Anti-Glycosyltransferase Antibody Development Service
Within the specialized field of glycobiology, precise tools are essential for studying the enzymes responsible for glycan synthesis. As a part of our broader Anti-Glycan Related Enzyme Antibody Development Service, Creative Biolabs provides a dedicated platform for generating high-specificity antibodies against glycosyltransferases. This includes critical targets such as xylosyltransferase I (XT-I), sialyltransferases (ST), and fucosyltransferases (FUT), which are pivotal in the biosynthesis of proteoglycans and glycoproteins associated with tumor microenvironments, such as in salivary gland adenoid cystic carcinoma and other malignancies.
Background: The Architects of the Glycome
Glycosyltransferases (GTs) are the creators of the biological code, catalyzing the transfer of saccharide moieties from activated nucleotide sugars to specific acceptor molecules—proteins, lipids, or growing glycan chains. This immense superfamily of enzymes, including the CAZyme antibody targets, dictates the structural diversity of the glycome. From the fucosyltransferase antibody targets that regulate core fucosylation to the sialyltransferase antibody targets that cap glycans with sialic acid, these enzymes are central to cell-cell adhesion, immune recognition, and signal transduction.
Functional Diversity & Clinical Relevance
The dysregulation of GT expression is a hallmark of many pathologies. For instance, aberrant expression of FUT8 (alpha-1,6-fucosyltransferase) drives malignant phenotypes in various carcinomas, while changes in ST6GAL1 levels are linked to tumor invasiveness. Specifically, enzymes like xylosyltransferase I (XT-I) initiate glycosaminoglycan (GAG) chain synthesis on core proteins, a process critical for the assembly of the extracellular matrix (ECM). Accurate detection using a specific glycosyltransferase antibody is crucial for deciphering these mechanisms.
Key Glycosyltransferase Targets in Cancer
| Target Enzyme | Primary Biological Function | Disease Association |
|---|---|---|
| FUT8 | Catalyzes core fucosylation of N-glycans (alpha-1,6 linkage) | Promotes tumor growth and metastasis in breast, lung, and prostate cancers. |
| ST6GAL1 | Adds alpha-2,6-sialic acid to N-glycans | Associated with stemness, drug resistance, and invasive potential in carcinomas. |
| XT-I | Initiates GAG synthesis on proteoglycans | Dysregulated in fibrosis and altered ECM composition in tumors like adenoid cystic carcinoma. |
| MGAT5 | Synthesizes beta-1,6-GlcNAc branched N-glycans | Enhances growth factor signaling and promotes metastasis. |
Biosynthesis Control in the Tumor Microenvironment (TME)
In tumors such as salivary gland adenoid cystic carcinoma, the altered biosynthesis of proteoglycans by neoplastic myoepithelial cells is a key area of investigation. The remodeling of the TME is often driven by changes in the expression of enzymes like xylosyltransferases and core fucosyltransferases. Developing a robust anti-glycan enzyme antibody toolset allows researchers to perform precise expression profiling via Western blot and immunohistochemistry, distinguishing tumor-specific glycan signatures from healthy tissue.
Pain Points in Anti-GT Antibody Discovery
Developing reliable antibodies against glycosyltransferases presents unique challenges that often hinder research progress:
High Homology
GT families share significant sequence similarity within catalytic domains (e.g., between ST3GAL antibody and ST6GAL1 antibody targets), leading to frequent cross-reactivity in standard polyclonal preparations.
Low Immunogenicity
Many GTs are evolutionarily conserved, making them poor immunogens in standard host species, resulting in low-titer or low-affinity antibodies.
Isoform Specificity
Distinguishing between functionally distinct isoforms requires precise epitope mapping to non-conserved regions, which is often overlooked in bulk peptide generation.
Complex Localization
Most GTs are Golgi-resident type II membrane proteins. Antibodies must be validated to recognize the enzyme in its native cellular context (IF) or denatured state (WB) without non-specific background from cytoplasmic proteins.
Our Custom Development Capabilities
We provide a comprehensive suite of development strategies designed to overcome the inherent challenges of targeting glycosyltransferases. Instead of a one-size-fits-all approach, we tailor the antigen design and host selection to the specific structural properties of your target enzyme.
