highly specific antibodies that target the active site or unique epitopes of heparanase, facilitating research into tumor metastasis, angiogenesis, and immune cell trafficking.
The Biology of Heparanase: A Master Regulator of ECM Remodeling
Heparanase is an endo-β-D-glucuronidase and is the only mammalian enzyme capable of cleaving heparan sulfate (HS) side chains of heparan sulfate proteoglycans (HSPGs). This activity is distinct from bacterial heparinases and plays a fundamental role in both normal physiology and disease pathology. The enzyme is initially synthesized as a latent 65 kDa precursor (pro-heparanase) that undergoes proteolytic processing in the lysosome. A linker segment is excised, yielding an active heterodimer composed of an 8 kDa subunit and a 50 kDa subunit.
The functional implications of heparanase activity are vast. By degrading HS in the basement membrane and ECM, heparanase loosens the structural barrier that confines cells, thereby permitting tumor cell invasion and metastasis. Furthermore, the ECM serves as a reservoir for heparin-binding growth factors such as VEGF, FGF, and HGF. Heparanase-mediated cleavage releases these sequestered factors, increasing their bioavailability and driving angiogenesis and tumor growth. Beyond its enzymatic role, heparanase also exerts non-enzymatic functions, including signaling through syndecan clustering and promoting autophagy via the TFEB pathway, which enhances chemoresistance in cancer cells.
Research Applications of Anti-Heparanase Antibodies
Given its dual role in enzymatic degradation and signaling, antibodies targeting heparanase are powerful tools for dissecting disease mechanisms. They are extensively used to study:
- Cancer Metastasis: Investigating the breakdown of physical barriers in the tumor microenvironment.
- Angiogenesis: Studying the release kinetics of VEGF and FGF from the ECM.
- Inflammation: Analyzing immune cell diapedesis and trafficking through vessel walls.
- Viral Infection: Understanding how viruses like HSV utilize HS for cellular entry.
- Kidney Disease: Exploring the role of glomerular basement membrane degradation in proteinuria.
Challenges in Developing Anti-Heparanase Antibodies
Developing high-affinity, neutralizing antibodies against heparanase presents several unique hurdles that standard antibody generation protocols often fail to address. The complexity of the enzyme's structure and its biological regulation requires a sophisticated approach to immunogen design and screening.
Active Site Access
The catalytic cleft of heparanase is a conserved pocket that can be difficult to target. Antibodies must sterically occlude this site or bind to the heparin-binding domains (HBDs) to effectively neutralize enzymatic activity.
Latent vs. Active
The latent 65 kDa pro-enzyme and the active heterodimer share many epitopes. Generating antibodies that specifically recognize the active form—essential for functional inhibition studies—requires precise counter-selection strategies.
HPA2 Homology
Heparanase-2 (HPA2) is a close homolog that lacks enzymatic activity but shares significant structural similarity. Antibodies must be rigorously screened to avoid cross-reactivity, ensuring that observed effects are due to HPA1 inhibition.
Cellular Uptake
Heparanase is internalized via cell surface receptors like LRP1 and syndecans. Antibodies intended to block non-enzymatic signaling must be capable of interfering with these specific receptor-ligand interactions.
Our Comprehensive Development Services
Creative Biolabs addresses these challenges through a specialized platform dedicated to anti-glycosidase antibody production. We utilize a combination of rational antigen design and high-throughput functional screening to deliver reagents that meet the rigorous demands of modern biomedical research.
Rational Immunogen Design
We design immunogens based on the crystal structure of the active heparanase heterodimer. This includes synthesizing peptides derived from the linker excision site (Gln157-Gln158) to target the active form specifically, as well as expressing recombinant protein with specific mutations to lock the enzyme in an open conformation for better epitope exposure.
Advanced Screening Platforms
Our screening workflow goes beyond simple binding. We employ enzyme neutralization assays using degradation-sensitive probes to identify clones that physically block HS cleavage. We also utilize flow cytometry to select antibodies that bind cell-surface heparanase or block its internalization.
Multi-Format Engineering
We can generate anti-heparanase antibodies in various formats, including full-length IgG, scFv, Fab, and VHH). VHHs are particularly useful for accessing the narrow catalytic cleft of the enzyme, a feat often difficult for conventional antibodies.
Specificity Validation
All candidates undergo rigorous cross-reactivity testing against HPA2 and other carbohydrate active enzyme antibodies targets. We also validate performance in Western Blot (distinguishing 65kDa vs 50kDa subunits) and IHC on tissue microarrays.
Request a Quote for Heparanase Antibody Development
Service Highlights
Neutralizing Potency
Antibodies screened for their ability to specifically block heparan sulfate degradation activity in vitro.
Epitope Specificity
Clear distinction between latent pro-heparanase (65 kDa) and the active heterodimer (50+8 kDa).
Species Cross-Reactivity
Options for antibodies that cross-react with mouse models, facilitating translational in vivo studies.
Advanced Validation
Validated via ECM degradation assays, invasion assays, and receptor internalization studies.
Published Data
A recent study highlights the development of a novel high-affinity monoclonal antibody (mAb), identified as A54, designed to specifically neutralize heparanase enzymatic activity. Unlike non-specific heparin-mimicking compounds that often induce adverse off-target effects, this IgG1 antibody demonstrates precise targeting capabilities. In vitro validation using sulfate-labeled extracellular matrix degradation assays confirmed that the antibody potently inhibits the cleavage of heparan sulfate side chains. Moreover, it was observed to interfere with the cellular uptake of the latent pro-enzyme, effectively blocking its conversion into the active form within lysosomes.