Anti-Sialyltransferase Antibody Development
Comprehensive antibody generation for ST3GAL, ST6GAL, and ST8SIA families. Essential for studying hypersialylation patterns involved in tumor metastasis and immune evasion.
Anti-Fucosyltransferase Antibody Development
Specialized development for FUT1, FUT2, FUT4, and FUT8. These antibodies are critical for analyzing core fucosylation and Lewis antigen biosynthesis in cancer and inflammation.
Anti-GlcNAc-transferase (GnT) Antibody Development
Targeting the N-acetylglucosaminyltransferases (MGATs/GnTs) that initiate N-glycan branching. Key tools for investigating the structural alteration of glycans in malignancy.
Anti-Galactosyltransferase (GalT) Antibody Development
Antibodies against beta-1,4-GalT and other family members. Facilitates the study of LacNAc synthesis, O-glycan elongation, and galactosylation levels in various tissues.
Technical Highlights & Advantages
Epitope Precision
Advanced bioinformatics to select isoform-unique peptide sequences, minimizing cross-reactivity between ST antibody family members.
Diverse Host Systems
From rabbits for high-affinity polyclonal antibodies to mouse/rat hybridomas for monoclonal consistency.
Rigorous Validation
Validated by WB, IHC, and ELISA using positive control lysates to guarantee performance.
One-Stop Solution
From polysialyltransferase antibody antigen synthesis to final purification and labeling services.
Start Your Project
Whether you need an anti-FUT antibody for core fucosylation analysis or an anti-sialyltransferase antibody for studying tumor metastasis, our team is ready to assist. We begin with a consultation to understand your target enzyme, desired host species, and intended applications.
Inquire About Anti-GT Antibodies
Published Data
Glycosyltransferases are pivotal regulators of tumor malignancy, yet their specific expression patterns and functional roles in clinical progression often remain under-characterized. A comprehensive 2025 study utilized high-specificity antibodies to investigate the clinical relevance of alpha-1,6-fucosyltransferase (FUT8) in prostate cancer. By analyzing expression levels across multiple independent patient cohorts comprising over 1500 clinical samples, the researchers established that FUT8 is significantly upregulated in high-grade and metastatic tumors compared to localized disease or benign hyperplasia.
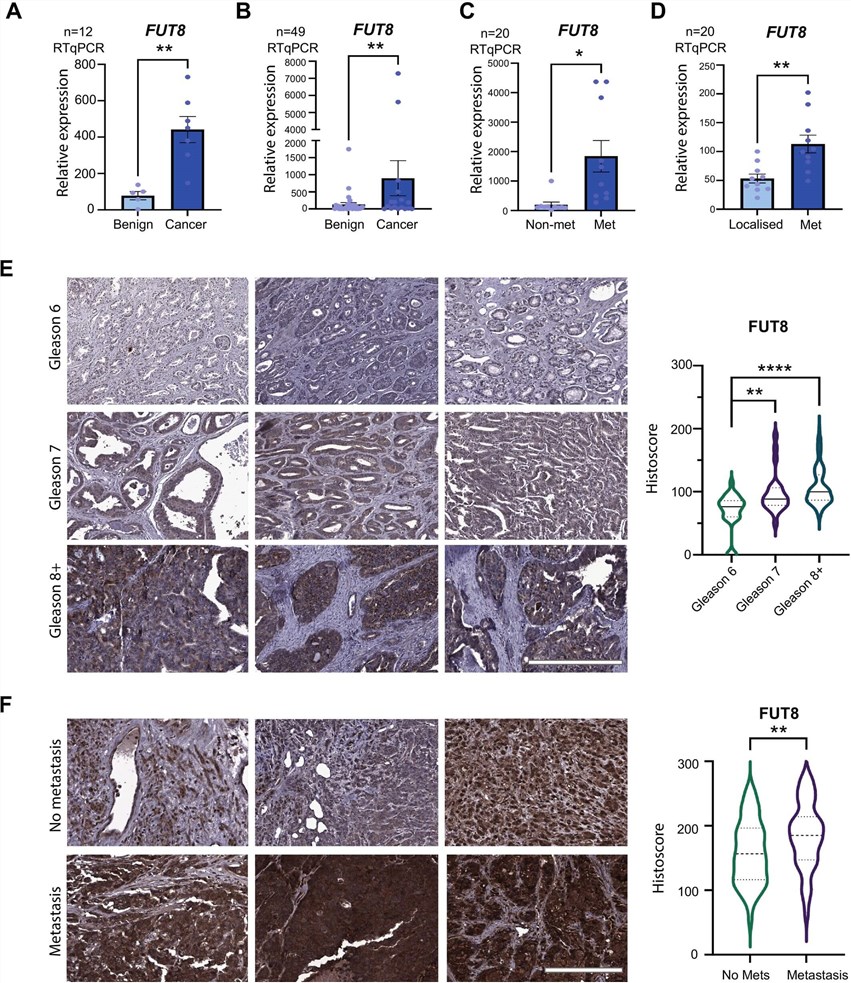 Fig.1
Immunohistochemical analysis reveals significant upregulation of FUT8 protein in high-grade prostate cancer tissue microarrays.1
Fig.1
Immunohistochemical analysis reveals significant upregulation of FUT8 protein in high-grade prostate cancer tissue microarrays.1