Structural elucidation through X-ray crystallography mapped the antibody's binding epitope to a region adjacent to the heparin-binding domain II (HBD-II) of the enzyme. This binding induces steric hindrance that occludes the active site cleft, preventing the substrate from accessing the catalytic groove. The study further evaluated the antibody's therapeutic potential in vivo, demonstrating a marked attenuation of tumor growth and metastasis in xenograft models of myeloma and glioma. Additionally, synergistic effects were observed when the antibody was administered alongside standard chemotherapies, such as gemcitabine, in pancreatic cancer models. These findings underscore the potential of developing specific anti-heparanase antibodies as a viable strategy to disrupt tumor-microenvironment cross-talk and inhibit cancer progression.
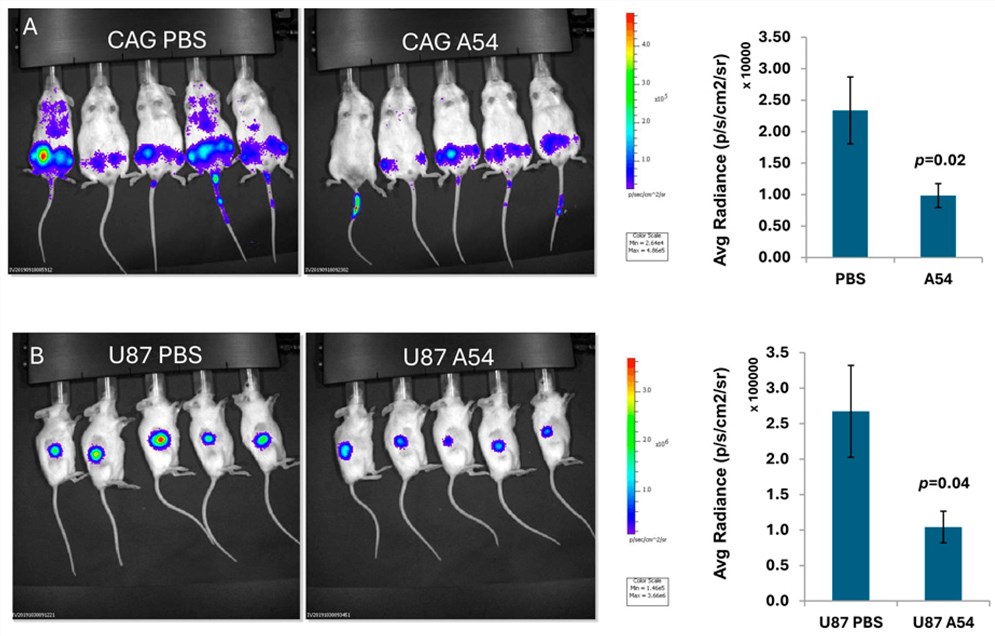 Fig.1 Anti-heparanase neutralizing antibody efficacy in xenograft models.1
Fig.1 Anti-heparanase neutralizing antibody efficacy in xenograft models.1
FAQs
How do you ensure the antibody neutralizes enzymatic activity?
We employ specific enzymatic assays where recombinant heparanase is incubated with sulfate-labeled extracellular matrix (ECM) or synthetic HS substrates. A neutralizing heparanase antibody will significantly reduce the release of degraded HS fragments compared to isotype controls.
Can you generate antibodies that distinguish between latent and active heparanase?
Yes. We can design immunogens targeting the linker segment (excised during activation) to detect only the latent form. Conversely, we can target the conformational epitopes unique to the 8+50 kDa heterodimer to specifically detect the active enzyme.
Does the antibody cross-react with Heparanase-2 (HPA2)?
Cross-reactivity is a major concern due to high homology. We include a counter-screening step using recombinant HPA2 to deplete cross-reactive binders, ensuring that the final antibody is specific to HPA1.
What species can you raise antibodies in?
We offer development in mice and rabbits for monoclonal antibodies. For specialized applications requiring deep tissue penetration or access to cryptic clefts, we also offer anti-glycosidase antibody development in llamas (VHH).
Are these antibodies suitable for flow cytometry?
Yes, we can screen for clones that bind to cell-surface associated heparanase. This is particularly useful for studying the non-enzymatic functions of the protein, such as receptor clustering and internalization.
What Our Customers Say
"We had struggled to generate a neutralizing antibody for heparanase for over a year. Creative Biolabs proposed a unique immunization strategy using the heterodimer. The resulting clones showed potent inhibition in our invasion assays. Exceptional scientific insight."
"The specificity of the antibody we received was impressive. It cleanly detected the 50kDa subunit in Western blots without cross-reacting with the latent form or HPA2. This has been crucial for our mechanism of action studies."
"Developing an anti-heparanase antibody for mouse models is challenging due to homology. Their team successfully delivered a surrogate antibody that works perfectly in our syngeneic models. Highly recommended service."
"The customized validation report was very detailed. They didn't just show ELISA data but included functional blocking assays which gave us confidence to move directly into in vivo experiments. Great turnaround time."
Reference:
- Barash, U.; Farhoud, M.; Odeh, M.; Huberman, E.; Wu, L.; Vlodavsky, I. "Heparanase-Neutralizing Monoclonal Antibody (mAb A54) Attenuates Tumor Growth and Metastasis." Cells 2025, 14, 1379. Distributed under Open Access license CC BY 4.0. https://doi.org/10.3390/cells14171379
Supports
- Glycosyltransferase & Glycosidase Substrate Microarray
- Glycosylation Analysis
- Custom Glycosylation of Biomolecules