Crucially, the study employed immunohistochemistry (IHC) on tissue microarrays to map the spatial distribution of the enzyme. The antibody-based detection revealed a robust correlation between intense FUT8 protein expression and aggressive disease phenotypes, including higher Gleason scores. Complementary analyses using lectin-based immunofluorescence and mass spectrometry imaging confirmed that this upregulation leads to aberrant core fucosylation of N-glycans on the cell surface. Functionally, the research demonstrated that FUT8 drives tumor growth, cell motility, and invasion in vivo, as knockdown of the enzyme significantly suppressed tumor burden in xenograft models. These data validate FUT8 as a critical driver of disease progression and highlight the essential role of specific anti-glycosyltransferase antibodies in identifying prognostic biomarkers and unravelling the complex glycobiology of the tumor microenvironment.
FAQs
Can you generate antibodies against specific CAZyme families beyond mammalian systems?
Absolutely. Our platform supports the development of CAZyme antibody tools for enzymes from various organisms, including plants, bacteria, and fungi, which are often used in glycobiotechnology.
How do you ensure the antibody distinguishes between ST3GAL and ST6GAL families?
We utilize bioinformatics to identify non-conserved regions in the sialyltransferase antibody targets. We then synthesize peptides corresponding to these unique sequences for immunization and perform negative screening against the homologous isoforms to eliminate cross-reactive clones.
Is it possible to detect expression of low-abundance enzymes like XT-I in tumor samples?
Yes. For low-abundance targets like Xylosyltransferase I, we recommend our high-affinity rabbit polyclonal antibody service or our recombinant antibody development platform. These approaches yield antibodies with superior sensitivity, suitable for detecting endogenous glycosyltransferase expression antibody signals in lysates from tissues like salivary gland adenoid cystic carcinoma.
Do you offer antibodies for B3GNT and MGAT family members?
Yes, we have extensive experience targeting branching enzymes. We can develop B3GNT antibody and MGAT antibody tools specifically validated for analyzing glycan branching patterns associated with tumor malignancy.
Can you produce antibodies targeting the catalytic domain specifically?
Yes, we can design antigens based on the catalytic domain structure to generate antibodies that may neutralize enzyme activity, which is useful for functional inhibition studies.
What Our Customers Say
"We required a specific antibody for XT-I to study proteoglycan synthesis in our carcinoma models. The polyclonal antibody Creative Biolabs generated was excellent for Western blot, showing a clean band at the expected molecular weight with no cross-reactivity."
"The anti-FUT8 antibody we ordered has been instrumental in our core fucosylation projects. It works perfectly in both IHC and WB. The team was very helpful in selecting the right immunogen strategy to avoid homology issues."
Reference:
- Bastian, K., Orozco-Moreno, M., Thomas, H. et al. "FUT8 is a critical driver of prostate tumour growth and can be targeted using fucosylation inhibitors." Cancer Medicine 14 (2025): e70959. Distributed under Open Access license CC BY 4.0, without modification. https://doi.org/10.1002/cam4.70959
Supports
- Glycosyltransferase & Glycosidase Substrate Microarray
- Glycosylation Analysis
- Custom Glycosylation of Biomolecules
